
Review Article
Austin J Anat. 2017; 4(1): 1064.
Haemolytic Uraemic Syndrome (HUS): Clinical Medicine Versus Clinical Anatomy
Muhammad SN¹*, Abdel Meguid E²* and Robert Novo’s³
¹Specialist Biomedical Scientist, Co-Founder and Chief in Research, Renal Patient Support Group (RPSG), UK
²Centre for Biomedical Sciences Education, School of Medicine, Dentistry and Biomedical Sciences, Queen’s University Belfast, UK
³Hospital Practitioner, Paediatric Nephrologic Unit, University Hospital, Lille, France
*Corresponding author: Muhammad SN, Specialist Biomedical Scientist, Co-Founder and Chief in Research, Renal Patient Support Group (RPSG), England, UK
Received: January 16, 2017; Accepted: February 13, 2017; Published: February 16, 2017
Abstract
Haemolytic Uraemic Syndrome (HUS) is an acquired disorder affecting mainly infants and children. The triad of this clinical syndrome is defined by: 1) Thrombotic or Microangiopathic Haemolytic Anaemia with schistocytes 2) Thrombocytopenia and 3) Acute Renal Failure (ARF) which can develop into Chronic Kidney Disease (CKD). The aim of this article is to provide an editorial/ commentary on the Clinical Medicine versus Clinical Anatomy of HUS.
HUS is the most common cause of Acute Renal Failure (ARF) in children with an equal sex incidence [1]. The annual incidence of VTEC infection varies geographically; it can range from 1 to 30 cases per 100,000 in industrialized countries. It is a rare syndrome post-puberty but it is also closely related to Thrombotic Thrombocytopenia (TTP) which is common in adults. The annual incidence of the Verocytotoxin-producing Esherichia Coli (VTEC) infection varies geographically from year to year, ranging from 1-30 cases per 100,000 in industrialized countries and is associated with HUS. HUS occurs in sporadic cases epidemics; between 1st January [2] and 31st December [2] in England, a total of 3717 cases were reported with evidence of Shiga Toxin-Producing E. Coli (STEC) infection; sometimes following outbreaks. In Hamburg [3], there was an outbreak with more than 900 cases. The disease has seasonal variation, being more common in the warmer months in children.
Renal histopathology is characterized by abnormal morphology applicable to afferent arterioles and glomeruli. The glomeruli show evidence of global sclerosis and glomerular thrombotic microangiopathy endothelial cell swelling; capillary wall thickening and glomerular basement membranes also evident. Interstitial fibro edematous change and tubular atrophy are marked. Arterial, arteriolar and capillary lumina are narrow with obstruction and intimal thickening. The nature of vascular involvement in the kidneys supports the hypothesis that HUS is mediated by systemic toxemia and endothelial cells are the primary target cells owing to action of Verocytotoxin. Histopathological findings provide clues not only to the diagnosis but also in the support of prognosis. Diffuse tubular interstitial change and global sclerosis indicate the degree of blood flow obstruction and prognosis. Renal blood flow obstruction caused by diffused arterial and arteriolar luminal stenosis may lead to irreversible changes in renal pathology
Keywords: Haemolytic Uraemic Syndrome (HUS); Clinical medicine; Clinical anatomy; Chronic kidney disease; Acute renal failure; Haematology
Introduction
Haemolytic Uraemic Syndrome (HUS) occurs due to Shigalike toxin activity via aberrant complement activation. HUS is typically classified into two primary types: 1) HUS due to infections, often associated with diarrohea (D+HUS, Shiga toxin-producing Escherichia Coli-HUS), with the rare exception of HUS due to a severe disseminated infection caused by Streptococcus; 2) HUS related to complement, such HUS is also known as “atypical HUS” and is not diarrohea associated (D-HUS, aHUS) [4]. Clinical features include proteinuria, renal impairment and history of E. coli diarrohea (hallmark of typical HUS). Encephalopathy is rare but can cause death. HUS is seen increasingly following outbreaks of infection with Verotoxin (VT)-producing organisms. It represents a growing public health problem and data suggest that more awareness of specific micro-organisms causing diarrhoea, (and those thus leading to HUS and/ or HUS related symptoms) may become more important in future health consultations [3].
E.coli 0157:H7 is the most commonly notified VT-producing organism in the UK and France. Clinical manifestations may vary from an asymptomatic infection to bloody diarrhoea, haemorrhagic colitis and HUS. Verotoxin Enterococcal (VTEC)-associated HUS was seen in up to 20% of patients in recent outbreaks, mainly affecting children [5]. Table 1 summarizes the defining classification of HUS and (Table 2) summarizes features of HUS, respectively.
HUS - Classifications
Typical
Infection related
Shiga toxin producing E. coli/ Shigella
Pneumococcal infection
HIV
Other viral or bacterial infections
Atypical
Complement factor abnormality
Factor H deficiency and Factor I deficiency
Miscellaneous
Cumulative Trauma Disorder (CTD), drugs, malignancy
Table 1: Classification of HUS.
HUS - Defining Features
- Renal involvement
- Evidence of coagulopathy –intravascular coagulation in the kidney
- Antipathy - marked fragmentation of red cells
- The disorder is associated with activation of neutrophils
- Typical HUS is secondary to GI infection with Verocytotoxin-producing E-coli 0157:H7 (VTEC), less often Shigella
- Complement activation used to attack foreign bodies
- Complement system highly regulated to prevent it from damaging healthy tissues/ organs
- Platelet activation, damage to endothelial cells (cells that line the blood vessels), white blood cell activation causing haemolysis
- Thrombotic Microangiopathy (TMA) – formation of blood clots in small vessels throughout the body and over time, causing multiple organ damage
Table adopted from (Blaser 2004; Licht et al. 2009)
Table 2: Defining features of HUS.
HUS epidemiology
HUS is the most common cause of Acute Renal Failure (ARF) in children with an equal sex incidence [1]. The annual incidence of VTEC infection varies geographically; it can range from 1 to 30 cases per 100,000 in industrialized countries. There is seasonal variation, thus being more common in warmer months. It is more common in those under 5-years.
Between 1st January [6] and 31st December [6] in England, a total of 3717 cases were reported with evidence of Shiga Toxin-producing E. Coli (STEC) infection, and the crude incidence of STEC infection was 1•80/100 000 person-years. Incidence was highest in children aged 1-4 years (7•63/100 000 person-years) [2].
HUS diarrohea association
Most common cause of HUS and intrinsic ARF in paediatrics in the UK, France and USA industrialized countries is diarrhoea. There are no specific therapies to treat the diarrohea. Mortality is as high as 8.5 % and up to 30 % of survivors may develop further Glomerular Filtration Rate (GFR) impairment or albuminuria [6].
Use of anti-motility drugs may increase the risk of developing HUS and it is a rare syndrome post-puberty [1]. Atypical HUS (aHUS) can be inherited or acquired and does not appear to vary by race, gender or geographic area. Data on the prevalence of aHUS is limited [7,8] (Table 3).
HUS - Signs
- Patients present with anaemia
- Petechiae, purpura, and fever are common
- GI bleeding is often found and a bloody diarrohea
- GI disease may be severe with haemorrhagic colitis, toxic megacolon, rectal prolapse, and bowel necrosis
- Neurological symptoms
- Cardiac involvement may lead to Congestive Heart Failure (CHF) and arrhythmias
- Microinfarcts in the pancreas (pancreatitis) or, rarely, Insulin Dependent Diabetes Mellitus (IDDM)
(Table adopted from Licht et al., 2009)
Table 3: HUS signs.
HUS and laboratory tests/ investigations
Haemolysis and red cell fragmentation are usually evident at presentation, although this rarely develops later in the disease even after platelet count improvement (Table 4). Coagulation studies are usually normal with mildly raised D-dimer titres in contrast to Disseminated Intravascular Coagulation (DIC) [9]. The Von- Willebrand Factor (VWF) levels are usually markedly raised during acute illness while analysis may/ may not show ultra-large multimers [10]. Poor prognostic features at presentation include a high neutrophil count [11]. Severe thrombocytopenia is uncommon but prolonged thrombocytopenia for more than 10 days is associated with long-term renal picture. Factor VIII levels do not correlate with clinical outcome [12]. Table 5 summarizes laboratory tests/ investigations according to Haematology specialty. Figure 1 depicts HUS red cell morphology under microscopy.
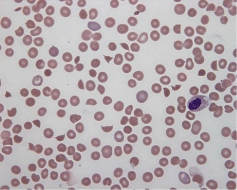
Figure 1: HUS red cell morphology under microscopy. RBC fragmentation
with obvious schistocytes/ helmet cells. Note decrease in platelets which is a
typical picture in HUS patients. Schistocytes are irregularly shaped fragments
with two pointed ends without central pallor (Barcellini & Fattizzo 2015).
Slide taken from (https://emedicine.medscape.com/article/779218-workup)
(accessed December 2016).
HUS - Symptoms
- The kidneys are swollen and pale
- Many flea-bite hemorrhages are on the surface
- GI involvement may lead to symptoms of an acute abdomen, with occasional perforation
- Hypertension
- Anuria – Oliguria, depending on overall GFR/ renal function
(Table adopted from Licht et al., 2009)
Table 4: HUS symptoms.
HUS - Haematology
Full Blood Count (FBC) highlights an anaemic picture on presentation
Coagulation highlights APTT and PT are both normal - suggests sepsis rather than HUS/TTP
Platelet and fibrin micro-thrombi is evident within the renal microvasculature
Thrombocytopenia is universal at some point in the illness
Haematinics highlight there is variability in Iron titres
Direct Coombes Test is Negative
Plasma Haptoglobin levels is decreased owing to Red Blood Cell (RBC) breakdown/ degradation
Raised Fraction Degradation Products (FDPs)
There is a slightly elevated D-Dimer
There may be microcytosis
Red cell enzymes and osmotic fragility are normal
Reticulocyte count is elevated
(Table adapted from Taylor et al., 1999; Taylor 2001a; Taylor 2001b; Taylor et al., 2004)
Table 5: HUS – Haematology.
Prognosis and treatment
Treatment is by supportive measure, and the main treatments are either Haemodialysis (HD) or Peritoneal Dialysis (PD) because of the ARF. The prognosis is dependent upon the aetiology and renal function; there is a poor prognosis if patient requires HD or PD more than 7 days, but good prognosis is conceivable with total recovery of renal function with no arterial hypertension, and no proteinuria. Children with intra-cerebral involvement may be treated with plasma exchange, but efficacy and long-term use is not advisable [13-15].
Major neurological dysfunction occurs in a third of patients in atypical HUS and less than 10% in typical HUS; is associated with a poor prognosis [7]. There should be supportive information made available for the patient and parent or career/guardian. Eculizumab is a monoclonal antibody that binds to C5 to prevent the formation of C5a and the membrane attack complex [7,8]. This treatment has become popular and can be helpful for patients who may have extra renal involvement such as that seen in typical HUS. Its efficacy and safety in the treatment of aHUS has been highlighted recently [7,8].
Management
Either HD or PD treatments is essential when renal function is altered. It is crucial to control hypertension, thus preventing any longer term complications (Figures 2 and 3). RBC transfusion in children is common owing to anaemia [13-15]. Fraction Frozen Plasma (FFP) has been administered in adults [8].
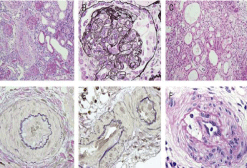
Figure 2: Histological Stains of a Kidney Biopsy Specimen – Day 37 following
Eculizumab Treatment characteristic of a HUS. (A) Almost all glomeruli give
evidence of global sclerosis or collapse (Periodic Acid Schiff stain [PAS];
×100). (B) Endothelial swelling is conspicuous and glomerular basement
membranes are focally duplicated (Periodic Acid Methenamine silver stain
[PAM]; ×1000). (C) Interstitial fibroedematous change and tubular atrophy
are marked (Haematoxylin and Eosin stain [H&E]; ×200). (D, E) Arterial and
arteriolar lumina stenosed or obstructed with intimal thickening (Elastica van
Gieson stain [EVG]; ×1000). (F) Mononuclear cells infiltrated into intima of
arteries (PASX1000). These findings are helpful demonstrating the diagnosis
of TMA diagnosis (Slide stain taken from Okuda et al. 2015).

Figure 3: HUS - fibrin histological stain. Fibrin histological stain highlighting
platelet-fibrin thrombi (red) in the glomerular capillaries, characteristic
of HUS/microangiopathic disorder, (taken from https://slideplayer.com/
slide/11018533/) (accessed December 2016).
Research perspectives
Haemoglobin (Hb) is the most direct indicator of clinical severity in haemolytic diseases. Its level may be close to normal values in mild forms (Hb >10 g/dL) or reduced in moderate (Hb 8–10 g/dL), severe (Hb 6–8 g/dL), and very severe (Hb 6 g/dL) forms (WHO 1989). In a differential diagnosis, an acute onset is more frequently observed in RBC enzymopathies involving the Pentose Phosphate (PP) shunt (e.g. glucose-6-phosphate-dehydrogenase, G6PD deficit) and in autoimmune haemolytic forms involving complement activation Autoimmune Haemolytic Anaemia (AIHA) caused by warm IgM, warm IgG + C, mixed, and Cold Agglutin Disease (CAD) (with thermal range close to physiological temperatures) and in Paroxysmal Nocturnal Haemoglobinuria (PNH) (Tables 6,7 and 8).
HUS – Key Morphological Features
Peripheral blood smear depicts striking red cell fragmentation
Findings of microangiopathic haemolytic anaemia
Deformed, irregular or helmet-shaped RBCs – schistocytes
Thrombocytopenia (counts of 50 x 109/l) or less
Neutrophil may be quite marked – it is a marker of adverse prognosis
Polychromasia (greyish tinge) may be marked
Bone Marrow is cellular – response to haemolysis/ red cell breakdown
(Table adapted from Taylor et al., 1999; Taylor 2001a; Taylor 2001b; Taylor et al., 2004)
Table 6: HUS key haematology morphological features.
HUS - Chemistry
Urea, Creatinine and Electrolytes demonstrate abnormally high results
Urine, if present, may contain protein and RBCs
Liver Function Test (LFTS) can be normal
Urine dipstick can show some protein leaking (24hr urine collection needed to quantify)
Electrophoresis can be helpful and important to establish extent of protein leaking
Blood Urea Nitrogen (BUN) is markedly elevated
HUS - Microbiology
Cultured species of E-coli is performed
Mainly looking for group Enterohaemarrohagic E-Coli (EHEC)
(Table adapted from Taylor et al., 1999; Taylor 2001a; Taylor 2001b; Taylor et al., 2004)
Table 7: HUS– chemistry/ microbiology.
HUS – Key Histopathological Features
Afferent arterioles and glomeruli inflammation
Ischaemic necrosis in renal cortex may occur from intravascular coagulation
Glomerular thrombosis and sclerosis
Hypercellularity
Sclerotic tuft
Fibrinous necrosis in places and some tubule-interstitial compartments show atrophic changes with infiltration of the inflammatory cells
Endocapillary swelling with diffuse arteriolar and arterial luminal stenosis due to the thickness and sclerotic changes of the media and intima
Renal Scarring
(Table adapted from Taylor et al., 1999; Taylor 2001a; Taylor 2001b; Taylor et al., 2004; Okuda et al., 2016)
Table 8: HUS key anatomical/histopathological features.
Summary
Clinical medicine
- Bloody’ diarrohea still most crucial element to be aware of in young patients
- Renal Function over time must be monitored
- Haemolytic parameters may be differently altered in the various conditions thus helping the differential diagnosis and coagulation must be monitored
- LDH is useful in evaluating response to treatment, as its level decreases along with the reduction of the haemolytic rate
- Polynucleosis is a factor of poor prognosis
- Glomerulus: hyalinized, thickened and sometimes split capillary walls due to endothelial swelling
- Tubules: atrophied and contains hyalinised casts
- Interstitium: fibrosed with lymphocystic infiltration
- Vessels: arterial and arteriolar sclerosis, intimal hyperplasia and fibrinoid necrosis with large deposits of fibrin-related materials in the capillary lumen. They are often occluded by thrombi.
Clinical anatomy/histopathologyg
Acknowledgement
This article is dedicated in loving memory to a dear friend Emma Marquick (1975 to 2003) who went through the whole HUS triad – Fourteen years on a Haemodialysis protocol and three transplants is what took her life.
References
- Sherbotie JR, Van HV, Axton R, Williamson K, Finn LS. Hemolytic uremic syndrome associated with Denys-Drash syndrome. Pediatr Nephrol. 2014; 12: 1092-1097.
- Byrne L, Jenkins C, Launders N, Elson R, Adak GK. The epidemiology, microbiology and clinical impact of Shiga toxin-producing Escherichia coli in England, 2009-2012. Epidemiol Infect. 2005; 143: 3475-3487.
- Schulz C, Schutte K, Jacobi CA, Hulsemann JL, Malfertheiner P. TV news and concerns about - the EHEC-outbreak 2011 in Germany. Z Gastroenterol. 2014; 52: 277-280.
- Talarico V, Aloe M, Monzani A, Miniero R, Bona G. Hemolytic uremic syndrome in children. Minerva Pediatr. 2016; 68: 441-455.
- Dundas S, Todd WT, Stewart AI, Murdoch PS, Chaudhuri AK, Hutchinson SJ. The Central Scotland 1996 Eschericia coli 0157 outbreak: predicotors of haemolytic uraemic. Syndrome⁄ thrombotic thrombocytopenic purpura and death. Clinical Infectious Diseases. 2001; 33: 923–931.
- Licht C, Pluthero FG, Li L, Christensen H, Habbig S, Hoppe B, et al. Plateletassociated complement factor H in healthy persons and patients with atypical HUS. Blood. 2009; 114: 4538-4545.
- Okuda Y, Ishikura K, Hamada R, Harada R, Sakai T, Hamasaki Y, et al. Membranoproliferative glomerulonephritis and C3. Glomerulonephritis: frequency, clinical features, and outcome in children. Nephrology. Carlton. 2015; 20: 286-292.
- Okuda Y, Ishikura K, Terano C, Harada R, Hamada R, Hataya H, et al. Irreversible severe kidney injury and anuria in a 3-month-old girl with atypical haemolytic uraemic syndrome under administration of eculizumab. Nephrology Carlton. 2006; 21: 261-265.
- Rose P, Chant I. Hematology of hemolytic–uremic syndrome. In: Eschericia coli 0157:H7 and other Shiga Toxin-Producing E. coli Strains (ed. by J.B. Kaper & A.D. O’Brien), p. 293. American Society for Microbiology, Washington. 1998.
- Rose PE, Enayat SM, Sunderland R, Short PE, Williams CE, Hill FGH. Abnormalities of factor VIII related protein multimers in the haemolytic uraemic syndrome. Archives of Disease in Childhood. 1984; 59: 1135–1140.
- Walters MD, Matthei IU, Kay R, Dillon MJ, Barratt TM. The polymorphonuclear leucocyte count in childhood haemolytic uraemic syndrome. Pediatric Nephrology. 1989; 3: 130–134.
- Milford D, Staten J, Mac Greggor I, Dawes J, Taylor CM, Hill FG. Prognostic markers in diarrhoea associated haemolytic uraemic syndrome: initial neutrophil count, neutrophil elastase and von Willebrand antigen. Nephrology, Dialysis and Transplantation. 1991; 6: 232–237.
- Taylor CM, Williams JM, Lote CJ, Howie AJ, Thewles A, Wood JA, et al. A laboratory model of toxin-induced hemolytic uremic syndrome. Kidney Int. 1999; 55: 1367-137.
- Taylor CM. Complement factor H and the haemolytic uraemic syndrome. Lancet. 2001; 358: 1200-1202.
- Taylor CM, Chua C, Howie AJ, Risdon RA. Clinico-pathological findings in diarrhoea-negative haemolytic uraemic syndrome. Pediatr Nephrol. 2004; 19: 419-425.