
Research Article
Austin Chromatogr. 2016; 3(1): 1040.
Chromatographic Separation of Calcium Isotopes using Benzo-18-Crown-6-Ether Resin and Acetic Acid Solution
Umehara S¹, Okumura S², Kishimoto T³, Fujii Y³*, Nomura M², Kaneshiki T² and Ozawa M²
¹Osaka University, Research Center for Nuclear Physics, Japan
²Tokyo Institute of Technology, Research Laboratory for Nuclear Reactors, Japan
³Osaka University, Graduate School of Science, Japan
*Corresponding author:Yasuhiko Fujii, Research Laboratory for Nuclear Reactors, Tokyo Institute of Technology, O-okayama, Meguroku, Tokyo, 152-8550, Japan
Received: January 18, 2016; Accepted: March 08, 2016; Published: March 14, 2016
Abstract
Chromatographic separation of 48Ca isotope was studied using an acetic acid solution as an eluent and a column packed with benzo-18-crown-6- ether resin. Prior to the chromatographic experiment, adsorption of calcium ions from acetic acid solutions was examined in batch mode experiments at different concentrations of acetic acid. The distribution coefficient Kd of calcium ions was observed to increase at highly concentrated acetic acid solutions. Chromatographic separation experiment, performed based on the batch mode adsorption test, showed that the heavy isotope 48Ca was enriched in the front boundary region. Separation coefficientε and HETP (height equivalent to a theoretical plate) were calculated from the experimental data. The observed ε of the calcium acetate experiment was 4.6 x10-3 and HETP was 2.2cm. These results suggest that separation coefficient ε is relatively larger than that of HCl solution system previously reported. The value of HETP of the present system was found to be very large, if compared with the HETPs of the HCl solution systems. The large ε is advantageous, but the large HETP is disadvantageous from the viewpoint of production of 48Ca enriched calcium. The large value of HETP is presumably due to the slow isotopic exchange between the solution phase and the resin phase.
Keywords: Benzo-18-Crown-6-Ether; Acetic acid; Isotope effect; Isotope separation; HETP; 48Ca
Abbreviations
HETP: Height Equivalent to a Theoretical Plate; B18C6E: Benzo- 18-Crown-6-Ether: NMR: Nuclear Magnetic Resonance
Introduction
Natural calcium consists of 40Ca, 42Ca, 43Ca, 44Ca, 46Ca, and 48Ca. Among these isotopes, 43Ca has the nuclear spin of 7/2 and is used as an NMR detectable calcium isotope. The radioisotope 47Ca, produced by (n, 2n) reaction of 48Ca or by (n,γ) reaction of 46Ca, can be used for medical purposes. In addition, 48Ca is regarded as a stable isotope but actually a double beta decay nuclide; two β rays are emitted simultaneously. The β decay is closely related to the characteristics of neutrino. The double β decay is considered to be a key to understand the physics of neutrino. For the neutrino research, the enrichment of 48Ca is anticipated, since its natural abundance is very low, only 0.187%.
Isotope separation processes based on the chemical exchange have been developed for light elements, such as hydrogen, lithium, boron, carbon, oxygen, and nitrogen. The enriched isotopes of D, 6Li, 7Li, 10B, 13C, 17O, 18O and 15N are commercially available. The principle of the isotope separation of these elements is based on the quantum effects in molecular vibration. Vibration energy remains even at zero degree K. Since the vibrational frequency depends on the mass of the isotope in the molecule, the vibrational energy shows the mass dependence and affects the thermodynamic properties of molecules, such as complex formation constants.
Calcium isotope separation has been studied using various chemical exchange processes. By using strongly acidic cation exchange resin, Aaltonen reported calcium isotope separation coefficient ε (48Ca/40Ca) as 8.7 x 10-4 [1]. By using strongly acidic cation exchange resin and EDTA eluent, Klinskii et al. reported ε (48Ca/40Ca) as -1.7 x 10-4. The minus value of ε shows the heavier isotope is enriched in the cation exchange resin [1].
Heumann et al. [1] studied calcium isotope fractionation in the systems of ion exchange resin, crown ether resin and criptand resin. Among the studied systems, criptand resin showed the largest isotope fractionation, or the largest isotope separation coefficient ε (48Ca/40Ca), as 12.6x10-3. Jepson et al. [2] studied 44Ca isotope separation by breakthrough chromatography with synthesized crown ether resin. The maximum value of ε (44Ca/40Ca) reported by Jepson is 2.7x10-3. From this value, ε (48Ca/40Ca) is theoretically expected to be 4.6 x10-3, which is 1.7 times as large as experimentally observed ε (44Ca/40Ca) Authors of the present work have developed crown ether resin embedded in fine porous silica beads. Using this type of resin, Hayasaka et al. [3], Fujii et al. [4], Oi et al. [5], Umehara et al. [6], and Okumura et al. [7], reported research work on calcium isotope separation.
In the case of crown ether resin, the isotope fractionation takes place in the isotopic exchange process between the calcium ions in the crown-ether resin and those in the outside solution. The exchange process of calcium ions is depicted in Figure 1. The solvents used by Heumann et al. and Jepson et al are chloroform/methanol mixed solutions. The present authors have used concentrated HCl solution and HCl/ethanol mixed solution for Ca isotope separation [3,4,6,7]. The typical values of observed ε are 3.6x10-3, and 3.0 x10-3 and 3.8x10-3 are listed in Table 1.
Solution
Migration (m)
Temperature (oC)
Feed rate (ml/hr)
Band velocity (m/day)
Separation coefficient
ex103
HETP
(cm)
Remarks
9M HCl
0.96
35
20
6.5
3.6±1.4
0.37
[6]
9M HCl
200
35
60
20
3.0±0.1
0.83
[6]
8M HCl 25% + Ethanol 75%
2.84
40
48
11.9
3.8±0.4
0.87
[7]
CH3COOH95% + H2O 5%
0.97
25
24
8.2
4.6±0.9
2.2
This work
Table 1: Experimental conditions and results.
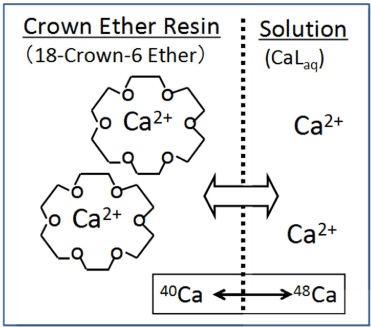
Figure 1: Isotope exchange and isotope fractionation between aqueous
solution and crown-ether resin.
To evaluate the performance of isotope enrichment by chromatography, there are two major factors, separation coefficient ε and HETP (height equivalent to a theoretical plate). The former is a factor showing the thermodynamic property of the isotope effect and the latter is a factor showing the kinetic property of the separation medium. The larger separation coefficient gives better productivity of the separation process. On the other hand, the smaller HETP gives better productivity of the separation process.
There are two types of chromatography; the elution type and the displacement type. Although the elution chromatography is usually applied for analytical chemistry, a displacement chromatography has been used for the separation of isotopes. A large number of stages are necessitated to pile up the enriched isotopes at the front boundary until the isotopic abundance ratio reaches to the desired enrichment degree. The displacement chromatography can build large number of separation stages inside the band. Figure 2 schematically illustrates the isotopic enrichment at the front boundary of a displacement chromatography.
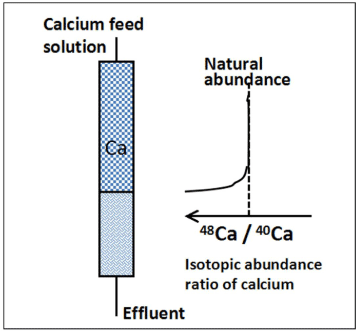
Figure 2: Illustration for chromatographic separation of calcium isotopes.
As shown above, many researchers studied the separation coefficients of different chemical systems, but in most work, other than the work by the present authors, HETP values were not clearly mentioned. In general, it is possible to obtain HETP of displacement chromatography by computational calculation on the isotopic analytical data, however in the small column chromatography, such calculation is most difficult. To determine HETP, we developed a set of theoretical equations derived for HETP of the displacement type, non-steady state chromatographic enrichment process. By using this equation, we calculated HETPs from our experimental data. Representative values of HETP observed in our previous work, 3.7mm, 8.3 mm and 8.7 mm, are listed in Table 1.
In the present paper, the theoretical background of chromatographic isotope separation is briefly mentioned and the experimental results on acetic acid / crown ether resin system are described.
Theoretical Background of Chromatographic Separation
Separation coefficient
The isotope effects or isotopic fractionations take place between coexisting chemical species A and B, when isotopes I1 and I2 are exchangeable between species A and B. The isotope separation factor α, which is frequently called as the isotope fractionation factor, is defined as the ratio of the isotopic abundance ratio [I1]/[I2] of the chemical species A against that of chemical species B.
Where rA and rB are the isotopic abundance ratios in chemical species A and B, respectively. The separation factor defined by above mentioned equation corresponds to the equilibrium constant K of the following isotopic exchange reaction between the chemical species A and B.
The deviation of α from unity, α – 1, is called separation coefficient.
In general, the isotope separation coefficient ε has the mass dependency expressed as,
WhereM1 and M2 are masses of isotopes I1 and I2, respectively, and ΔM is the mass difference between M1 and M2. T is absolute temperature and c is a constant relating to the binding energy between the isotopes and the surrounding atoms. It is understandable from Equation (4) that the isotope effects of heavy elements become very small, because ε is inversely proportional to the product M1M2.
In spite of the small isotope separation coefficient, isotope enrichment is observed in the chromatographic separation process where a large number of separation stages are involved in the column packed with adsorbent. A number of studies on chromatographic isotope separation have been made on lithium, boron, carbon, nitrogen and even uranium by using ion exchange resins.
The separation coefficient is determined by using the experimental data observed by a displacement chromatography [3-9];
Where qi is the moles of isotopic ion in the effluent fraction, R is the mole fraction of an isotope, subscript i and o represent the effluent fraction number and the original feed solution, respectively, and Q is total moles of isotopes adsorbed in the resin. The summation is taken for all the enriched fractions of the effluent. When the abundance of the concerned isotope Ro is very small, (1-Ro) is approximated as unity, and Equation (1) is simplified to,
Where ri is isotopic abundance ratio, and in the present work ri is [48Ca]/[40Ca] of calcium in effluent fraction i. The isotopic abundance ratio r is measured by mass spectrometry.
Above mentioned ε is a physico-chemical separation coefficient associating with the adsorption process. The capacity of Q is the amount of ions of concerned isotopes adsorbed in the resin. It is noted that the separation medium contains the isotopes in the solution phase, of which space is expressed as the dead volume. From the viewpoint of process engineering, we need an engineering separation coefficient based not on the amount of adsorbed isotopes in the resin but on the amount of fed isotopes into the separation system. The engineering separation coefficient εp is defined using the total feed Qt introduced into the separation system as,
And therefore,
HETP
The theory of HETP for displacement-type chromatographic enrichment in non-steady state was developed in the references [8,9]. According to the theory, HETP of the experimental separation system is calculated by the following equations,
Where k is an experimentally determined slope coefficient, H is HETP, L is the migration length of the front boundary of the adsorption band, and |Xi-L| is the distance between the front boundary and the inner-band location Xi corresponding to the effluent volume of fraction i. The 2nd term on the right hand side of Equation (9) is the maximum enrichment observed at the front boundary and regarded as a constant independent of the fraction number i. Although Xi is a hypothetical term, the value of (Xi- L) is converted to the effluent volume deference between fractions i and the breakthrough point (Vi - VB) by using the conversion factor L/VB where VB is the breakthrough volume.
The minus sign is added on the right hand side in Equation (11)
because there are relations of Xi
For the convenience of graphical calculation of slope coefficient k, ln (ri–ro) can be plotted against Vi or (Vi - VB), neglecting the last term ln (rL–ro) in Equation (12).
Experimental
Materials
The crown ether resin was synthesized in our laboratory by condensation polymerization. Usually it is difficult to control small particle diameter in the condensation polymerization. Our special technique applied in the present work is the use of porous silica beads, MST8 produced by Mizusawa Chem. The diameter of the beads is ca. 50 μm and the size of the pores in the beads is approximately 1000 nm. The raw materials of benzo crown, bisphenol A, trichlor-acetic acid, chloroform and paraformaldehyde were mixed with silica beads in a 300 cm3 eggplant-shaped flask. The organic raw materials are liquefied above 80°C and absorbed in the silica beads. The flask was heated to 90oC for totally 8 hours and further heated at 120°C for 4 hours. The benzo-18-crown-6-ether resin was synthesized in the porous silica beads. After the heating, the synthesized rein was washed with methanol several times and dried at room temperature. The organic chemicals used for the synthesis of B18C6E resin were purchased from Wako Pure Chemical Industries, Ltd.
Adsorption of calcium in acetic acid solutions
Prior to the chromatographic experiment, adsorption of calcium ions on the B18C6E resin in the acetic acid solution was examined by a batch mode adsorption test. A portion of B18C6E resin, 2.0 g, was put in a 50 ml plastic vial and added with 25 ml acetic acid solution containing calcium acetate at 50 ppm. The concentration of acetic acid was varied from 1 M to 17.5 M (mol/L).The vials for the adsorption test were placed in a shaking bath, AS ONE GK-0259-02, for 24 hours at 25°C. The vials were shaken at 160 rpm. After the batch mode adsorption experiments, the solution in each bottle was sampled and the concentration of calcium was measured by an atomic absorption spectrophotometer ANA-182F made by Tokyo Photoelectric Co. Ltd with the mode of flame photometry at the wave length of 622 nm.
Chromatography
Chromatographic isotope separation experiment was conducted by using synthesized B18C6E resin packed in glass columns with 8mm inner diameter and 1m long. The resin bed height was 97 cm. Calcium acetate 0.1 M in acetic acid solution (95 % CH3COOH, 5 % H2O) was charged into the column at 25°C by using a double-plunger type, high-pressure corrosion-resistant pumpNP-KX-110U made by Nihon Seimitsu Kagaku Co. The pressure of the feed solution was monitored with a pressure gauge. The flow rate of feed solution was 0.4 ml/min. At this flow rate, observed velocity of the adsorption band was 8.2 m/d. The effluent was sampled in fractions by using a fraction collector, ADVANTEC CHF100AA; the sampling time was 5 minutes for each fraction. The sampling volume was 2 ml in each fraction. The concentration of calcium in the fraction was measured in the same way as mentioned in the earlier section.
Mass spectrometry
The isotope abundance ratios of 48Ca and 40Ca of the calcium in the eluent fractions were measured, by using a mass spectrometer MAT 261, produced by Finnegan MAT, equipped with a doublefilament thermal-ionization ion source. Since the ionization energy of calcium is 6.113 eV, thermal ionization is appropriate to produce stable ion beams.
A portion of each sampled fraction was taken and added with droplets of conc. HI solution and heated to dryness. To convert the chemical form of sampled calcium from calcium acetate to CaI2 for mass analysis, the sample was again added with droplets of conc. HI solution and heated to evaporate excess water to obtain concentrated CaI2.
It is a usual process of calcium mass spectrometry that the CaI2 sample loaded on the evaporation filament is strongly heated and CaI2 sample is converted to CaO on the filament. In this process we can see the mass discrimination in the conversion and evaporation processes of CaO. We developed a different heating method in our previous work [10]. The evaporation filament loaded with ca 10 μg CaI2 is not strongly heated but mildly heated and we obtain CaI2 vapor. Then the CaI2 vapor is ionized at ionization filament. Rhenium filaments are used for both evaporation and ionization in the present work.
The heating currents for the generation of calcium ions, or thermo-ionization, are 0.4 A for evaporation filament and 2.4 ~ 2.8 A for ionization filament. The procedure of thermo-ionization mass spectrometry of calcium is similar to one described in the earlier work [10].
Results and Discussion
Adsorption of calcium
The adsorption capability of the adsorbent is expressed with the distribution coefficient Kd, which is determined by the following equation using the experimentally observed concentration of the chemical species in question,
Where Co and Ce are the initial and the equilibrium concentrations of calcium ions in the solution phase, respectively, Vs is the volume of the solution phase and wa is the weight of adsorbent.
The measured distribution coefficients Kd of calcium is plotted in Figure 3. It is seen that the Kd value becomes very large at the very high concentration of acetic acid. Then the Kd values are drastically decreased by addition of water. The value of Kd approx.6000 is much higher than that of conc. HCl systems previously reported [7]. The reason why the Kd is so large in the case of acetic acid is not fully explained but it is conceivable that the dielectric constant of acetic acid reported as 6.2 is much lower than the value of water, 80. The dielectric constant of acetic acid is similar to those of organic solvents. Since the crown-ether resin is considered as an organic material consisting of ether, phenol, benzene etc., calcium ions are easily extracted from the organic solvent to the resin phase, when both phases are consists of non-polar materials.
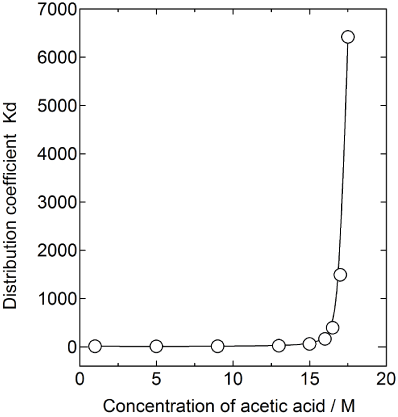
Figure 3: Distribution coefficient of calcium adsorbed on B18C6E resin in
acetic acid solution.
Apparently highly concentrated acetic acid is applicable for chromatographic separation process as a solvent. The large difference in the Kd values is seen between the concentrations 17.5 M and 15M. Care should be taken to keep the concentration of the solution constant in handling the acetic acid solution as the eluent. However, simultaneously, the large difference in Kd could be advantageous from the viewpoint of the regeneration of the resin for cyclic use.
The regeneration procedure is as follows. The eluent of calcium in a concentrated acetic acid solution is charged into the column for enrichment process. After the elution of the calcium adsorption band, the resin is charged with low concentration acetic acid solution to remove the calcium ions adsorbed in the resin. Then the resin is rinsed with conc. acetic acid. Finally the column is again fed with calcium in conc. acetic acid solution. The regeneration is expected to become easy when the process is accomplished only by changing the concentration of the same type of solution.
Chromatography
Taking account of the observed distribution coefficients for acetic acid solutions, the composition of eluent of chromatography was chosen as 0.1 M Ca(CH3COOH)2 in the solvent of 95 % CH3COOH and 5% H2O. The chromatogram of calcium eluted from the bottom of the column is shown in Figure 4 (top). The results of the frontal analysis of isotopic ratio [48Ca]/[40Ca] are also plotted in Figure 4 (bottom). It is confirmed that the heavy isotope 48Ca is accumulated in the front boundary region. This tendency accords with the previously observed isotope fractionation [3-7].
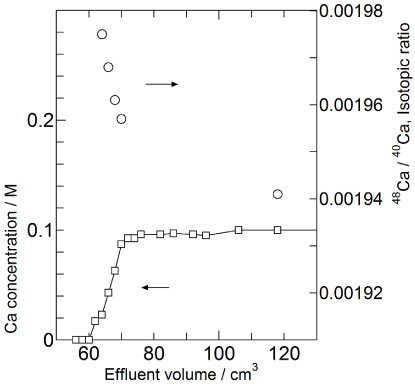
Figure 4: (Top) 48Ca isotope abundance ratio observed at the front boundary.
(Bottom) Concentration of calcium in effluent at the front band boundary.
The selected experimental conditions and the results of chromatographic experiment are listed in Table 1. Using the equations mentioned in the earlier section of the present work, the separation coefficient ε was calculated as 4.6x103. The error of ε observed for short migration chromatography is quite large and is estimated to be about 20%, or ± 0.9x10-3, in the case of the present experimental work.
The previously observed separation coefficients ε for 9M HCl solution [6] and 8M HCl / Ethanol solution [7] are also listed in Table 1. The error range of ε for the short migration of ca.1m using 9MHCl is also large. However the error range of ε for 200m migration is sufficiently small due to the large isotope separation developed at the front band boundary. If we compare the present acetic acid system with 9M HCl with 200m migration, ε of the acetic acid system is larger than that of HCl solution system. To develop the practical enrichment process, the large separation coefficient is very important. The plant size is inversely proportional to the square of ε. As long as the value of ε is concerned, acetic acid is promising as a working solution.
Above mentioned ε is a physicochemical separation coefficient appearing in the adsorption of calcium isotopes on B18C6E resin. The engineering separation coefficients εp is calculated by using Equation (8) as 2.1x10-3. Since the Ca concentration of the feed solution is relatively high, 0.1 M, in the present work, εp was found to decrease to less than a half of ε. The engineering separation coefficient is important for the designing of the enrichment plant and expected to be enhanced by using low-concentration feed solution.
Another important factor to evaluate the efficiency of the isotopic enrichment process is HETP. According to Equation (12), ln (rL – ro) is plotted against the effluent volume difference between fraction i and extreme front boundary (Vi – VB).The plots are shown in Figure 5. It is seen in Figure 5 that the plots fall on a linear line. The slope of the line divided by the conversion factor (L/VB) gives k in Equations (9) and (12).
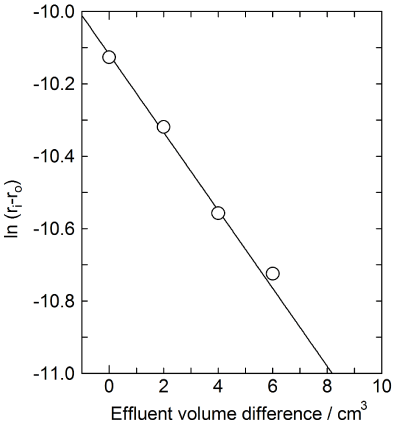
Figure 5: Deviation of isotopic ratios at the band front.
The observed value of HETP calculated by using Equation (10) in the present work is 2.2 cm and listed in Table 1. This value is also quite large if compared with HETPs of other systems in Table 1. The large value of HETP means the slow exchange rates of calcium isotopes between the adsorbent phase and the solution phase. The reason, why HETP of the present system is so large, has not yet been elucidated, but probably it is related to the large Kd value of the present system. The Kd values of conc. HCl systems measured under conditions similar to the present work are ca. 20 [7]. This value of Kd is much smaller than the Kd of the present system of conc. acetic acid, ca. 6000.
The Kd is a kind of equilibrium constant for the adsorption of calcium. In general, the equilibrium constant is expressed as the ratio of the forward reaction rate constant kf to the backward reaction rate constant kb. The large Kd value suggests that the backward reaction rate constant is smaller than the forward reaction rate constant. In the case of very large Kd value, the rate constant of backward reaction or the complex decomposition reaction is estimated to become very small. For the chemical exchange isotope separation, the large reaction rates are very important to obtain small HETP. In the acetic acid solution, it is estimated that the crown ether compound of calcium is so stable that the calcium ions are not easily detached from the solid organic phase to the solution phase. The HETP is a kinetic factor and can be improved by adjusting operational conditions and the resin structure.
Conclusion
Adsorption of calcium ion on B18C6E resin was studied using acetic acid solution. Since calcium ions showed large Kd value at highly concentrated acetic acid solution, chromatographic experiment was conducted with acetic acid solution, 95% CH3COOH and 5% H20, at 25°C. Separation coefficient observed in the present work was 4.6x10- 3.This value is larger than those of HCl systems previously reported. But the observed HETP of the acetic acid system, 2.2 cm is very large.
The synthesis of B18C16E resin developed in the present work showed the low cost production process. This technique is applicable to the large scale production of the crown ether resin. The crown ether resin embedded in fine particle porous silica beads may be applicable to chemical separation and purification of various cations in aqueous solution. Typical example may be separation of radioactive strontium in aqueous solution. Development of separation technology using crown ether resin is anticipated.
Acknowledgement
This work was financially supported by the Japan Society for the Promotion of Science, Grant-in-Aid for Young Scientists (A) (JSPS KAKENHI Grant Number 23684015) and Grant-in-Aid for Scientific Research (S) (JSPS KAKENHI Grant Number 24224007).
References
- Heumann KG, Schiffer HP, Spiess W. Enrichment of heavy calcium isotope by ion exchange resins with cyclopolyethers as anchor groups. Schmidt HL, Foerstel H, Heinzinger K, editors. In: Stable Isotope. Elsevier. 1982; 711-718.
- Jepson BE, Evans WF. Calcium Chemical Exchange. In: Mound Activities in Chemical and Physical Research: January–December 1989, MLM-3654. 1990; 4-26.
- Hayasaka K, Kaneshiki T, Nomura M, Suzuki T, Fujii Y. Calcium ion selectivity and isotope effects studied by using benzo-18-crown-6 resin. Progress in Nuclear Energy. 2008; 50: 510-513.
- Fujii Y, Nomura M, Kaneshiki T, Sakuma Y, SuzukiT, Umehara S, et al. Mass dependence of calcium isotope fractionations in crown-ether resin chromatography. Isotopes in Environmental and Health Studies. 2010; 46: 233-241.
- Nemoto S, Nomura M, Oi T. Calcium isotope fractionation in liquid chromatography with crown ether resin in methanol medium. Isotopes Environ Health Stud. 2013; 49: 257-268.
- Umehara S, Kishimoto T, Kakubata H, Nomura M, Kaneshiki T, Suzuki T, et al. A basic study on the production of enriched isotope 48Ca by using crown-ether resin. Prog theor Exp Phys. 2015; 053C03.
- Okumura S, Umehara S, Fujii Y, Nomura M, Kaneshiki T, Ozawa M, et al. Separation of calcium-48 isotope by crown ether chromatography using ethanol/hydrochloric acid mixed solvent. J Chromatogr A. 2015; 1415: 67-72.
- Fujii Y, Aida M, Okamoto M, Oi T. A Theoretical Study of Isotope Separation by Displacement Chromatography. Sep Sci Technol.1985; 20: 377-392.
- Aida M, Fujii Y, Okamoto M. Chromatographic enrichment of 10b by using weak-base anion-exchange resin. Sep sci technol. 1986; 21: 643-654.
- Fujii Y, Hoshi J, Iwamoto H, Okamoto M, Kakihana H. Calcium isotope effects in ion exchange electro migration and calcium isotope analysis by thermo-ionization mass spectrometry. Z Natureforsch. 1985; 40a: 843-848.