
Research Article
Austin J In Vitro Fertili. 2015; 2(3): 1021.
Interpretation: Real Time Assessment on Immotile but Viable Spermatozoa for Intracytoplasmic Sperm Injection (Icsi): an Embryologists Outlook
Javed Aamir¹*, Ashwini LS¹, Debashree Ganguly², Murugan S³, Muthiah SS³, Shabin Kainoth4 and Gopinath Rami Reddy4
¹Department of Embryology, Base Fertility Medical Science Pvt Ltd, India
²Jahar Infertility Research Institute, India
3Institute of Biomedical Research, India
4R & D in Life Sciences Biotechnology Research Lab, Dayananda Sagar Institutions, India
*Corresponding author: Javed Aamir, Department of Embryology, BFMS, KHB Colony, Basaveshwarnagar, Bangalore – 79, India
Received: July 16, 2015; Accepted: August 10, 2015; Published: September 23, 2015
Abstract
Objective: To uncover the best method for real time assessment on immotile but viable spermatozoa for Intracytoplasmic Sperm Injection (ICSI)
Design: Method Development, Comparison and Analysis.
Setting: Base Fertility Medical Science Pvt Ltd
Intervention(S): None.
Material and Method: To help in selection, quite a few techniques have been developed to recognize viable spermatozoa from the immotile portion. Amongst the foremost well-known used approaches are:
(i) The hypo-osmotic swelling test (ii) Modified hypo osmotic swelling test (iii) Chemical compound for initiation of tail actions (iv) The sperm tail flexibility test (v) Laser-assisted immotile sperm selection (vi) Birefringence-polarization microscopy and (vii) Raman micro spectroscopy***.
Interpretation: A healthy and viable spermatozoon is the most importantly required for commencement of fertilization development and progression in ICSI. Usually motility is the crucial and principal sign used to conclude a sperm’s viability. Nevertheless, in every Assisted Reproduction Technology (ART) procedure likely In Vitro Fertilization (IVF) assisted conception units cases are experienced in which not an iota or only little spermatozoa are motile. This inevitable dilemma can occur in treatment cycles (couple undergoing treatment) entailing ejaculated samples but, is utmost frequent and general in cases where surgically extracted testicular spermatozoa are retrieved.
Conclusion: All this can be used routinely in an IVF laboratory with each having both potencies and limitations. The purpose of this petite review is to spot light on the technical concern involved in the routine of each of these techniques and to focus the probenefits and drawbacks of each one approach.
Keywords: Immotile Spermatozoa; Viable spermatozoa; ART and ICSI
Introduction
ICSI involves injecting a live viable healthy single sperm straight into an oocyte in order to achieve fertilization. The fertilized egg that is now an embryo is then transferred to the woman’s uterus for the further development. Viable spermatozoa are a prerequisite for successful ICSI. For an ART laboratory, the principal means of finding a spermatozoon’s vitality is by its ability to be in motion. Subsequently, where the embryologist will be encountered with a dilemma when no noticeable motility can be detected. Absolute asthenozoospermia is caused by two unlike incidents. The first incident is a genetically caused imperfection in the composition of spermatozoa, the upshot is immotility because of the structural incapability of these spermatozoa to be in motion as, for example, observed in kartagener’s syndrome. The erstwhile most vital one is based on very assorted incidents and immotility is a consequence of oxidative stress, infection or several other manipulations [1-3]. Immotility in testicular biopsy samples is common in respect to normal event. Several studies have shown that immotile spermatozoa from the ejaculated samples [4-9] or retrieved from testicular biopsies [10-12] can be used to fertilize oocytes and successfully reproduce viable pregnancies. Though, immotile spermatozoa seem to develop embryos but with lower quality and result in lower pregnancy rates [13]. In every day lab practice the embryologist has unavoidably to face the use of immotile spermatozoa. Nevertheless the potentiality with arbitrarily selected immotile spermatozoa are technically reasonable, the employ of only viable immotile spermatozoa is a extra preferable approach which enhances the success rates for these stern cases. For that cause, quick and simple routine laboratory techniques which can identify between in viable and viable but immotile spermatozoa are compulsory. The current manual of the World Health Organization (WHO-2010) for assessment and processing of human semen enclose no proposals for the identification of immotile but viable spermatozoa. The mere section which not directly pact with vitality testing is the chapter which illustrate benchmark techniques advised for the in vitro diagnostic evaluation of vitality, particularly: the eosin test, which may be used in permutation with negrosin, and the Hypo-Osmotic Swelling (HOS) test. The eosin test categorizes viable spermatozoa principally on the exclusion dye whilst the HOS test assesses membrane properties. Intended for the reason of sperm selection for ICSI, the eosin test is totally unsuitable as it commence a potentially unsafe material onto the spermatozoa. Consequently, quite a lot of research have been available and published which explored substitute techniques which could best be utilized for rapid and simple application in a routine In Vitro Fertilization (IVF) laboratory [14].
Of lately a new technique has been expanded which uses unique polarization microscopy to find variations in head birefringence as marker of acrosome integrity [15,16]. The array of birefringence is reliant on sperm motility and morphology, is associated with intact acrosome reaction. Conversely as this advancement necessitates highly complicated supplemented microscopic paraphernalia which is not generally accessible, as a result this application remains in the weak sphere of most IVF/ ART laboratories.
The foremost commonly used modus operandi are:
(i) The hypo-osmotic swelling test.
(ii) Modified hypo osmotic swelling test.
(iii) The sperm tail flexibility test.
(iv) Chemical stuffs for initiation of tail action.
(v) Laser-assisted immotile sperm selection.
(vi) Birefringence polarization microscopy.
(vii) Raman micro spectroscopy***.
Each of these will be furthermore illustrated in the following segments.
Hypo-Osmotic swelling test: Figure 1a
As a substitute to the dye exclusion test (e.g. Eosin test), HOS test is the single method which is incorporated to the latest edition of the WHO’s 2010 manual. The procedure involves of exposing the spermatozoa to a hypo-osmotic medium (Sperm preparation medium is mixed with sterile water in the ration of 1:1) followed by incubation , the final evaluation is done by microscopic examination .By this mode of action the sperm encounters osmotic challenge which hampers the tails intact membranes to curve or swell which are henceforth viable [17,18] after the physiological changes seen in the sperm the spermatozoa are exposed to normo-osmotic fluid wherein they regain normal shape prior to injection [19]. The WHO manual states that if the spermatozoon has to be clinically used then the incubation time (normally 30 min) has to be reduced to 5 mins. Moreover the method to be followed in an ART setup are not mentioned or specified, but assorted alterations to the fundamental protocol have been recommended [20-23]. An alternative method to the HOS test is commonly practised in ART procedures comprises of the collection of individual spermatozoa with the help of ICSI needle. The collected spermatozoa are transferred in a hypo-osmotic droplet so that their viability are determined by the test (i.e. curved or swollen tail) after that it is taken in the ICSI needle again and transferred to the normal media and finally washed to re-equilibrate it [24].
The main problem arising in the HOS test is the basic principle of the test itself, the nature of the sperm to get curved or swollen due to the hypo-osmotic media which makes the collection of the spermatozoa by an ICSI needle difficult .An alternative method to this problem is to take in the spermatozoa head first into the ICSI needle and partially releasing it from ICSI needle instead of completely placing it in the hypo-osmotic solution so that the initial part of the tail remains inside [25]. However some customized adaptation are also been tried, in which a single spermatozoa is transferred to the HOS solution and once the tail curving is visible is transferred back into the fertilization solution washed and used for the ICSI injection. Some time to observe of this tail curving, as the whole spermatozoa is been place in the HOS solution a new approach can be used to hold the sperm first from the head and only the tail can be place in the HOS solution which will avoid the steric hindrance which in turn help in collecting sperm easily On the other hand if the tail curling happening in the 10-20 sec, then the spermatozoa is taken in the needle swiftly and washed in the medium and place back to the PVP drop. Nevertheless this technique is not 100% trustable some time pseudo positive results are observed [26]. Regardless of the procedure the most important factor is that the washing of the spermatozoa cannot be bypassed, though sometime increases the time of selection.
The result has been observed in all the case like the testicular biopsies (both frozen and fresh) and even in the case of the kartagener’s syndrome [27-29].
Note: This method is not suitable for spermatozoa that have been processed, particularly cryopreserved then thawed or column-washed samples, as these unexpectedly build up tail swellings.
Modified hypo-osmotic swelling test: Figure 1b
A modified hypo-osmotic solution is again one of the solutions for accessing the motility of the viable but immotile spermatozoa and was used to select viable ejaculated and testicular spermatozoa to perform ICSI .Observation from 27 treatment cycles from patients with complete absence of sperm motility. The treatment cycles consisted of 15 cycles in which ejaculated spermatozoa were used and 12 cycles in which testicular spermatozoa were used (Liu et al., 1997). The modified hypo-osmotic solution consisted of 50% culture medium and 50% deionized water and was shown in previous in-vitro studies to be superior to the original solution used in the classical hypo-osmotic swelling test. Fertilization was attained in 37.3% of the oocytes injected. Embryos were placed in 70.4% of the cycles with a mean of 2.0 embryos per cycle. There were no statistically noteworthy differences between the ejaculated sperm subjects and the testicular sperm subjects in the fertilization rate (42.7 versus 30.1%) or in the cleavage rate (92.7 versus 77.3%). Four pregnancies achieved two in the ejaculated sperm group and two in the testicular sperm group, a pregnancy rate of 14.8%. All pregnancies were singletons. It is concluded that the use of this solution to select viable but immotile spermatozoa for ICSI is a simple and practical method and is associated with acceptable fertilization and pregnancy rates [30].
Sperm tail flexibility test: Figure 1c
As specified by the name itself STFT is completely based on the examination of the selected (usually single spermatozoa). In this process the tip of the sperm tail is been slightly touched by the ICSI injecting pipette and the sperm which shows the slight movement irrespective of head movement is been considered as motile and further been used for ICSI. On the other hand if the head moves along with the tail the sperm is considered to be non-viable this technique can be used for both frozen and fresh TESE-ICSI cycles irrespective fertilization rates whether immotile or motile sperm were injected 65.7% vs. 74.3% frozen, 73.4% vs. 64.4% fresh, respectively [30,31].
However the method may not be suited well for the cryopreserved and thawed ejaculated sample because of the random tail curling .The advantages of this physical touch procedure over those that use activating substances are many folds. Foremost the method do not cause any change the functional and structural integrity of spermatozoa,
Secondly as there is no extra additive been added so no cleaning steps are required, so spermatozoa can be injected directly. Finally and most importantly, it will avoid all possible detrimental effects of the additives to the developing embryo. Even so, it must be noted that the technique is not 100% accurate and requires a high level of experience and skills and a time consuming procedure [32,33].
Activating substances: Figure 1d
The scientific use of sperm assortment with chemical compounds has increased consideration recently; exclusively two compounds belonging to the same family of methylxanthine derivatives are applied: theophylline and pentoxifylline mutually induce sperm motility by reducing phosphodiesterase activity and thus escalating intracellular cAMP levels. The aptitude of pentoxifylline to enhance and prolong sperm motility was first identified in the end of 1970s [34,35] with prevalent employ in the following decades. Foremost successful experiments intended at enhancing motility in cases of asthenozoospermia or oligozoospermia where fertilization collapse in preceding IVF cycles was prevail over by incubation with pentoxifylline. Whereas pentoxifylline had an outcome on motility and grade of progression [36,37].
The five focal techniques for immotile sperm assortment: the HOS test and M-HOS, activators for initiation of tail faction movements, the involuntary and mechanical Sperm Tail Flexibility Test (STFT) and the Laser Assisted Immotile Sperm Selection (LAISS). Attentiveness had no affect on acrosome reaction [38]. The relevance of pentoxifylline for immotile spermatozoa in TESE fraction was explained quite a few years later [39] where the supplementation of 3.6 mM pentoxifylline was found to provoke a notable and considerable motility subsequent to 30 min incubation. Pentoxyfylline has also been employed to provoke tail movements in immotile testicular spermatozoa and in ejaculated spermatozoa with very low motility [40]. Specifically, it significantly advanced fertilization rates from 55.9% in the untreated to 66.0% in the treatment group [41]. Unluckily these enhancements were not reflected in the related clinical pregnancy rates, as no difference was found between samples where pentoxifylline was added and non-treated controls. Even though, implantation and pregnancy rates were similar to those of a control group where normal motility was microscopically noticed [42,43] Irrespective of these pronouncements there is no doubt that the supplementation of pentoxifylline significantly decreases the time spent finding and choosing motile spermatozoa. In a study evaluating HOS treated against pentoxifylline recognized spermatozoa, the addition of pentoxifylline established to be radically more successful in terms of both fertilization (62.05% vs. 41.07%) and clinical pregnancy rates 32% vs. 16% [19]. Even though the procedures were revealed to be very consistent, the lesser efficiency of the HOS test was clarified as being caused by the assortment of pseudo positive spermatozoa. Additional to the convenience of pentoxifylline in IVF/ ART, the compound has revealed to also be useful in the finding of spermatozoa with structural defects. Although at preliminary, it seemed unfeasible, nevertheless pentoxifylline was able to stimulate ejaculated spermatozoa from a patient with Kartagener’s syndrome which were in turn selected, used for ICSI and ended in a viable pregnancy [44].
Of lately theophylline, another member of the methylxanthine family has been employed in a forthcoming clinical sibling oocyte tryout where it was noted to have an instantaneous but diminutiveterm effect on sperm motility. Once accumulated the motile spermatozoa were moved to a PVP fall, immobilized by an ICSI injecting pipette and stored until employ. Incubation of immotile frozen - thawed testicular retrieved spermatozoa with theophylline was found to be successful in more or less in all patients treated (98.5%) and proven to have the further advantages of petite finding time, notably increased fertilization rates – mounting from 63.3% to 79.9% in the treatment faction – and also blastulation rates. Also increased implantation and clinical pregnancy rates were accomplished. An apprehension in reverence to the safety and non toxic effect of these chemical compounds has taken place mostly as a consequence of the pronouncements of animal studies. Pentoxifylline concentrations in the micro molar scale had no affect on blastocyst formation during culture and co-culture or on implantation until the egg cylinder stage [45 a,b] but if concentrations increased to the millimolar range, lethal radical effects causing to a two-cell cleavage arrest became prominent. If induced for a shorter time that is 30 min, at these increased concentrations, the block did not emerge and the embryos carry oned to the blastocyst stage, though with lower cell numbers. But yet again, if submitted to uterus they implanted but an blight and impair egg cylinder stage emergence was again obvious [46]. Analogous discovery to the mouse have been described in the hamster, where increased concentrations (i.e. 0.45 and 0.9 nM) of pentoxifylline were set up to negatively influence blastocyst formation at the same time as lower concentrations (23 μM) truly improved embryo development . Thus so far, no proof-based clinical studies assessing the accurate effects of these compounds on embryos and human spermatozoa have been available. Still, if used in a clinical trial where births have been accounted following theophylline or pentoxifylline treatment, no anomalies have been recorded.
Laser assisted immotile sperm selection: Figure 1e
Over the last decade the application of lasers in IVF/ART has come in diverse aspects, from abetting assisted hatching to embryo biopsy, recently it has incident a further revolution as numerous novel applications concerning its use on spermatozoa have come out [47 a,b]. Laser provoked permeabilization and immobilization of the spermatozoa membrane being were the foremost described modus operandi. Membrane permeabilization is a precondition for high-quality good fertilization rates and regularly performed using an ICSI injecting pipette. It is a fairly consistent technique but one which is extremely bended and dependent on the handiness, skill and experience of the embryologist. By swaping the needle with a laser shot of between 2 and 3 mJ (millijoule) strength, the resulting permeabilization can be accomplished with higher precision and correctness while reducing the reliance on the operator’s skill altitude. The worth of the technique was set up and the first birth accounted over decade ago [48,49]. The analogous technique of laser sperm immobilization was principally developed as a way of bypassing the need for PVP, a feasible toxic agent. Here a double shot of a 1.48 lm (luminous flux) wavelength laser is used to firstly stop then permanently arrest the chosen spermatozoon [50]. On the other hand if no such alteration is seen then the spermatozoon is non-viable. The use of the technique on ejaculated asthenozoospermic samples and immotile spermatozoa from testicular biopsies explained that its capacity to identify numbers of viable spermatozoa in a sample was similar to that of the HOS test (22.0% vs. 21.5%). Fertilization rate enhanced significantly from 20.4% in the randomly selected TESE spermatozoa group to 45.4% in the laser selection group; accordingly the take home baby rate increased from 5.9% to 19.0%. The laser can also be functional for cryopreserved ejaculated samples even though the spontaneous curling might reduce the precision (personal experience). An example of the successful application of this laser assortment even in severe conditions was the recognition of viable spermatozoa in a patient with primary ciliary dyskinesia which were subsequently used for ICSI and lead to a pregnancy [51,52].
Birefringence-polarization microscopy: Figure 1f
This modus operandi presupposes that polarization microscopy of a sperm with birefringent head and midpiece (reacted sperm) entails healthy mature sperm. As stated by [53] viable human spermatozoa are naturally and physically birefringent, while in pathological state of affairs, dead, necrotic spermatozoa are lack of birefringence because of the complete deficiency of usual conventional sperm surface and texture. Birefringence capacity may recognize diverse sperm regions because of their different reaction to polarized light (i.e. a normal progressive spermatozoa and immotile but viable spermatozoa illustrates a non-luminous acrosome and a luminous, typical-sized compact nucleus and flagellum) while the luminous acrosome can be seen in the anomalous spermatozoa [54]. In 2009 chattopadhyay [55] confirmed pregnancy rate of 46% when Intra cytoplasmic sperm injection was carried out by injecting a sperm with birefringent head. Moreover, [56] stated that the assortments of birefringent spermatozoa amplify the probability and possibility of recognizing vital spermatozoa having a complete intact DNA even though it is not motile. Nevertheless, more comparative and virtual research studies are required to be performed because this procedure is steeply expensive.
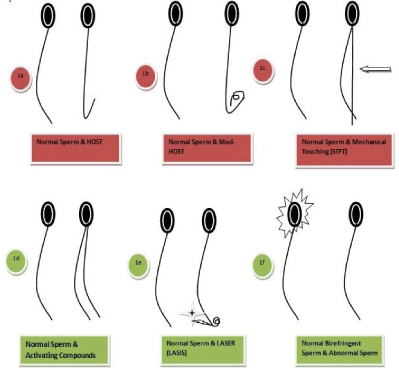
Figure 1: 1a-1f: Sperm selection: Modus Operandi.
a) The hypo-osmotic swelling test.
(b) Modified hypo osmotic swelling test.
(c) The sperm tail flexibility test.
(d) Chemical stuffs for initiation of.tail action.
(e) Laser-assisted immotile sperm.selection.
(f) Birefringence. Polarization microscopy.
Raman micro-spectroscopy***
As a quick, label-free and non-invasive enlightening practice, Raman spectroscopy has been extensively utilized for the examination of biological tissues, any modifications of molecular structure and chemical modules during pathological processes would be revealed via the differences on Raman spectrum. In Andrology, interests in Raman spectroscopy had just come out. In this overview, we abridged the development about the usefulness of Raman spectroscopy in Andrology; the literatures were gathered from various database and Ovid database terms associated with testis, prostate, single sperm cell and seminal plasma. Except for single end testing, applying Confocal Raman Micro spectroscopy (CRM) that merges Raman spectroscopy with a confocal optical microscope, chemical map of single spermatozoa cell might be developed with location of identical attribute being allocated to a précised color. Raman image facilitated definite identification of different parts of sperm, yet very diminutive irregularities could be differentiated, such as vacuoles in the sperm head [57,58].
However this procedure is extremely accurate but highly technical regardless of fertilization process, cleavage rate and takes home baby rate.
Conclusion
Various tests for the assortment and selection of viable immotile spermatozoa subsist, each having their own set of benefits and drawbacks but none gratifying and fulfilling all the requirements of IVF/ART. Which modus operandi is appropriate for which laboratory relies upon the type of procedures undertaken, the equipment accessible and, most vital, the training, dexterity and expertise and proficiency of the personnel. However, concerning the different immotile spermatozoa types the most excellent test is the LAISS. It can be employed even for cryopreserved ejaculated spermatozoa as the impulsive and spontaneous occurring tail swellings do not largely partiality the result of the test.
This is in contrast to the HOS and Mod-HOS test as well as to the STFT and Birefringence microscopy which both are restricted in selecting frozen thawed spermatozoa samples. In use of practicability the technique of choice would also be the laser test, as it is rapid and trouble-free and no further substances have to be used which might cause impairment to the spermatozoa or to the embryos as for example it might be the case with the activating compounds. Furthermore the laser test require no further incubation or treatment of the spermatozoa as it is obligatory with the HOS test or the activating substances. And in terms of reliability and consistency the laser test is undoubtedly superior to the tail flexibility test.
However, further studies need evaluation the laser with the other accessible tests would be needed to support this observation. In spite of which method is used, the six modus operandi illustrated in this petite scientific review have evidently been shown to produce higher fertilization rates and, to some extent, higher pregnancy rates in the complex couples with asthenozoospermia or immotile testicular spermatozoa. As such the integration of in any case one of the described modus operandi should be a necessity of a modern IVF/ ART laboratory.
Raman Micro-Spectroscopy***: A technique as a viable clinical tool still needs additional improvements in this technology.
Acknowledgement
The authors are extremely grateful to Proprietor, Base Fertility Medical Science Pvt Ltd, MIG 1/14, KHB Colony, 1st stage, 1st Cross, 60 ft Road, Havanur Circle Bangalore – 560079.
References
- Aitken RJ, Mattei A, Irvine S. Paradoxical stimulation of human sperm motility by 2-deoxyadenosine. J Reprod Fertil. 1986; 78: 515-527.
- Lecomte PJ, Barthelemy C, Nduwayo L, Hamamah S. Necrospermia: etiology and management. In: Male sterility and motility disorders. Proceeding of the International Symposiun on Male Sterility for Motility Disorders, sponsored by Serono Symposia USA (eds. S Hamamah, R Mieusset, F Olivennes & R Frydman). Springer Publishing Group, Paris, France. 1998; 65–78.
- Ortega C, Verheyen G, Raick D, Camus M, Devroey P, Tournaye H. Absolute asthenozoospermia and ICSI: what are the options? Hum Reprod Update. 2011; 17: 684-692.
- Aparicio NJ. Therapeutical use of pentoxifylline in disturbed male fertility. Singapore. Med J. 1979; 20: 43-51.
- Kahraman S, Tasdemir M, Tasdemir I, Vicdan K, Ozgur S, Polat G, et al. Pregnancies achieved with testicular and ejaculated spermatozoa in combination with intracytoplasmic sperm injection in men with totally or initially immotile spermatozoa in the ejaculate. Hum Reprod. 1996; 11: 1343-1346.
- Nijs M, Vanderzwalmen P, Vandamme B, Segal-Bertin G, Lejeune B, Segal L, et al. Fertilizing ability of immotile spermatozoa after intracytoplasmic sperm injection. Hum Reprod. 1996; 11: 2180-2185.
- Vandervorst M, Tournaye H, Camus M, Nagy ZP, Van Steirteghem A, Devroey P. Patients with absolutely immotile spermatozoa and intracytoplasmic sperm injection. Hum Reprod. 1997; 12: 2429-2433.
- Wang CW, Lai YM, Wang ML, Lee JD, Soong YK. Pregnancy after intracytoplasmic injection of immotile sperm. A case report. J Reprod Med. 1997; 42: 448-450.
- Barros A, Sousa M, Angelopoulos T, Tesarik J. Efficient modification of intracytoplasmic sperm injection technique for cases with total lack of sperm movement. Hum Reprod. 1997; 12: 1227-1229.
- Barkay J, Bartoov B, Ben-Ezra S, Langsam J, Feldman E, Gordon S, et al. The influence of in vitro caffeine treatment on human sperm morphology and fertilizing capacity. Fertil Steril. 1984; 41: 913-918.
- Kahraman S, Isik AZ, Vicdan K, Ozgur S, Ozgun OD. A healthy birth after intracytoplasmic sperm injection by using immotile testicular spermatozoa in a case with totally immotile ejaculated spermatozoa before and after Percoll gradients. Hum Reprod. 1997; 12: 292– 293.
- Shulman A, Feldman B, Madgar I, Levron J, Mashiach S, Dor J. In vitro fertilization treatment for severe male factor: the fertilization potential of immotile spermatozoa obtained by testicular extraction. Hum Reprod. 1999; 14: 749-752.
- Stalf T, Mehnert C, Hajimohammad A, Manolopoulos K, Shen Y, Schuppe HC, et al. Influence of motility and vitality in intracytoplasmic sperm injection with ejaculated and testicular sperm. Andrologia. 2005; 37: 125-130.
- Chan PJ, Tredway DR, Corselli J, Pang SC, Su BC. Combined supravital staining and hypoosmotic swelling test. Hum Reprod. 1991; 6: 1115-1118.
- Correa JR, Zavos PM. The hypoosmotic swelling test: Its employment as an assay to evaluate the functional integrity of the frozen-thawed bovine sperm membrane. Theriogenology. 1994; 42: 351-360.
- Magli MC, Crippa A, Muzii L, Boudjema E, Capoti A, Scaravelli G, et al. Head birefringence properties are associated with acrosome reaction, sperm motility and morphology. Reprod Biomed Online. 2012; 24: 352-359.
- Bourne H, Richings N, Liu DY, Clarke GN, Harari O, Baker HW. Sperm preparation for intracytoplasmic injection: methods and relationship to fertilization results. Reprod Fertil Dev. 1995; 7: 177-183.
- Casper RF, Meriano JS, Jarvi KA, Cowan L, Lucato ML. The hypo-osmotic swelling test for selection of viable sperm for intracytoplasmic sperm injection in men with complete asthenozoospermia. Fertil Steril. 1996; 65: 972-976.
- Mangoli V, Mangoli R, Dandekar S, Suri K, Desai S. Selection of viable spermatozoa from testicular biopsies: a comparative study between pentoxifylline and hypoosmotic swelling test. Fertil Steril. 2011; 95: 631-634.
- Eilts BE. Theoretical aspects of canine semen cryopreservation. Theriogenology. 2005; 64: 692-697.
- Liu J, Tsai YL, Katz E, Compton G, Garcia JE, Baramki TA. High fertilization rate obtained after intracytoplasmic sperm injection with 100% nonmotile spermatozoa selected by using a simple modified hypo-osmotic swelling test. Fertil Steril. 1997; 68: 373-375.
- Sallam HN, Farrag A, Agameya AF, Ezzeldin F, Eid A, Sallam A. The use of the modified hypo-osmotic swelling test for the selection of viable ejaculated and testicular immotile spermatozoa in ICSI. Hum Reprod. 2001; 16: 272–276.
- Esteves SC, Varghese AC. Laboratory handling of epididymal and testicular spermatozoa: What can be done to improve sperm injections outcome. J Hum Reprod Sci. 2012; 5: 233-243.
- Hoskins DD, Hall ML, Munsterman D. Induction of motility in immature bovine spermatozoa by cyclic AMP phosphodiesterase inhibitors and seminal plasma. Biol Reprod. 1975; 13: 168-176.
- Gould KG, Young LG, Hinton BT. Alterations in primate sperm motility with maturation and during exposure to theophylline. Am J Primatol. 1988; 15: 325–336.
- Carreras A, Ramirez JP, Mendoza C. Sperm plasma membrane integrity measurement: a combined method. Andrologia. 1992; 24: 335-340.
- Ved S, Montag M, Schmutzler A, Prietl G, Haidl G, van der Ven H. Pregnancy following intracytoplasmic sperm injection of immotile spermatozoa selected by the hypo-osmotic swelling-test: a case report. Andrologia. 1997; 29: 241-242.
- Jayaprakash D, Kumar KS, Shivaji S, Seshagiri PB. Pentoxifylline induces hyperactivation and acrosome reaction in spermatozoa of golden hamsters: changes in motility kinematics. Hum Reprod. 1997; 12: 2192-2199.
- Westlander G, Barry M, Petrucco O, Norman R. Different fertilization rates between immotile testicular spermatozoa and immotile ejaculated spermatozoa for ICSI in men with Kartagener's syndrome: case reports. Hum Reprod. 2003; 18: 1286-1288.
- Sallam HN, Farrag A, Agameya AF, El-Garem Y, Ezzeldin F. The use of the modified hypo-osmotic swelling test for the selection of immotile testicular spermatozoa in patients treated with ICSI: a randomized controlled study. Hum Reprod. 2005; 20: 3435-3440.
- De Oliveira NM, Vaca Sanchez R, Rodriguez Fiesta S, Lopez Salgado T, Rodriguez R, Bethencourt JC, et al. Pregnancy with frozen-thawed and fresh testicular biopsy after motile and immotile sperm microinjection, using the mechanical touch technique to assess viability. Hum Reprod. 2004; 19: 262–265.
- Kordus RJ, Price RL, Davis JM, Whitman-Elia GF. Successful twin birth following blastocyst culture of embryos derived from the immotile ejaculated spermatozoa from a patient with primary ciliary dyskinesia: a case report. J Assist Reprod Genet. 2008; 25: 437-443.
- Geber S, Lemgruber M, Taitson PF, Valle M, Sampaio M. Birth of healthy twins after intracytoplasmic sperm injection using ejaculated immotile spermatozoa from a patient with Kartagener's syndrome. Andrologia. 2012; 44: 842-844.
- Koutsarova N, Todorov P, Koutsarov G. Effect of pentoxifylline on motility and longevity of fresh and thawed dog spermatozoa. J Reprod Fertil Suppl. 1997; 51: 117-121.
- De Turner E, Aparicio NJ, Turner D, Schwarzstein L. Effect of two phosphodiesterase inhibitors, cyclic adenosine 3':5'-monophosphate, and a beta-blocking agent on human sperm motility. Fertil Steril. 1978; 29: 328-331.
- Kruger TF, Menkveld R, Stander FS, Lombard CJ, Van der Merwe JP, van Zyl JA, et al. Sperm morphologic features as a prognostic factor in in vitro fertilization. Fertil Steril. 1986; 46: 1118-1123.
- Yovich JM, Edirisinghe WR, Cummins JM, Yovich JL. Preliminary results using pentoxifylline in a pronuclear stage tubal transfer (PROST) program for severe male factor infertility. Fertil Steril. 1988; 50: 179–181.
- Mladenovic I, Micic S, Pearson RM, Genbacev O, Papic N. Effects of pentoxifylline on human sperm parameters in vitro. J Assist Reprod Genet. 1994; 11: 495-499.
- Lewis SE, McKinney KA, Thompson W. Influence of pentoxifylline on human sperm motility in asthenozoospermic individuals using computer-assisted analysis. Arch Androl. 1994; 32: 175-183.
- Nassar A, Mahony M, Morshedi M, Lin MH, Srisombut C, Oehninger S. Modulation of sperm tail protein tyrosine phosphorylation by pentoxifylline and its correlation with hyperactivated motility. Fertil Steril. 1999; 71: 919-923.
- Kovacic B, Vlaisavljevic V, Reljic M. Clinical use of pentoxifylline for activation of immotile testicular sperm before ICSI in patients with azoospermia. J Androl. 2006; 27: 45-52.
- Terriou P, Giorgetti HC, Spach JL, Salzmann J, Urrutia V, Roulier R, et al. Pentoxifylline initiates motility in spontaneously immotile epididymal and testicular spermatozoa and allows normal fertilization, pregnancy, and birth after intracytoplasmic sperm injection. J Assist Reprod Genet. 2000; 17: 194–199.
- Griveau JF, Lobel B, Laurent MC, Michardière L, Le Lannou D. Interest of pentoxifylline in ICSI with frozen-thawed testicular spermatozoa from patients with non-obstructive azoospermia. Reprod Biomed Online. 2006; 12: 14-18.
- Okada H, Tatsumi N, Kanzaki M, Fujisawa M, Arakawa S, Kamidono S. Formation of reactive oxygen species by spermatozoa from asthenospermic patients: response to treatment with pentoxifylline. J Urol. 1997; 157: 2140-2146.
- (a) Tournaye H, Van der Linden M, Van den Abbeel E, Devroey P, Van Steirteghem A. Effects of pentoxifylline on in-vitro development of preimplantation mouse embryos. Hum Reprod. 1993; 8: 1475–1480. (b) Tournaye H, Van der Linden M, Van den Abbeel E, Devroey P & Van Steirteghem A. Effect of pentoxifylline on implantation and post-implantation development of mouse embryos in vitro. Hum Reprod. 1993; 8: 1948–1954.
- Negri P, Grechi E, Tomasi A, Fabbri E, Capuzzo A. Effectiveness of pentoxifylline in semen preparation for intrauterine insemination. Hum Reprod. 1996; 11: 1236-1239.
- (a) Montag M, Rink K, Delacretaz G, van der Ven H. Laser-induced immobilization and plasma membrane permeabilization in human spermatozoa. Hum Reprod. 2000; 15: 846–852. (b) Montag M, Klose R, Koster M, Rosing B, van der Ven K, Rinck K, et al. Application of non-contact laser technology in assisted reproduction. Med Laser Appl. 2009; 24: 57–64.
- Yildirim G, Ficicioglu C, Akcin O, Attar R, Tecellioglu N, Yencilek F. Can pentoxifylline improve the sperm motion and ICSI success in the primary ciliary dyskinesia? Arch Gynecol Obstet. 2009; 279: 213-215.
- Ebner T, Moser M, Tews G. Possible applications of a non-contact 1.48 microm wavelength diode laser in assisted reproduction technologies. Hum Reprod Update. 2005; 11: 425-435.
- Vazquez JM, Martinez EA, Martinez P, Garcia-Artiga C, Roca J. Hypoosmotic swelling of boar spermatozoa compared to other methods for analysing the sperm membrane. Theriogenology. 1997; 47: 913-922.
- Pérez-Llano B, Lorenzo JL, Yenes P, Trejo A, García-Casado P. A short hypoosmotic swelling test for the prediction of boar sperm fertility. Theriogenology. 2001; 56: 387-398.
- Gerber PA, Kruse R, Hirchenhain J, Krussel JS, Neumann NJ. Pregnancy after laser-assisted selection of viable spermatozoa before intracytoplasmatic sperm injection in a couple with male primary cilia dyskinesia. Fertil Steril. 2008; 89: 1826.
- Baccetti B. Microscopical advances in assisted reproduction. J Submicrosc Cytol Pathol. 2004; 36: 333-339.
- Collodel G, Federico MG, Pascarelli NA, Geminiani M, Moretti E. Natural sperm birefringence can be used to estimate sperm viability and morphology. Syst Biol Reprod Med. 2010; 56: 465-472.
- Chattopadhyay R, Ghosh S, Goswami SK, Goswami M, Chakravarty B. Selection of birefringent sperm head under polscope and its effect on outcome of ICSI in azoospermia and complete asthenozoospermia. Hum Reprod. 2009; 24: 70-73.
- Crippa A, Magli MC, Paviglianiti B, Boudjema E, Ferraretti AP, Gianaroli L. DNA fragmentation and characteristics of birefringence in human sperm head. Hum Reprod. 2009; 24: i95. O-234.
- Kubasek WL, Wang Y, Thomas GA, Patapoff TW, Schoenwaelder KH, Van der Sande JH, et al. Raman spectra of the model B-DNA oligomer d(CGCGAATTCGCG)2 and of the DNA in living salmon sperm show that both have very similar B-type conformations. Biochemistry. 1986; 25: 7440-7445.
- Aamir Javed, Ashwini LS. Oocyte Degeneration Subsequent Intracytoplasmic Sperm Injection (ICSI). Medical Science. 2014; 13: 9-15.