
Case Report
Austin J Med Oncol. 2015;2(1): 1015.
Gastric Adenocarcinoma in an Excluded Stomach Diagnosed by Double Balloon Enteroscopy
Deena Midani1, Adam C. Ehrlich1, Truptesh H Kothari2* and Stephen J. Heller3
1Temple University School of Medicine, Philadelphia, PA,USA
2University of Rochester Medical Center, Rochester, NY,USA
3Fox Chase Cancer Center, Philadelphia, PA, USA
*Corresponding author: Truptesh H. Kothari MD, MS, Assistant Professor of Medicine, Director, Developmental Endoscopy Lab at University of Rochester (DELUR), Division of Gastroenterology and Hepatology, University of Rochester Medical Center, Rochester, NY 14642, USA
Received: February 26, 2015; Accepted: April 06, 2015; Published: April 08, 2015
Abstract
Gastric bypass is a common surgical procedure for obesity. A patient presented many years after a Roux-en-Y gastric bypass surgery with clinical evidence of metastatic malignancy of unknown primary. When biopsy of a liver lesion suggested a gastrointestinal primary tumor, we performed an upper double balloon enteroscopy to examine the excluded stomach and found a primary gastric cancer. To our knowledge, this is the first report of using this method to identify such a tumor, and this report demonstrates the feasibility of such an approach.
Introduction
Gastric bypass with Roux-en-Y reconstruction (RYGB) is an increasingly common procedure for the treatment of morbid obesity [1,2]. Although rare, gastric cancers developing in the excluded stomach have been reported in the RYGB patient population. These cancers often present in advanced stages due to limited accessibility of the excluded stomach to direct visualization [3]. This case demonstrates the ability to diagnose gastric cancer in the excluded stomach without surgery using upper double balloon enteroscopy (UDBE).
Case Report
A 43 year old female with family history of ovarian and endometrial cancers initially presented with nausea, anorexia, and severe right lower quadrant pain. She had undergone RYGB fourteen years earlier but at presentation was morbidly obese by body mass index (BMI). Initial evaluation at an outside institution, including routine upper endoscopy exploring the gastric pouch and colonoscopy, was normal. Imaging including ultrasound and CT scan of the abdomen and pelvis, also done at an outside institution, revealed a right ovarian mass, abdominal and pelvic ascites, multiple liver nodules and omental nodularity concerning for carcinomatosis. Ultrasoundguided core needle biopsy of a liver lesion at our institution showed moderately differentiated adenocarcinoma with tumor cells positive for CK7, PCEA, CDX-2, CK-19, and focal CK20suggestive of a primary gastrointestinal or biliary neoplasm. Review of her previous CT scan suggested a mass in her excluded stomach (Figure 1).
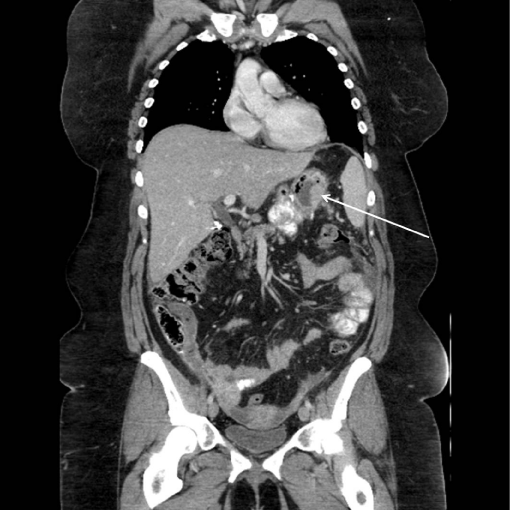
Figure 1: Coronal CT scan with thickening of gastric wall in excluded
stomach.
Options for examining this portion of her stomach included attempting double balloon enteroscopy, CT-guided percutaneous biopsy, or laparoscopic-assisted transgastric endoscopy (with a surgically-placed access port into the stomach). We elected to pursue upper double balloon enteroscopy (UDBE).
After traversing the relatively short Roux limb with the double balloon enteroscope, we identified the biliopancreatic limb and reached the excluded stomach via a retrograde approach (Figure 2). Upon traversing the pylorus, we found a large, fungating, infiltrating tumor that encompassed much of the antrum and body of the stomach (Figure 3). Biopsies of this mass confirmed poorlydifferentiated gastric adenocarcinoma (Figure 4) and were consistent with the pathologic diagnoses of the initial core liver biopsy specimen and pleural and ascitic fluid cytology. Immunohistochemical stains of the gastric specimen revealed an overall equivocal HER2 status, later confirmed to be negative by FISH analysis.
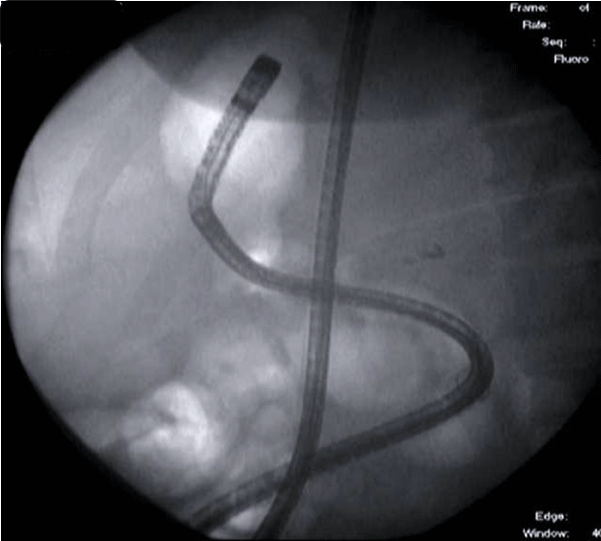
Figure 2: Fluoroscopic image of double balloon enteroscope in stomach via
retrograde approach (note: gastric bubble).
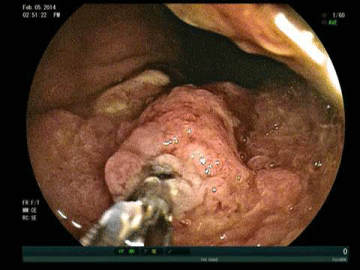
Figure 3: Large, fungating, infiltrating tumor in antrum and body of excluded
stomach.
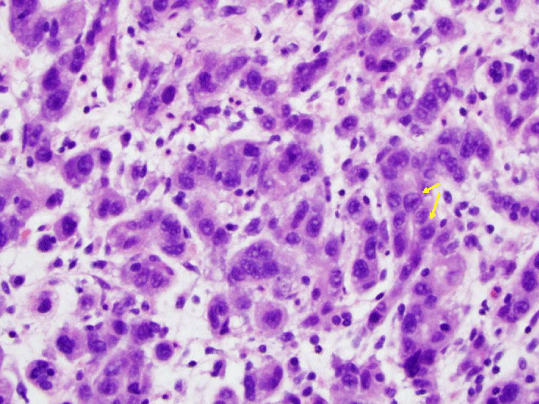
Figure 4: Pathology specimen stained with hematoxylin and eosin revealing
poorly-differentiated gastric adenocarcinoma.
The patient elected to receive an initial round of palliative chemotherapy at our institution and pursue additional care with her local oncologist. Despite further treatment, she died four months later.
Discussion
Although rare, there are several case reports of gastric cancers arising in the excluded stomach of patients who have undergone RYGB [3-11]. A recent review of the literature by Kuruba et al. [3] includes eight cases of cancer diagnosed in the excluded stomach among other reports of esophagogastric cancers following gastric bypass surgeries. The mean age of these patients was 54, ranging from 38 to 71. Five patients were female and three male. Five of the eight patients underwent RYGB as their initial bariatric procedure. Three of these five patients ultimately died from their cancer. The authors emphasize their concern regarding the delay in diagnosis in these patients. In this series, the mean duration of symptoms prior to the diagnosis of malignancy was 18 months [10,11].
Diagnosis may be delayed due to failure to recognize symptoms in a timely manner. In their review of the literature, Scozzari et al. [10] hypothesize that early symptoms related to underlying malignancy are mistaken for routine post-operative complaints. Furthermore, patients may mistakenly attribute ongoing weight loss to the expected results of their RYGB rather than an underlying malignancy [10]. Therefore careful attention must be paid to patients with new or evolving upper gastrointestinal complaints in the years following bariatric surgery.
These cancers usually present in advanced stages due to the difficulty in evaluation of the excluded stomach and have traditionally been identified either by laparoscopic-assisted endoscopy or by surgery. These surgical procedures are often technically difficult and are associated with operative morbidity. Although timeconsuming, UDBE offers the ability to evaluate the excluded stomachendoscopically without additional surgery [12,13]. UDBE may not be feasible in patients with a long roux limb as part of their surgical reconstruction. In these patients, surgery remains a viable option [12,13].
To our knowledge, this is the first reported case of using UDBE to diagnose gastric adenocarcinoma in an excluded stomach from RYGB. This case highlights the importance of considering a neoplasm arising in a surgically excluded stomach as part of the differential diagnosis during evaluation of a potential gastrointestinal tumor. It demonstrates that UDBE is a safe and feasible method to access the excluded stomach and pancreato-biliary tree. In the appropriate clinical setting, endoscopic surveillance of the excluded stomach is recommended following RYGB. Larger case series confirming the safety and feasibility of this technique would be useful.
References
- Livingston EH. Procedure incidence and in-hospital complication rates of bariatric surgery in the United States. Amer J Surg 2004; 188: 105-110.
- Santry HP, Gillen DL, Lauderdale DS. Trends in bariatric surgery procedures. JAMA 2005; 294: 1909-1917.
- Kuruba R, Jawad M, Karl RC, Murr MM. Technique of resection of esophageal adenocarcinoma after Roux-en-Y gastric bypass and literature review of esophagogastric tumors after bariatric procedures. Surg Obes Relat Dis 2009; 5: 576-81.
- Abellan I, Ruiz de Angulo D, Parrilla P. Incidental gastric gastrointestinal stromal tumor (GIST) in the excluded stomach after Roux-en-Y gastric bypass: a case report and review of the literature. Surg Obes Relat Dis 2014; 10: e13-4.
- Watkins BJ, Blackmun S, Kuehner ME. Gastric adenocarcinoma after Roux-en-Y gastric bypass: access and evaluation of excluded stomach. Surg Obes Relat Dis 2007; 3: 644-647.
- Corsini DA1, Simoneti CA, Moreira G, Lima SE Jr, Garrido AB. Cancer in the excluded stomach 4 years after gastric bypass. Obes Surg 2006; 16: 932-934.
- Escalona A1, Guzmán S, Ibáñez L, Meneses L, Huete A, et al. Gastric cancer after Roux-en-Y gastric bypass. Obes Surg 2005; 15: 423-427.
- Khitin L, Roses RE, Birkett DH. Cancer in the gastric remnant after gastric bypass: a case report. Curr Surg 2003; 60: 521-523.
- Raijman I, Strother SV, Donegan WL. Gastric cancer after gastric bypass for obesity: case report. J Clin Gastroenterol 1991; 13: 191-194.
- Scozzari G, Trapani R, Toppino M, Morino M Esophagogastric cancer after bariatric surgery: systematic review of the literature. Surg Obes Relat Dis 2013; 9: 133-142.
- Kuga R, Safatle-Ribeiro AV, Faintuch J, Ishida RK, Furuya CK Jr, et al. Endoscopic findings in the excluded stomach after Roux-en-Y gastric bypass surgery. Arch Surg 2007; 142: 942-946.
- Elton E, Hanson BL, Qaseem T, Howell DA Diagnostic and therapeutic ERCP using an enteroscope and a pediatric colonoscope inlong-limb surgical bypass patients. Gastrointest Endosc 1998; 47: 240–249.
- Moreels TG, Hubens GJ, Ysebaert DK, Op de Beeck B, Pelckmans PA Diagnostic and therapeutic double-balloon enteroscopy after small bowel Roux-en-Y reconstructive surgery. Digestion 2009; 80: 141-147.