
Research Article
Austin Biochem.2016; 1(1): 1003.
Partial Characterization of an Interaction between Kinesin Associated Protein 3 (Kap3) of Kinesin-2 and the Actin Cytoskeleton
Tenney A, Price M, Macatangay J, Hatt D, Kline T and Berezuk MA*
Department of Biology and Chemistry, Azusa Pacific University, USA
*Corresponding author: Berezuk MA, Department of Biology and Chemistry, Azusa Pacific University, 675 E Foothill Blvd, Azusa, CA 91702, USA
Received: September 29, 2016; Accepted: November 10, 2016; Published: November 14, 2016
Abstract
Kinesin-2 is a plus-end directed microtubule based motor involved in the transport of diverse intracellular cargoes. It has been specifically shown to function in systems that help define sub cellular structures and cell morphology in general. The non-motor accessory subunit of kinesin-2, KAP3, is proposed to act as a multifunctional linker between the motor subunits and an assortment of target cargoes. Human KAP3 contains an array of eleven armadillo repeats, a well characterized motif common to various protein-protein interactions. Previous work showed that native kinesin-2 and bacterially expressed KAP3 armadillo repeat bearing fragments can co-cycle with actin filaments in vitro. However, these studies left the putative KAP3-actin interaction poorly characterized and did not address what in vivo significance, if any, a KAP3- actin interaction might have. Here we report that KAP3 associates with actin polymers exclusively through its central core of armadillo repeats. Conversely, KAP3 appears incapable of binding to monomeric, globular actin in solution. Co-localization of over expressed GFP-tagged full length KAP3 with phalloidin stained actin filaments in cell culture substantiates an in vivo consequence to our biochemical description. This work helps to refine our understanding of the KAP3-actin interaction in vitro and expands it to a cellular context. Thus, providing further insight into how KAP3 facilitates kinesin-2 transport activities, specifically those related to the formation and maintenance of specific cellular processes and cell shape.
Keywords: Kinesin-2; Actin; Cytoskeleton
Introduction
Kinesins are a class of predominantly plus-end directed, microtubule-based motors capable of transforming the energy generated from ATP hydrolysis into mechanical force for intracellular transport [1-6]. While all kinesins share nearly identical motor domains, their non-motor components and accessory subunits are quite varied [4,7-11]. Kinesin-2 is one of the plus-end directed members of the kinesin super family and is essential for metazoan life [9,12]. It is unique amongst kinesin motors in that it consists of two configurations of heterologous motor subunits (kinesin-2 A/B or A/C) and a non-motor, accessory protein, KAP3 [13-15]. The kinesin-2 holoenzyme has been shown to function in the intracellular transport of organelles, viruses and RNAs as well as having roles in the regulation of cell polarity, cell-cell adhesion, mitosis, neurite extension, intraflagellar transport and cilium assembly/maintenance [9,16-18]. However, the exact mechanisms of these activities and how kinesin-2 associates with the myriad of cargoes involved is poorly understood. Most studies implicate the KAP3 subunit of kinesin-2 as the likely mediator of target molecule/organelle recognition and attachment to the motor subunits [2,19-29]. While promising, these reports have only identified a small proportion of the potentially large number of cargo associating molecules present for KAP3 and kinesin-2 to bind within the cell.
Structurally, KAP3 contains a central grouping of armadillo repeats [8,14,30], a common protein interaction motif often found in proteins that associate with the actin cytoskeleton [31-36]. Previous work using His-tagged KAP3 fragments identified an association between the armadillo repeats of KAP3 and actin [27]. This novel finding provided us with a model for kinesin-2 recognizing a specific cargo in the context of a known functional interaction [22,37]. Here we report studies which confirm that the armadillo repeat regions of KAP3 are capable of co-cycling with actin filaments in vitro. However, we are unable to detect any direct interaction between these same fragments and globular actin. To address the cellular significance of the observed biochemical interaction, we cloned a Green Fluorescent Protein (GFP) fusion construct of full length human KAP3 into a mammalian expression vector. Transient transfection and overexpression in COS-7 cell culture coupled with fluorescence staining of actin filaments revealed specific co-localization between GFP-KAP3 and filamentous actin. Our continued characterization of this novel interaction furthers our understanding of how these distinct cytoskeletal systems can communicate and potentially regulate one another in performing a variety of cellular functions.
Materials and Methods
His-tagged protein expression and purification
KAP3 fragment constructs cloned into the His-tagged pRSET B bacterially expression vector [27] were a generous gift from T.A. Schroer. The coding region for KAP3 included in each clone corresponds to the N-terminus [KAP3 N; a.a. 1-161], armadillo repeats 1-3 (KAP3 A1-3; a.a 162-333), 4-8 (KAP3 A4-8; aa. 334-534), 9-11 (KAP3 A9-11; aa. 535 -662) and the C terminus (KAP3 C; aa. 663-772) of human KAP3 (Figure 1). Plasmids were transformed into Rosetta (DE3) competent cells (EMD Millipore, Billerica, MA). Cultures were grown and protein expression induced with 0.1 μM IPTG for 16 hours at 37 C. Cells were lysed by sonication and sterile filtrate loaded onto a HiTrap™ Chelating HP column (GE Healthcare, Marlborough, MA) for binding (binding buffer; 20 mM Tris-HCl pH 7.5, 500 mM NaCl, 10 mM imidazole) and elution (0 to 100% gradient of elution buffer; 20 mM Tris-HCl pH 7.5, 500 mM NaCl, 500 mM imidazole) using an AKTA®purifier FPLC system (GE Healthcare, Marlborough, MA).
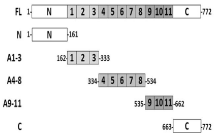
Figure 1: Schematic of full length human KAP3 protein (FL) and cloned
fragments used. Amino acid numbers defining the sequence for each
construct are as labelled. Amino terminus (N), carboxyl terminus (C) and
armadillo repeats (numbered 1 – 11) within the sequence are as shown.
Actin co-cycling
Actin co-cycling experiments were performed as previously described [27]. Briefly, human platelet actin (Cytoskeleton Inc., Denver, CO) and His-tagged KAP3 fragment samples were made to 1 mg/ml and 0.25 mg/ml, respectively, in G-buffer (5 mM Tris- Cl pH 8.0, 0.2 mM NaCl, 0.2 mM ATP, 0.5 mM DTT). Actin was polymerized by addition of 10 X F-buffer (final concentration: 5 mM Tris-Cl pH 8.0, 2 mM MgCl2, 5 mM NaCl, 1 mM ATP, 0.5 mM DTT) and incubated for 4 hours at 30°C. Polymerized F-actin was incubated with His-tagged KAP3 (final ratio 30:1 m/m) for 30 minutes at 30 C. Samples were centrifuged at 150,000 g for 90 minutes at 30°C. Supernatants were saved. Pellets were resuspended in G-buffer and incubated overnight on ice. Samples were spun at 150,000 g for 90 minutes at 4°C. Supernatants were removed, supplemented with 10X F-buffer and incubated for actin polymerization as before. Pellets were resuspended in G-buffer and saved. Samples were spun at 150,000 g for 90 minutes at 30°C, supernatants were saved and pellets resuspended in G-buffer.
Actin affinity pulls down
A 50μl suspension of BioMag Streptavadin (Qiagen, Inc., Valencia, CA) was incubated with 10μg biotinylated actin protein (Cytoskeleton, Inc., Denver, CO) in 500μl G-buffer for 1 hour at room temperature. Beads were separated using a Qiagen 12-tube magnet (Qiagen, Inc., Valencia, CA) and washed with binding buffer (20 mM Tris, pH 7.5, 250 mM NaCl, 10 mM imidazole). Test proteins (25μg each, supplemented with 25μg bovine serum albumin (Sigma-Aldrich, St. Louis, MO) carrier protein) in 500μl binding buffer were added and incubated overnight at 4°C. Beads were separated and washed with binding buffer. Beads were resuspended in 1X SDS-PAGE sample buffer for analysis.
SDS-PAGE, Coomassie staining and immunoblotting
SDS-PAGE was performed as described [38]. Coomassie staining of SDS-PAGE gels was performed with Coomassie Brilliant Blue G-250 (Bio-Rad Laboratories Inc., Hercules, CA). Western transfer and immunoblotting were performed according to the Mini Trans- Blot and Clarity Western ECL detection kit protocols (Bio-Rad Laboratories Inc., Hercules, CA). Blots were visualized using a ChemiDoc XRS+ imaging system (Bio-Rad Laboratories, Hercules, CA). Primary antibody used was anti-Xpress™ mAb (Invitrogen, Carlsbad, CA) Secondary antibody used was HRP conjugated goat anti-mouse IgG (Life Technologies, Carlsbad, CA).
Cloning of GFP-tagged Full Length KAP3 Construct
Full-length human KAP3 DNA (coding region a.a. 1-772, (Figure 1)) was obtained from restriction digest of the KAP3 pRSET B vector clone previously described [27] using XhoI and EcoRI cut sites. Gel purified restriction fragment was ligated into the pAcGFP1-C2 mammalian expression vector (Clonetech Laboratories Inc., Mountain View, CA) and transformed into JM109 competent cells (Promega Corporation, Madison, WI) for selection. Purified plasmid construct (GFP-KAP3 FL) was confirmed by restriction digest and DNA sequencing.
Cell Culture, Transfection and Cytochemistry
COS-7 cells were grown on sterile glass coverslips (Neuvitro Corporation, El Monte, CA) and maintained in Dubellco’s Modified Eagles Media (DMEM) (Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum (FBS) (Invitrogen, Carlsbad, CA). Transfection was performed per manufacturer’s protocol using Polyfect® reagent (Qiagen, Valencia, CA) [39]. Filamentous actin staining was performed 48 hours post-transfection according to protocol with rhodamine-Phallacidin (Sigma-Aldrich, St. Louis, MO). Cover slips were mounted in ProLong Diamond Antifade Mountant with DAPI (Life Technologies, Carlsbad, CA).
Microscopy
Cells were observed by fluorescent microscopy using a Zeiss LSM 700 confocal microscope (Carl Zeiss Inc., Thornwood, NY). Still frame images in the fluorescent green (GFP) red (Alexa Fluor 568) and blue (DAPI) channels were imaged and overlayed using Zeiss- ZEN software (Carl Zeiss Inc., Thornwood, NY).
Results and Discussion
To date, most interactions identified between kinesin-2 and putative cargoes have involved its non-motor accessory protein, KAP3 [9,19-28,40]. Sequence analysis of KAP3 across a variety of species reveals a central stretch of armadillo repeats [13,14]. The armadillo repeat itself is a common motif employed in a variety of protein-protein associations [31,32]. Many armadillo repeat proteins, including the canonical Armadillo protein, are known to associate with the actin cytoskeleton [31-36]. Taken together, these particulars make it logical to infer that KAP3 may be an actin binding protein.
Previous work indicated that the native kinesin-2 holoenzyme and bacterially expressed fragments of KAP3 containing the armadillo repeats could co-cycle with actin filaments in vitro [27]. Our continued work into this putative association confirms and further substantiates the ability of KAP3 to complex with filamentous actin (F-actin) through its armadillo repeats (Figure 2). For each of the three KAP3 armadillo repeat fragments tested, we observed co-fractionation with filamentous actin (P1, Figure 2). Neither of the non-armadillo repeat fragments of KAP3 used showed an ability to bind F-actin in this assay. Resuspension of the F-actin pellets with bound protein in G-buffer followed by a second round of centrifugation released the actin and bound protein into the soluble fraction (S2, Figure 2). None of the KAP3 fragments pelleted non-specifically (P2, Figure 2). Addition of F-buffer to the S2 fractions allowed for the actin to re-polymerize and be pelleted again during a third round of centrifugation. Once again, the KAP3 armadillo repeat protein present in S2 fractionated into the pellet with F-actin (P3, Figure 2).
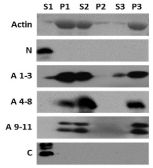
Figure 2: SDS-PAGE with Coomassie staining (actin) or immunoblotting
(KAP3 fragments) of samples from actin co-cycling assays. Supernatant (S)
and pellet (P) fractions from each round (1-3) of centrifugation are labeled S1/
P1 (polymerized F-actin), S2/P2 (depolymerized, monomeric G-actin), and
S3/P3 (cycled F-actin) respectively. KAP3 fragment proteins are identified
to the left of each immunoblot. Immunoblotting was performed using anti-
Xpress™ mAb (Invitrogen, Carlsbad, CA).
At the molar ratios of actin: KAP3 fragment used (between 20:1 and 10:1) all detectable protein from the KAP3 A 4-8 and KAP3 A 9-11 samples pelleted initially (P1) and after cycling (P3) of the actin (Figure 2). The KAP3 A 1-3 samples did not show as complete an ability to co-cycle. We consistently observed a detectable proportion of KAP3 A 1-3 not binding to F-actin during the initial centrifugation (P1) and after cycling (P3) (Figure 2). This outcome would seem to support a model in which this segment of KAP3 does not have as high an affinity for F-actin as the other two fragments tested. A possible explanation for this is a disruption in armadillo repeat sequence found near the middle of repeat 2, in an area previously shown to associate with the actin binding protein fodrin [20].
One of the major limitations of the actin co-cycling assay is that it does not directly assess whether or not our KAP3 fragments can interact with G-actin. To address this issue, we developed an in vitro actin affinity pull down assay in which biotinylated actin monomers were immobilized on sepharose beads (Figure 3A). Results from these assays indicate that none of our KAP3 fragments are capable of binding to immobilized, monomeric G actin (Figure 3B & C) while the majority of the known G-actin binding protein, profilin, did [41,42] (Figure 3A). This result appears to be in opposition to what was previously observed by far western [27]. However, the technical constraints of a far western necessitate validation by an appropriate solution based experiment [43]. Our assay does just that. Additionally, parallel work in which the His-tagged KAP3 fragments were immobilized on nickel beads and used to attempt pull down of G-actin from solution were similarly unable to detect any interaction (data not shown). Further substantiating our conclusion that the KAP3 armadillo repeat fragments that co-cycle with F-actin are incapable of associating with G-actin.
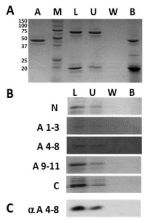
Figure 3: Actin affinity pulls down assay.
A. SDS-PAGE with Coomassie staining of actin affinity pulls down assay
for profilin. Mw Marker (M) (Sizes in kDa to left), actin coated beads (A),
pre-incubation test protein load (L), post-incubation unbound test protein
(U), pooled washes (W) and beads with bound protein (B) fractions are as
labelled.
Major bands correspond to actin (~43 kDa), profilin (~18 kDa) and BSA (~66
kDa).
B. SDS-PAGE with Coomassie staining of actin affinity pulldown assays for
KAP3 fragments. Samples loaded are consistent with those above in A. KAP3
fragment proteins are identified to the left of each gel.
C. SDS-PAGE and immunoblotting of actin affinity pull down assay for KAP3
A4-8 fragment. Immunoblotting was performed using anti-Xpress™ mAb
(Invitrogen, Carlsbad, CA).
Finally, we aimed to validate the in vivo significance of the characterized biochemical interaction. To do so, we cloned the full length human KAP3 DNA sequence into a GFP-tagged mammalian expression vector and transformed it into COS-7 cells. Over expression of GFP-KAP3FL in COS-7 cell culture showed a cortical staining pattern consistent with the expected morphology of the actin cytoskeleton (Figure 4A). Staining of F-actin with rhodamine-Phallacidin (Figure 4B) indicated that the observed GFP-KAP3FL expression pattern indeed overlapped with actin filaments (Figure 4C). Co-localization was most apparent cortically. Not as pronounced, though frequently observed, was GFP-KAP3FL fluorescence coinciding along the length of larger cytoplasmic actin assemblies.
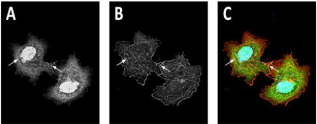
Figure 4: GFP-KAP3 FL/actin co-localization in COS-7 cell culture.A. Localization of GFP-tagged full length human KAP3 overexpressed in
COS-7 cells.B. Actin filaments stained using rhodamine-Phallacidin (Sigma-Aldrich, St.Louis, MO).C. Co-localization (merged images from A and B) of overexpressed GFPKAP3FL (green) and rhodamine-Phallacidin stained actin filaments(red) in COS-7 cell culture with DAPI (blue) counterstain. Arrows indicate
representative areas of co-localization.
Conclusion
By following an optimized actin co-cycling protocol, we show that, in agreement with the previous report, bacterially expressed KAP3 armadillo repeat fragments co-cycle with actin, while two nonarmadillo repeat fragments corresponding to the N and C-terminus of KAP3 do not. Furthermore, the development of an in vitro G-actin binding assay allows us to conclude that none of our KAP3 fragments are capable of binding to G-actin in solution. Lastly, while there is still more analysis to be done on the characterization of the KAP3-actin interaction in vivo, our initial observations confirm that the results from our solution binding studies may have a functional significance in living systems.
References
- Miki H, Setou M, Kaneshiro K, Hirokawa N. All kinesin super family protein, KIF, genes in mouse and human. Proc Natl Acad Sci USA. 2001; 98: 7004-7011.
- Goldstein LS, Yang Z. Microtubule-based transport systems in neurons: the roles of kinesins and dyneins. Annu Rev Neurosci. 2000; 23: 39-71.
- Hirokawa N, Noda Y, Tanaka Y, Niwa S. Kinesin superfamily motor proteins and intracellular transport. Nat Rev Mol Cell Biol. 2009; 10: 682-696.
- Verhey KJ, Kaul N, Soppina V. Kinesin assembly and movement in cells. Annu Rev Biophys. 2011; 40: 267-288.
- Wozniak MJ, Milner R, Allan V. N-terminal kinesins: many and various. Traffic. 2004; 5: 400-410.
- Wang W, Cao L, Wang C, Gigant B, Knossow M. Kinesin, 30 years later: Recent insights from structural studies. Protein Sci. 2015; 24: 1047-1056.
- Yamazaki H, Nakata T, Okada Y, Hirokawa N. KIF3A/B: a heterodimeric kinesin superfamily protein that works as a microtubule plus end-directed motor for membrane organelle transport. J Cell Biol. 1995; 130: 1387-1399.
- Cole DG, Chinn SW, Wedaman KP, Hall K, Vuong T, Scholey JM. Novel heterotrimeric kinesin-related protein purified from sea urchin eggs. Nature. 1993; 366: 268-270.
- Scholey JM. Kinesin-2: a family of heterotrimeric and homodimeric motors with diverse intracellular transport functions. Annu Rev Cell Dev Biol. 2013; 29: 443-469.
- Hirokawa N, Tanaka Y. Kinesin superfamily proteins (KIFs): Various functions and their relevance for important phenomena in life and diseases. Exp Cell Res. 2015; 334: 16-25.
- Fu MM, Holzbaur EL. Integrated regulation of motor-driven organelle transport by scaffolding proteins. Trends in cell biology. 2014; 24: 564-574.
- Marszalek JR, Goldstein LS. Understanding the functions of kinesin-II. Biochim Biophys Acta. 2000; 1496: 142-150.
- Wedaman KP, Meyer DW, Rashid DJ, Cole DG, Scholey JM. Sequence and submolecular localization of the 115-kD accessory subunit of the heterotrimeric kinesin-II (KRP85/95) complex. J Cell Biol. 1996; 132: 371-380.
- Yamazaki H, Nakata T, Okada Y, Hirokawa N. Cloning and characterization of KAP3: a novel kinesin superfamily-associated protein of KIF3A/3B. Proc Natl Acad Sci U S A. 1996; 93: 8443-8448.
- Berezuk MA, Schroer TA. Fractionation and characterization of kinesin II species in vertebrate brain. Traffic. 2004; 5: 503-513.
- Hirokawa N. Stirring up development with the heterotrimeric kinesin KIF3. Traffic. 2000; 1: 29-34.
- Akhmanova A, Hammer III. Linking molecular motors to membrane cargo. Curr Opin Cell Biol. 2010; 22: 479-487.
- Lechtreck KF. IFT-Cargo Interactions and Protein Transport in Cilia. Trends Biochem Sci. 2015; 40: 765-778.
- Shimizu K, Shirataki H, Honda T, Minami S, Takai Y. Complex formation of SMAP/KAP3, a KIF3A/B ATPase motor-associated protein, with a human chromosome-associated polypeptide. The Journal of biological chemistry. 1998; 273: 6591-6594.
- Takeda S, Yamazaki H, Seog DH, Kanai Y, Terada S, Hirokawa N. Kinesin superfamily protein (KIF3) motor transports fodrin-associating vesicles important for neurite building. J Cell Biol. 2000; 148: 1255-1265.
- Jimbo T, Kawasaki Y, Koyama R, Sato R, Takada S, Haraguchi K, et al. Identification of a link between the tumour suppressor APC and the kinesin superfamily. Nat Cell Biol. 2002; 4: 323-327.
- Deacon SW, Serpinskaya AS, Vaughan PS, Lopez Fanarraga M, Vernos I, Vaughan KT, et al. Dynactin is required for bidirectional organelle transport. J Cell Biol. 2003; 160: 297-301.
- Teng J, Rai T, Tanaka Y, Takei Y, Nakata T, Hirasawa M, et al. The KIF3 motor transports N-cadherin and organizes the developing neuroepithelium. Nat Cell Biol. 2005; 7: 474-482.
- Sadananda A, Hamid R, Doodhi H, Ghosal D, Girotra M, Jana SC, et al. Interaction with a kinesin-2 tail propels choline acetyltransferase flow towards synapse. Traffic. 2012; 13: 979-991.
- Guillaud L, Wong R, Hirokawa N. Disruption of KIF17-Mint1 interaction by CaMKII-dependent phosphorylation: a molecular model of kinesin-cargo release. Nat Cell Biol. 2008; 10: 19-29.
- Schonteich E, Wilson GM, Burden J, Hopkins CR, Anderson K, Goldenring JR, et al. The Rip11/Rab11-FIP5 and kinesin II complex regulates endocytic protein recycling. J Cell Sci. 2008; 121: 3824-3833.
- Berezuk MA. Analysis of the invitro activity and cargo binding characteristics of kinesin-2 [dissertation]. Baltimore, MD: The Johns Hopkins University; 2005.
- Carpenter BS, Barry RL, Verhey KJ, Allen BL. The heterotrimeric kinesin-2 complex interacts with and regulates GLI protein function. J Cell Sci. 2015; 128: 1034-1050.
- Tateno M, Kato S, Sakurai T, Nukina N, Takahashi R, Araki T. Mutant SOD1 impairs axonal transport of choline acetyltransferase and acetylcholine release by sequestering KAP3. Hum Mol Genet. 2009; 18: 942-955.
- Khan ML, Ali MY, Siddiqui ZK, Shakir MA, Ohnishi H, Nishikawa K, et al. C. elegans KLP-11/OSM-3/KAP-1: orthologs of the sea urchin kinesin-II, and mouse KIF3A/KIFB/KAP3 kinesin complexes. DNA research: an international journal for rapid publication of reports on genes and genomes. 2000; 7: 121-125.
- Andrade MA, Perez-Iratxeta C, Ponting CP. Protein repeats: structures, functions, and evolution. Journal of structural biology. 2001; 134: 117-131.
- Hatzfeld M. The armadillo family of structural proteins. International review of cytology. 1999; 186: 179-224.
- Riggleman B, Wieschaus E, Schedl P. Molecular analysis of the armadillo locus: uniformly distributed transcripts and a protein with novel internal repeats are associated with a Drosophila segment polarity gene. Genes & development. 1989; 3: 96-113.
- McCrea PD, Turck CW, Gumbiner B. A homolog of the armadillo protein in Drosophila (plakoglobin) associated with E-cadherin. Science. 1991; 254: 1359-1361.
- Choi HJ, Huber AH, Weis WI. Thermodynamics of beta-catenin-ligand interactions: the roles of the N- and C-terminal tails in modulating binding affinity. The Journal of biological chemistry. 2006; 281: 1027-1038.
- Coates JC. Armadillo repeat proteins: beyond the animal kingdom. Trends in cell biology. 2003; 13: 463-471.
- Berezuk MA, Schroer TA. Dynactin enhances the processivity of kinesin-2. Traffic. 2007; 8: 124-129.
- Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970; 227: 680-685.
- Tang MX, Redemann CT, Szoka FC Jr. In vitro gene delivery by degraded polyamidoamine dendrimers. Bioconjug Chem. 1996; 7: 703-714.
- Shimizu K, Kawabe H, Minami S, Honda T, Takaishi K, Shirataki H, et al. SMAP, an Smg GDS-associating protein having arm repeats and phosphorylated by Src tyrosine kinase. The Journal of biological chemistry. 1996; 271: 27013-27017.
- Mockrin SC, Korn ED. Acanthamoeba profilin interacts with G-actin to increase the rate of exchange of actin-bound adenosine 5'-triphosphate. Biochemistry. 1980; 19: 5359-5362.
- Pollard TD. Actin and Actin-Binding Proteins. Cold Spring Harb Perspect Biol. 2016; 8.
- Vaughan KT, Vallee RB. Cytoplasmic dynein binds dynactin through a direct interaction between the intermediate chains and p150Glued. J Cell Biol. 1995; 131: 1507-1516.