
Case Report
Austin J Emergency & Crit Care Med. 2015;2(1): 1010.
Acute Cholecystitis Caused by Methicillin-resistant Staphylococcus aureus: A Rare Case Report
Po-An Su1,2 and Wen-Liang Yu3,4*
1Department of Internal Medicine, Section of Infectious Diseases, Chi-Mei Medical Center, Tainan
2Department of Pharmacy, Chia-Nan University of Pharmacy and Science, Taiwan
3Department of Intensive Care Medicine, Chi Mei Medical Center, Taiwan
4Department of Medicine, Taipei Medical University, Taiwan
*Corresponding author: Wen-Liang Yu, Department of Intensive Care Medicine, Chi-Mei Medical Center, 901 Chung-Hwa Road, Yungkang City, Taiwan
Received: November 21, 2014; Accepted: January 07, 2015 Published: January 09, 2015
Case Report
Acute cholecystitis is usually a mixed infection most commonly by Escherichia coli accompanied with Enterococcus species and anaerobes. Methicillin-resistant Staphylococcus aureus (MRSA) rarely causes the disease entity. However, it is uncertain whether anti- MRSA therapy should be started for acute cholecystitis for a patient with end-stage renal disease (ESRD) in the emergency medicine.
An 84-year-old man with ESRD on maintenance hemodialysis presented at the Emergency Department of our hospital with epigastric pain for one day. Other symptoms were poor appetite, nausea, vomiting and general malaise for one week. Physical examination revealed a temperature of 39.5oC, a heart rate of 94 beats per minute, a respiratory rate of 17 breaths per minute and a blood pressure of 117 /70 mmHg. Mild abdominal distension and epigastric tenderness with rebounding pain were noticed. Initial blood test revealed a white blood-cell count of 12,400/uL, a platelet count of 61,000/uL and a hemoglobin level of 10.4 g/dL. Chest X-ray revealed borderline cardiomegaly with widening and tortuosity of thoracic aorta. The computed tomography of the abdomen revealed gallstones complicated with acute cholecystitis (Figure 1). After insertion of percutaneous transhepatic gallbladder drainage (PTGBD), the patient became hypotensive and was admitted to the intensive care unit. Biochemistry data included a C-reactive protein of 166 mg/L (normal, <3); albumin, 3.1 g/dL; blood urea nitrogen, 43 mg/dL; creatinine, 4.08 mg/dL; serum glutamic-oxaloacetic transaminase, 35 IU/L; serum glutamic pyruvic transaminase, 12 IU/L; lipase, 64 IU/L; total bilirubin, 1.86 mg/dL and lactate 1.4 mmole/L. Empirical antibiotic therapy with piperacillin-tazobactam was used. The results of blood cultures were of no growth. The bile culture yielded MRSA susceptible to minocycline, tigecycline, teicoplanin, vancomycin (MIC, 1 ìg/mL) and fusidic acid, but resistant to oxacillin and clindamycin. Antibiotic was shifted to intravenous tigecycline (50 mg every 12 hours) for 1 week. The patient was getting better and transferred to general ward. He was discharged uneventfully after 10 days of hospitalization. Antibiotic was shift to oral fusidic acid (500 mg tid) for 4 weeks as prolonged antibiotic suppression therapy. One month later, T-tube cholecystography via PTGBD cholangiogram revealed filling defects in gallbladder and non-opacification of cystic duct by contrast medium (Figure 2). The patient underwent selective laparoscopic cholecystectomy after 2 months of discharge. Multiple black pigmented stones in the thicken-wall gallbladder with sticky pus formation were found and pathology of gallbladder confirmed acute cholecystitis with cholelithiasis.
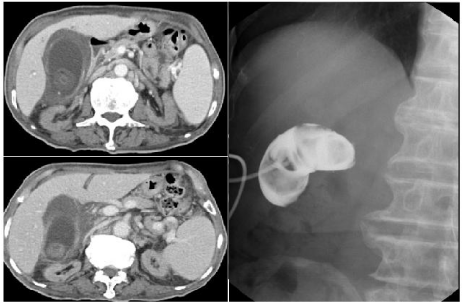
Figure 1 and 2 : (Right): T-tube cholecystography revealed filling defects in gallbladder and non-opacification of cystic duct by contrast medium.
Teranishi et al. reported the first case of acute cholecystitis caused by MRSA, which was published in the Japanese literature [1]. Nguyen and Ramus presented an MRSA gallbladder empyema and septicaemia in a previous vascular surgical patient with a prosthetic implant, which was the first case reported in the English literature [2]. Similar to our case, Martin et al reported a MRSA cholecystitis in an ESRD patient with previous recurrent MRSA bacteremia, which was believed to cause the MRSA cholecystitis [3]. Because our case did not have previous or concurrent MRSA bacteremia, we believe that our case was a primary MRSA cholecystitis. Kim et al. reported a human immunodeficiency virus-infected patient with acute cholecystitis caused by a community-associated strain of MRSA [4]. Nepal et al. reported successful treatment of MRSA cholecystitis by using vancomycin therapy and laparoscopic cholecystectomy [5].
Cases of MRSA-associated acute cholecystitis have rarely been reported, and usually in an immunocompromised patient. ESRD could be the risk for MRSA bactibilia, thus anti-MRSA agent could be reasonably considered for treating acute cholecystitis in an ESRD patient visiting an emergency room of acute health-care facilities. Compared to vancomycin, tigecycline has higher concentration in the bile and could be suggested as an alternative antibiotic to vancomycin.
In conclusion, our case experienced significant response to tigecycline therapy for the MRSA infection, which should be added to the growing list of etiology causing acute cholecystitis, particularly in ESRD patients. The risk factor for the MRSA infection of our case could be attributed to frequent exposure to the dialysis setting.
References
- Teranishi T, Okamoto Y, Hata N, Matsuoka M, Ogawa M, Kiyota K, et al. [A case of toxic shock syndrome and acute cholecystitis caused by MRSA]. Nihon Shokakibyo Gakkai Zasshi. 2002; 99: 165-169.
- Nguyen DQ, Ramus NI. A fatal case of MRSA septicaemia and gallbladder empyema. Int J Surg. 2004; 2: 120-121.
- Martin J, Miller D, Ménard GE. Treatment considerations for recurrent MRSA bacteremia leading to cholecystitis. J Gen Intern Med. 2011; 26: 669-672.
- Kim J, Gregson DB, Church DL. A case of acute cholecystitis caused by methicillin-resistant Staphylococcus aureus in an immunocompromised patient. Can J Infect Dis Med Microbiol. 2011; 22: e7-9.
- Nepal SK, Giri S, Panday K. Successful treatment of methicillin-resistant Staphylococcus aureus bacteraemia and cholecystitis. BMJ Case Rep. 2012; 2012.