
Case Report
Austin J Clin Neurol 2016; 3(2): 1094.
A Case of Sjögren’s Syndrome Associated with Only Seropositive Foraquaporin-4 Antibodies without Clinical Evidences of Neuromyelitisoptica
Do-Hyung Kim*
Department of Neurology, Eulji University School of Medicine, South Korea
*Corresponding author: Do-Hyung Kim, Department of Neurology, Eulji University Hospital, School of Medicine, Eulji University, DunsanSeo-ro 95, Seo-gu, Daejeon, 35233, South Korea
Received: September 26, 2016; Accepted: November 04, 2016; Published: November 08, 2016
Abstract
Neuromyelitisoptica (NMO) is characterized by optic neuritis and longitudinally extensive transverse myelitis. With the recent discovery of an antibody to aquaporin-4 (NMO-IgG) which has high sensitivity and specificity for diagnosing NMO, the coexistence of Sjögren’s syndrome (SS) and Neuromyelitisoptica spectrum disorder (NMOSD) has been increasing reported. We report a case of SS with a positive antibody to aquaporin-4 without any evidence of myelitis or clinical features of NMO or NMOSD. To our knowledge, antibody to aquaporin-4 positivity in SS without clinical evidence of NMO was unusual case.
Keywords: Neuromyelitisoptica; Sjogren’s syndrome; Aquaporin 4; Rheumatoid factor
Introduction
Sjögren’s syndrome is an autoimmune exocrinopathy characterized by exocrine dysfunction of lacrimal and salivary glands and by dysfunction even in the exocrine gland of female genital tract, skin, nose, trachea and gastrointestinal tract. Neuromyelitisoptica is an idiopathic, severe, and inflammatory demyelinating disease of the central nervous system that preferentially affects the optic nerve and spinal cord. NMO-IgG autoantibody as a disease specific marker of NMO arises from B cells, binds to aquaporin 4 expressed on astrocyte foot process. Detecting antibody to aquaporin-4 (anti AQP4-IgG) can be used very helpfully as a serological test in diagnosing NMO and NMOSD.
The role of autoimmunity in NMO was the association with other autoimmune diseases such as thyroiditis, systemic lupus erythematosus (SLE) or SS in 10-40% of patients [1], and those were accepted concept. Although anti AQP4-IgGis frequently found in patients with connective tissue disorders combined with neurological symptoms suggestive of NMO or NMOSD, there have not been reports of discovery of anti AQP4-IgG in any patients of connective tissue disorders without clinical evidences of NMO or NMOSD [2]. Herein, we describe a patient with serologic and radiologicconfirmed SS who showed a serum anti AQP4-IgG without evidences of myelopathy, optic neuropathy and any central nervous system involvement.
Case Presentation
68-year-old Korean woman was consulted to the neurology department because of generalized weakness and bilateral droopy eyelid. From 19 years ago, she has been managed with a diagnosis of Grave’s disease. The patient displayed weight loss of about 4-5 kg during a year without visible muscle atrophy. Although there was reduction in neck muscle power, she denied dysphasia or dysarthria. Symptoms of shoulder and forearm pain, dry mouth and ocular fatigue were accompanied along with reduction in lacrimation and blurred vision.
In neurological examination, except for bilateral droopy eyelids and deterioration of neck muscle power (Grade IV), other abnormalities were not observed. The laboratory test showed positive antinuclear and Ro (SS-A), La (SS-B) antibodies and an increased erythrocyte sedimentation rate of 86 mm/hour. In addition, the fluorescent antinuclear antibody (speckled pattern), anti-RNP, antidsDNA, anti-Smith, anti-TM antibody test, and rheumatoid factor were positive. Thyroid function test revealed mild reduction in free T4 (0.8 ng/dL, normal 0.89-1.76 ng/dL) with normal T3 (70 ng/dL, normal 65-150 ng/dL), thyroid stimulating hormone (0.66 μIU/mL, normal 0.55-4.78 μIU/mL), thyroglobulin antibody (22.2 U/mL, normal 0.0-60.0 U/mL), thyroid microsomal antibody (54 U/mL, normal 0.0-60.0 U/mL) and thyroid stimulation hormone receptor antibody (0.92 IU/L, normal 0.0-1.75 IU/L). Salivary gland scan revealed decreased secretary and excretory function in both parotid and submandibular glands (Figure 1). Schirmer test revealed Sjögren’s dry eye (4 mm/5 mm). The cerebrospinal fluid study and brain magnetic resonance imaging (MRI) were normal. Nerve conduction study, electromyography, visual evoked potential, repetitive nerve stimulation test, and neostigmine test were within normal limits. Chest computed tomography was also normal, and screening for the tumor markers (alpha fetoprotein, carcinoembryonic antigen, cancer antigen-125, 19-9, anti-Hu, Ri, and Yo) and anti-acetylcholine receptor antibody were normal.
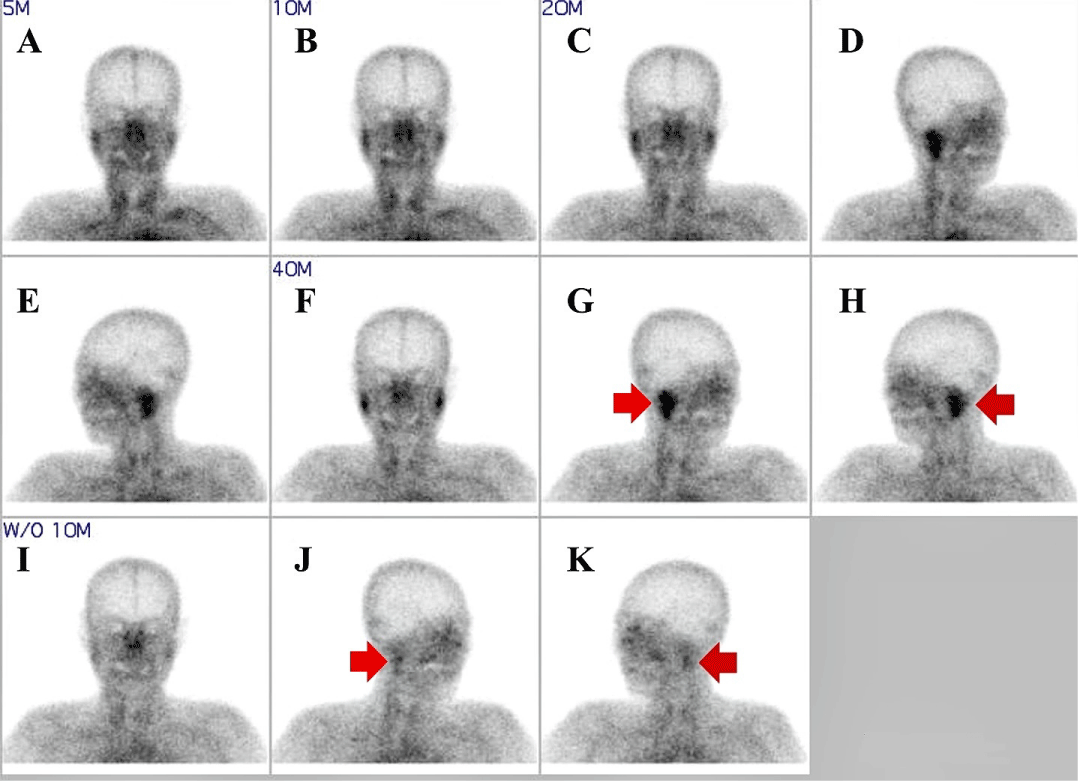
Figure 1: 99mTc-pertechnetate salivary gland scintigraphy showed that
delayed uptake and decreased excretory function ((G), (H) red arrow) in both
salivary gland. However, wash out function was normal ((J), (K) red arrow).
Whole spine MRI and anti AQP4-IgG test were carried out in order to discern whether the patient’s neurological symptoms were associated with myelopathy associated with SS or NMOSD. There was no finding suggests myelopathy, however, anti AQP4-IgG was positive by indirect immunofluorescence method.
Discussion
Aquaporin is a water channel present in the central nervous system and is found mostly in periventricular area, the medullary floor of the fourth ventricle, area postrema, neocortex, hippocampal dentate gyrus, medial habenular nucleus, choroid plexus, retina and optic nerve. Currently, total 11 types of aquaporin subtypes have been discovered. Among these, aquaporin-4 subtype is thought to be in charge of key role in transportation of substances between blood vessels and brain. anti AQP4-IgG combines with pia mater and cerebral microvessel present in the Virchow-Robin space to induce inflammatory demyelinating disorder of central nervous system [3]. Lennon, et al. reported a serum auto anti body, anti AQP4-IgG, the presence of which was 73% sensitive and91% specific for clinically defined NMO [1].
Increasing evidences about the role of autoimmunity in NMO have revealed the associations with autoimmune diseases such as thyroiditis, SLE or SS. Anti-nuclear autoantibodies were detected in the serum of about 50% of patients with NMO [4]. Non-organ specific auto-antibodies (particularly anti-Ro) are seen more frequently in the serum of patients with recurrent transverse myelitis or relapsing NMO (77%) than in those with monophasic disease (33%) [5] Our patient met the diagnostic criteria for SS and maybe with other autoimmune disorder, such as SLE and Grave’s disease. SS can involve nervous system approximately in 20% cases and large portions of them are accompanied with NMO or NMOSD. In previous study, 3% of patients with NMO met the international criteria for the diagnosis of SLE or SS, and 78%were seropositive for anti AQP4-IgG [4] NMO patients who met the diagnostic criteria for SLE or SS but did not have optic neuritis or myelitis were anti AQP4-IgGseronegative [1] and seropositive patients may manifest limited forms of the disease, which includes isolate recurrent optic neuritis, recurrent transverse myelitis [6]. Until now, anti AQP4-IgG has not been found in autoimmune disorders that had no manifestations of myelitis or optic neuritis suggestive of NMO or NMOSD [7]. Therefore, transverse myelitis or optic neuritis is likely to occur in patients with SLE or SS who are seropositive for anti AQP4-IgG, which signifies the coexistence of two autoimmune diseases. Previous report had shown that cancer coexistence with NMOSD was 5% in large cohorts but up to 15% in cohorts with older patients and the authors speculated correlation with inflation of anti AQP4-IgG and underlying oncologic disease [8]. However, as the normal result of tumor marker, the cancer relation could be excluded in our patient. Another previous report had shown that a case of just isolated seropositive for anti AQP4- IgG without any neurological deterioration [9]. They suggested that certain immunologic events (possibly infections and other immune activations) that increased blood-brain barrier (BBB) permeability are needed to allow anti AQP4-IgG to cross BBB. And myelitis or optic neuritis would be possible only after the anti AQP4-IgG cross the BBB to bind to AQP4 on astrocytes. Isolated seropositive for anti AQP4-IgG could also be related to the immunologic events weren’t happened in our patient.
Although, in the majority of the aforementioned reported cases, anti AQP4-IgG are found only when connective tissue disease (CTD) is accompanied with NMO [10], finding anti AQP4-IgG in CTD without the accompaniment of neurological symptoms of NMO has not been reported. It suggests that SS may be highly associated with NMOSD and anti AQP4-IgG, and that it needs to have long term observation, whether SS precede NMOSD or not. Therapeutically, whether aggressive immunosuppressive therapy is needed to prevent progression of SS or SLE to NMO or NMOSDs should be further investigated.
References
- Wingerchuk DM, Lennon VA, Lucchinetti CF, Pittock SJ, Weinshenker BG. The spectrum of neuromyelitis optica. Lancet Neurol. 2007; 6: 805-815.
- Zhang B, Zhong Y, Wang Y, Dai Y, Qiu W, Zhang L, et al. Neuromyelitis optica spectrum disorders without and with autoimmune diseases. BMC Neurol. 2014; 14: 162.
- Jacob A, Matiello M, Wingerchuk DM, Lucchinetti CF, Pittock SJ, Weinshenker BG. Neuromyelitis optica: changing concepts. J Neuroimmunol. 2007; 187: 126-138.
- Pittock SJ, Lennon VA, Wingerchuk DM, Homburger HA, Lucchinetti CF, Weinshenker BG. The Prevalence of Non-Organ-Specific Autoantibodies and NMO-lgG in Neuromyelitis Optica (NMO) and Related Disorders. Neurology. 2006; 66: A307.
- Hummers LK, Krishnan C, Casciola-Rosen L, Rosen A, Morris S, Mahoney JA, et al. Recurrent transverse myelitis associates with anti-Ro (SSA) autoantibodies. Neurology. 2004; 62: 147-149.
- Pandit L. Neuromyelitis optica spectrum disorders: An update. Ann Indian Acad Neurol. 2015; 18: S11-S15.
- Pittock SJ, Lennon VA, de Seze J, Vermersch P, Homburger HA, Wingerchuk DM, et al. Neuromyelitis optica and non organ-specific autoimmunity. Arch Neurol. 2008; 65: 78-83.
- Rossel T, Zekeridou A, Ochsner F, Renaud S. Aquaporin-4-positive myelitis associated with Sjogren syndrome and colonic adenocarcinoma. Neurol Neuroimmunol Neuroinflamm. 2015; 2: e103.
- Nishiyama S, Ito T, Misu T, Takahashi T, Kikuchi A, Suzuki N, et al. A case of NMO seropositive for aquaporin-4 antibody more than 10 years before onset. Neurology. 2009; 72: 1960-1961.
- Weinshenker BG, Wingerchuk DM, Vukusic S, Linbo L, Pittock SJ, Lucchinetti CF, et al. Neuromyelitis optica IgG predicts relapse after longitudinally extensive transverse myelitis. Ann Neurol. 2006; 59: 566-569.