
Case Report
Austin J Clin Neurol 2015;2(6): 1055.
Headache Attributed to Petrous Apicitis without Symptoms of Acute Otitis Media
Young-Il Rho*
Department of Pediatrics, Chosun University School of Medicine, Korea
*Corresponding author: Young Il Rho, Department of Pediatrics, Chosun University Hospital, 365 Pilmoondaero, Dong-gu, Gwangju, 501-717, South Korea
Received: April 07, 2015; Accepted: May 10, 2015; Published: May 30, 2015
Abstract
The classical findings of petrous apicitis, also known as Gradenigo’s syndrome, include abducens nerve palsy, deep facial pain and symptoms of acute otitis media. With the widespread use of antibiotics, petrous apicitis has become an uncommon disease. However, when it occurs, it may be silent or inconspicuous at first and gradually progress to a life-threatening stage. Petrous apicitis has conventionally been treated with aggressive surgical methods. However, recent reports have described good outcomes of more conservative medical treatments with minimal surgical intervention. Here, we report a case involving a 12-year-old child with petrous apicitis without symptoms of acute otitis media, who presented with persistent headaches since 14 days. On day 2 of admission, he developed diplopia and lateral limitation of the left eye. Medical treatment resulted in favorable outcomes, and the child was in good health without headaches or abducens nerve palsy at the 2-month follow-up.
Keywords: Headache; Petrous apicitis; Otitis media
Introduction
Petrous apicitis due to otitis media has become a rare disease, particularly in children, after the widespread availability and use of antibiotics. This condition is characterized by ipsilateral abducens nerve palsy, pain in the region of the first and second divisions of the trigeminal nerve and symptoms of acute otitis media [1]. In addition, patients present with headache, diplopia, tinnitus, and trigeminal and facial nerve palsies. The clinical presentation can vary. When petrous apicitis occurs with silent or inconspicuous symptoms of otitis media, it is more dangerous and frequently fatal [2,3]. The atypical symptoms require special attention. Petrous apicitis has traditionally been treated with aggressive surgical methods. However, recent reports have described good outcomes of more conservative therapy with high-dose broad-spectrum antibiotics and/or minimal surgical intervention [4,5]. Here, we describe the clinical and neuroimaging finding of a 12-year-old boy with petrous apicitis, without symptoms of acute otitis media who presented with persistent headaches since 14 days. His condition was successfully managed with medical treatment alone.
Case Presentation
A 12-year-old boy was admitted to the pediatric neurology department with a chief complaint of daily severe headaches since 14 days. He described the headaches as follows: severe, pulsatile, 3~6 hour duration, behind the left eye, in the forehead and left temporal regions, and associated with nausea. Routine analgesics were ineffective. His medical history indicated an upper respiratory infection that occurred a month back and lasted for 3 days.
On admission, he denied any preceding infectious symptoms, including fever, sore throat, and sinus congestion, cough and ear pain. His vital signs were as follows: body temperature, 36.6°C; pulse rate, 91-beats/min; and blood pressure, 110/75 mmHg. The findings of physical examination were normal, and he was alert with no other abnormal neurological findings. Laboratory investigations revealed the following: white blood cell count 12,800/mm3, with 8,960/mm3 polymorphonuclear neutrophils; C-reactive protein, 5.43 mg/dL (reference range, <0.3); and erythrocyte sedimentation rate, 62 mm/h. His hemoglobin level, platelet count, routine chemistries, liver function profile, ammonia concentration, and electrolyte level were within reference ranges. Culture results and virus studies were normal. A diagnosis of migraine without aura was initially considered. However, the following day, he complained of double vision and developed fever. Furthermore, he was unable to fix a lateral gaze with the left eye, which suggested left abducens nerve palsy (Figure 1). Ophthalmologic examination did not reveal papilledema, and otoscopic examination finding were normal. He exhibited confusion, dysphasia, and bilateral facial palsy.
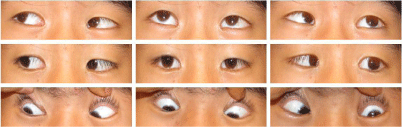
Figure 1: Lateral gaze palsy of the left eye 2 days after admission.
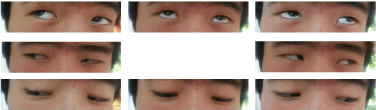
Figure 5: Improved lateral gaze with the left eye at the 2-month follow-up.
Computed tomography images (bone window) demonstrated bony erosion along the cortex surrounding the petrosal pyramid with widened petroclivial fissure, although there was no fluid in the middle ear (Figure 2). Brain magnetic resonance imaging (MRI) demonstrated T1 low- and T2 high-signal intensity lesions involving the left petrous bone, with strong enhancement and diffuse narrowing of the left petro-cavernous internal carotid artery. Highsignal intensity in the left mastoid air cells on T2-weighted images (Figure 3). Cerebrospinal fluid examination showed normal findings.

Figure 2: (a) Axial and (b) coronal computed tomography images (bone
window) before treatment. Bony erosion is demonstrated along the cortex
surrounding the petrosal pyramid (straight arrows), with a widened petroclivial
fissure (asterisk).

Figure 3: (a) A T1-weighted axial image, (b) T2-weighted axial image, and
(c, d) enhanced axial and sagittal T1-weighted images show T1 low- and T2
high-signal intensity lesions involving the left petrous bone (straight arrows)
with strong enhancement and diffuse narrowing of the left petro-cavernous
internal carotid artery (curved arrow). (b) A T2-weighted image shows
heterogeneous high-signal intensity in the left mastoid air cells (asterisk).
The patient was treated with intravenous antibiotic therapy including amoxicillin-clavulanic acid, gentamycin and metronidazole for 4 weeks, following which his symptoms significantly subsided. Twelve days later, his fever and headaches disappeared, and 4 weeks later, he showed partial recovery from diplopia and the abducens nerve palsy. The patient was discharged without antibiotic therapy and showed further recovery from abducens nerve palsy at the 1-month follow-up.
At the 2-month later follow up, T1-weighted MRI showed a decrease in the size of the enhanced region in the left petrous bone compared with that on previous MRI studies. T2-weighted MRI showed no heterogeneous signal intensity and pneumatization of the left mastoid air cells (Figure 4). Clinically, the child was in good health without headaches and abducens nerve palsy (Figure 5).

Figure 4: Follow-up Magnetic resonance (MR) images obtained 2 months
after discharge, (a) a T1-weighted axial image, (b) T2-weighted axial image,
and (c, d) enhanced axial and coronal T1-weighted images show a decrease
in the size of the enhanced portion (straight arrows) in the left petrous bone
compared with that on previous MR images. The T2-weighted image shows
no heterogeneous signal intensity and pneumatization of the left mastoid air
cells (asterisk).
Discussion
Petrous apicitis due to otitis media, which was first described by Giuseppe Gradenigo in 1907, is characterized by ipsilateral abducens nerve palsy, pain in the region of the first and second divisions of the trigeminal nerve, and symptoms of acute otitis media [1]. Although petrous apicitis, also known as Gradenigo syndrome, was common and frequently fatal in the pre-antibiotic era, it is now a rare entity with the wide availability of antibiotics, particularly in children.
The incidence of petrous apicitis is reported to be approximately 2/100,000 children with acute otitis media [6]. The petrous apex lies between the inner ear and the clivus in the most medial portion of the temporal bone and communicates with the middle ear through well-aerated cells. The petrous apex is in contact with the cranial nerves, vascular structures, dura, and the brain, which may be affected by inflammation in the petrous apex. Petrous apicitis may cause direct invasion because of bone destruction or may spread via venous channels [7]. In addition, the infection may spread outside the petrous apex to affect the meninges and cranial nerves. The present patient complained of persistent headaches, abducens nerve pals, and diplopia without otitis media. The presence of atypical symptoms is not an unusual presentation of Gradenigo’s syndrome; this condition frequently present without all typical symptoms. Chole and Donald reported a case series involving eight petrous apicitis, none of whom presented with all typical symptoms [5,8]. Such cases require special attention.
The condition is generally diagnosed on the basis of clinical findings, which are generally characteristic. Imaging with CT and MRI is important to identify the extent of abnormalities and distinguish petrous apicitis from other lesions involving the petrous apex, such as cholesterol granuloma, cholesteatoma, osteomyelitis, and neoplasms [9].
Untreated petrous apicitis can rapidly become life-threatening because of meningitis, empyema, brain abscess, venous thrombosis, cranial nerves palsies, and internal carotid stenosis. This condition has transitionally been treated with surgical care, but recently improved antibiotic regimens have allowed for the consideration of non surgical therapy [4,5,10]. Burston et al. reported a 6-yearold child with Gradenigo’s syndrome who was managed without mastoidectomy and showed full recovery [4]. Kong et al. reported a 10-year-old child with acute otitis media-induced petrous apicitis who was successfully treated by ventilation tube insertion [10]. Giber et al. described a 5-year-old boy with atypical Gradenigo’s syndrome without clinical otitis media diagnosed by clinical and neuroimaging examination, why showed favorable outcome after medical therapy alone [5].
In conclusion, we reported the clinical and neuroimaging findings of a child with petrous apicitis without symptoms of acute otitis media who present with persistent headaches since 14 days and were successfully managed with complete recovery after medical treatment alone. Although petrous apicitis, without symptoms of acute otitis media is rare, special attention is required because it may be inconspicuous or silent at first and gradually progress to become life-threatening.
Acknowledgment
Witten informed consent was obtained from the patient to publish the report.
References
- Gradenigo G. Uber die Paralyse des N. Abduzens bei Otitis. Arch Ohrenheilk. 1907; 74: 149-158.
- Bravo D, Machová H, Hahn A, Marková H, Otruba L, Mandys V, et al. Mastoiditis complicated with Gradenigo syndrome and a hypertrophic pachymeningitis with consequent communicating hydrocephalus. Acta Otolaryngol. 2007; 127: 93-97.
- Hananya S, Horowitz Y. [Gradenigo syndrome and cavernous sinus thrombosis in fusobacterial acute otitis media]. Harefuah. 1997; 133: 284-286, 335.
- Burston BJ, Pretorius PM, Ramsden JD. Gradenigo's syndrome: successful conservative treatment in adult and paediatric patients. J Laryngol Otol. 2005; 119: 325-329.
- Gibier L, Darrouzet V, Franco-Vidal V. Gradenigo syndrome without acute otitis media. Pediatr Neurol. 2009; 41: 215-219.
- Goldstein NA, Casselbrant ML, Bluestone CD, Kurs-Lasky M. Intratemporal complications of acute otitis media in infants and children. Otolaryngol Head Neck Surg. 1998; 119: 444-454.
- Stamm AC, Pinto JA, Cóser PL, Marigo C. Nonspecific necrotizing petrositis: an unusual complication of otitis in children. Laryngoscope. 1984; 94: 1218-1222.
- Chole RA, Donald PJ. Petrous apicitis. Clinical considerations. Ann Otol Rhinol Laryngol. 1983; 92: 544-551.
- Jackler RK, Parker DA. Radiographic differential diagnosis of petrous apex lesions. Am J Otol. 1992; 13: 561-574.
- Kong SK, Lee IW, Goh EK, Park SE. Acute otitis media-induced petrous apicitis presenting as the Gradenigo syndrome: successfully treated by ventilation tube insertion. Am J Otolaryngol. 2011; 32: 445-447.