Case Report
Austin J Med Oncol. 2014;1(1): 3.
A Case of Bronchial Wall Angiosarcoma with Complete Response to Pazopanib
Braik T1*, Chechani V3, Badine E1, Sussman Z4 and Williams M2
1Department of Hematology and Medical Oncology, Texas Tech. University, USA
2Department of Pathology, Head & Neck section, UT MD Anderson Cancer Center, USA
3Department of Internal Medicine, Eastern New Mexico Medical Center, USA
4Department of Pathology, Eastern New Mexico Medical center, USA
*Corresponding author: Braik T, Department of Hematology and Medical Oncology, Texas Tech. University, Kymera Cancer Center, 407 W. Country Club Rd, Roswell, NM 88201, USA
Received: July 19, 2014; Accepted: September 03, 2014; Published: September 05, 2014
Abstract
Angiosarcomas are a rare subtype of sarcoma, which are mesenchymal derived tumors. Specifically, these malignant and highly aggressive tumors arise from endothelial-cell vascular or lymphatic origin. Because of their rapid clinical course, treatment is often challenging and the prognosis remains poor. As vascular derived neoplasms, angiosarcomas typically express endothelial markers such as vascular endothelial growth factor (VEGF), an observation that has stimulated an interest in using therapeutic agents that targets angiogenic pathways in this rare disease. We present a case of angiosarcoma originating in the lumen of the right main bronchus that was successfully treated with a multi-targeted receptor tyrosine kinase inhibitor Pazopanib after failing first line chemotherapy.
Keywords: Angiosarcoma; Lung cancer; Pazopanib
Case Presentation
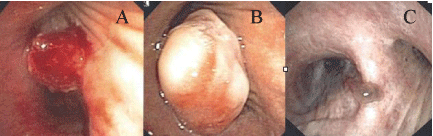
A 71-year-old man presented with significant hemoptysis of two days duration. His medical history was significant for severe emphysema from chronic tobacco abuse. His chest X-ray was negative for evidence of infection or malignancy. He underwent a diagnostic bronchoscopic evaluation, which revealed an actively bleeding irregularly surfaced lesion arising from the posterior wall of right main bronchus and the right upper lobe carina (Figure 1A). Bronchial brushing and tissue biopsy were performed and tissue was sent for pathologic examination.
Figure 1: tumor initial appearance at diagnosis, B. tumor after failure of paclitaxel and bevacizumab, C.Tumor after treatment with Pazopanib
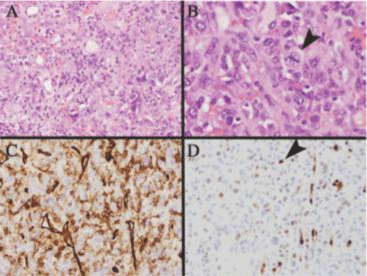
On pathologic review, the tissue was highly cellular with irregular epithelioid cells with high nuclear to cytoplasmic ratio, growing in sheets (Figure 2). Mitotic figures were frequent with abnormal forms present. The background consisted of blood, necrosis and inflammation. While a high-grade malignancy was confirmed histologically, the tumor lineage was uncertain and required immunohistochemical evaluation for further characterization. Immunohistochemical markers associated with carcinomas (keratins) were negative as were melanoma markers (s100, HMB45 and Mart1). Secondary to the discohesion within the tumor and hemorrhagic nature of the tumor, a CD31 vascular marker was performed and showed the tumor was diffusely positive. ERG, a transcription factor show to be expressed in endothelial lesions [1] also showed focal nuclear positivity confirming vascular origin and final classification as angiosarcoma.
Figure 2: Pathologic evaluation of angiosarcoma: A-B) hematoxylin and eosin stained tissue sections showing large tumor cells admixed with inflammation and hemorrhage. An abnormal mitoses is seen (B at arrow). C-D) Immunohistochemical evaluation shows the tumor cells are C) diffusely positive for CD31 a vascular marker (brown cytoplasmic staining reaction), and D) positive in the nuclei (at arrow) for ERG a transcription factor seen in endothelial lesion. Both immunomarkers support lineage and classification as angiosarcoma.
PET/CT scans for evaluation of distant metastases only showed small nodules in the right main stem bronchus with increased FDG activity. As the patient was not a surgical candidate because of his severe emphysema he was started on paclitaxel 80 mg/m2 I.V days 1,8,15 every 21 days and bevacizumab 15 mg/kg I.V every 21 days. After 3 cycles of treatment, bronchoscopy was repeated to evaluate tumor response. The tumor had markedly grown and became pedunculated and prevented entry to the bronchus intermedia and right upper lobe with the bronchoscope (Figure 1B). No active bleeding was noted. A new polypoidal lesion was seen in left main stem bronchus. Owing to the refractory nature of his tumor, Pazopanib was started at 800 mg PO once daily and subsequently held on day 21 secondary to grade 3 mucositis and liver toxicity. Pazopanib was restarted a week later at a 600 mg with no further complications.
Three months after initiating pazopanib, bronchoscopy was repeated which showed a marked response of the tumor to pazopanib with complete resolution of the tumor in the right main bronchus (Figure 1C). The patient remains asymptomatic and tolerating 600 mg of once daily pazopanib and his repeat CT scan at eight months since diagnosis shows no evidence of metastatic disease.
Discussion
Angiosarcoma is a rare malignancy. Because soft tissue sarcomas in aggregate account for approximately 1% of adult malignancies [2], the number of cases of any particular histologic type is exceedingly small. Angiosarcoma, as a specific entity, has been associated with radiation therapy (i.e. following breast cancer treatment or radiation at other sites for other malignancies), may arise in long standing tissues with lymphedema, or in sun exposed regions particularly the head and neck and may also arise de novo without associated risk factor. Secondary to their rarity, there are no randomized trials and few prospective studies for angiosarcoma and most published reports of treatment are retrospective case series. No evidence-based recommendations can be made for specific angiosarcoma subtypes and patients with this rare disease should be referred to specialist centers. Radical surgery with complete (R0) resection is the primary treatment of choice; however the tumors often infiltrate beyond what visually apparent leading to difficulty is obtaining complete resection. Cytotoxic chemotherapy is the primary treatment option for metastatic angiosarcoma, although the evidence base for this is limited [3]. The main drug groups used are anthracyclines, ifosfamide, and taxanes. Over the past decade there has been growing interest in the use of taxane chemotherapy for the treatment of angiosarcoma. Response rates in other soft-tissue sarcomas have been disappointing, but taxanes have antiangiogenic activity and are therefore of particular interest in the management of angiosarcomas. The only prospective phase II study of chemotherapy for angiosarcoma, investigated the use of weekly paclitaxel in 30 patients with locally advanced and metastatic disease [4]. Of these patients, 19 (63%) were chemotherapy-naive, 11 (37%) had previously received anthracycline-based chemotherapy. Only five (17%) showed a partial response; however, three of these patients proceeded to surgery and two showed a pathological-complete response.
Areas of relative hypoxia and markers of dysfunctional angiogenesis are common features of soft tissue sarcomas. Because angiosarcomas are endothelial-cell tumors, there is great interest in the role of angiogenesis and the angiogenic factors associated with their pathogenesis, and how they might be used as targets for treatment. Results of a phase II study of single-agent bevacizumab (a monoclonal antibody against vascular endothelial growth factor VEGF-A) suggested significant activity [5]. Use of broad-spectrum tyrosine-kinase inhibitors to target VEGFRs has been assessed in several phase II studies. The largest of these included 37 angiosarcomas within a larger study of 122 advanced-soft-tissue sarcomas treated with sorafenib. Partial responses were seen in four patients and one complete response was seen within the angiosarcoma group, giving an overall-response rate of 14% with a median progression-free survival of 3.8 months [6]. Thalidomide, an immunomodulatory drug with antiangiogenic effects and known therapeutic efficacy in other sarcomas such as kaposi’s sarcoma, also has been tried with successful results [7].
We report a case of bronchogenic angiosarcoma with complete response to pazopanib. Preclinical studies have consistently described aberrant expression of pazopanib target kinases in many histologic sub-types. Angiosarcoma cells exhibit overexpression of multiple components of the VEGF pathway, including VEGFRs, hypoxia-inducible factor 1-a, TIE-1 and TIE-2 [8]. Pazopanib is a multi-tyrosine kinase inhibitor of vascular endothelial growth factor receptor VEGFR-1, VEGFR-2, VEGFR-3, PDGFR-a and -b, fibroblast growth factor receptor-1 and -3, and KIT as well as other emerging targets, including B-RAF proteins. Preclinical models of pazopanib demonstrated inhibition of ligand-induced auto-phosphorylation of VEGFR-2, KIT, and PDGFR-b receptors, and impaired FGF and VEGF-mediated angiogenesis as well as xenograft growth in multiple human cancers [9]. Pazopanib is the first molecular targeted agent approved by the FDA for sarcomas since imatinib for GIST with median progression free survival of 4.6 months in a landmark phase III study (the PALETTE trial) [10]. The large study population enrolled to ensure statistical power led to increased in the heterogeneity of the population and diluted the impact of the isolated patients with superior benefit. However, in the PALETTE study, histologic subtype (leiomyosarcoma vs. synovial sarcoma vs. other) did not correlate with PFS, although the specific histologies of the 6% of patients who achieved partial response were not reported. Our case and other case reports detail dramatic responses in rarer sarcomas that are generally lumped into ill-characterized ‘other’ categories: a patient with pericardial angiosarcoma obtained a durable partial remission with pazopanib following paclitaxel chemotherapy, with stable disease over a 10-month follow-up period [11]. The complete response seen with pazopanib rather than bevacizumab in this case could be due to the difference in the mechanism of disrupting tumor angiogenesis between the two drugs, pazopanib targets multiple vascular endothelial growth factor receptors, while bevacizumab targets the ligand of these receptors. The Dramatic response seen in our case and the preclinical rationale in angiosarcomas strongly supports clinical investigation of pazopanib and other multitargeted tyrosine kinase inhibitors.
Conclusion
Pazopanib should be evaluated in a prospective study in the first line treatment of angiosarcomas.
References
- Miettinen M, Wang ZF, Paetau A, Tan SH, Dobi A, et al. ERG transcription factor as an immunohistochemical marker for vascular endothelial tumors and prostatic carcinoma. Am J SurgPathol. 2011; 3: 432-441.
- Budd GT. Management of Angiosarcoma. Current Oncology Reports. 2002; 4: 515-519.
- Young RJ, Brown NJ, Reed MW, Hughes D, Wall PJ. Angiosarcoma. Lancet Oncol. 2010; 11: 983-991.
- Penel N, Bui BN, Bay JO, Cupissol D, Ray-Coquard I, et al. Phase II trial of weekly paclitaxel for unresectable angiosarcoma: the ANGIOTAX Study. J Clin Oncol. 2008; 26: 5269-5274.
- Agulnik M, Okuno SH, Von Mehren M. An open-label multicenter phase II study of bevacizumab for the treatment of angiosarcoma and epithelioidhemangioendotheliomas. Ann Oncol. 2013; 24: 257-263.
- Maki RG, D’Adamo DR, Keohan ML, Saulle M, Schuetze SM, et al. Phase II study of sorafenib in patients with metastatic or recurrent sarcomas. J Clin Oncol. 2009; 27: 3133-3140.
- Raina V, Sengar M, Shukla NK, Deo SS, Mohanty BK, et al. Complete Response From Thalidomide in Angiosarcoma After Treatment of Breast Cancer. J Clin Oncol. 2007; 25: 900-901.
- Park M, Ravi V, Araujo D. Inhibiting the VEGF-VEGFR pathway in angiosar-coma, epithelioid hemangioendothelioma, and hemangiopericytoma/solitary fibrous tumor. Curr Opin Oncol. 2010; 22: 351-355.
- Wilky BA, Meyer CF, Trent JC. Pazopanib in sarcomas: expanding the PALETTE. Curr Opin Oncol. 2013; 25: 373-378.
- Van der Graaf WT, Blay JY, Chawla SP, Kim DW, Bui-Nguyen B, et al. Pazopanib for metastatic soft-tissue sarcoma (PALETTE): a randomised, double-blind, placebo-controlled phase III trial. Lancet. 2012; 379: 1879-1886.
- Ong P, Greulich S, Schumm J, Backes M, Kaufmann M, et al. Favorable Course of Pericardial Angiosarcoma Under Paclitaxel Followed by Pazopanib Treatment Documented by Cardiovascular Magnetic Resonance Imaging. Circulation. 2012; 126: 279-281.