
Case Report
Austin J Med Oncol. 2015; 2(2): 1020.
Coexistence of Acute Lymphoblastic Leukemia with Lupus Erythematosus in a Male Child: A Rarest of Rare Association
Narayan S¹*, Murli P¹, Kapoor A¹, Sharma N¹ and Beniwal S²
¹Department of Radiation Oncology, S.P. Medical College and Associated Group of Hospital, India
²Department of Medical Oncology; S.P. Medical College and Associated Group of Hospital, India
*Corresponding author: Satya Narayan, Department of Radiation Oncology, S.P. Medical College, Room no 26, PG Hostel, PBM Hospital Campus, Bikaner, Rajasthan, India
Received: February 02, 2015; Accepted: August 28, 2015; Published: September 05, 2015
Abstract
The association of systemic lupus erythematosus (SLE) and acute lymphoblastic leukemia (ALL) is rarely reported in literature for the pediatric age group. Usually, SLE precedes the onset of lymphoproliferative disease, but the malignancy can occur earlier or even simultaneously. We report a case of 15 year old male child 12 year back diagnosed as SLE, recently on routine investigation incidentally diagnosed as ALL.
SLE is an autoimmune disease characterized by inflammation of blood vessels and connective tissue resulting in multisystem involvement. The clinical manifestations are extremely variable. An estimated 10% to 20% of patient’s diagnosis or onset of SLE prior to adulthood. Childhood SLE affects girls more often than boys (8:1), even in the prepubescent age group (4:1). The childhood SLE is usually more severe and has a poorer prognosis than adult. The hallmark of SLE is presence of antinuclear antibody (ANA). The association of SLE and acute lymphoblastic leukemia (ALL) in pediatric patients only few cases reported in literature. Usually, SLE precedes the onset of lymphoproliferative disease, but the neoplasia can occur earlier or even simultaneously.
Keywords: Systemic lupus erythematosus; Acute lymphoblastic leukemia; Anti-double stranded DNA
Case Presentation
A 15 year old male child was a known case of lupus nephritis, on routine follow up complete blood count reveled high TLC. Peripheral blood film analysis suggests acute lymphoblastic leukemia (Figure 1). On flowcytometry analysis shows 60% blasts which are CD79a+, CD45+, CD34+, HLADR+, CD20+, CD38+, CD19+, CD10+, CD22 HTG + were positive and CD13,33,3 and MPO were negative. The flocytometry suggests B cell acute lymphoblastic leukemia. The karyotype of child was 46XY and no structural abnormality, no hypo or hyper diploidy was seen (Figure 2). The Philadelphia chromosome was not evident in any of the metaphases. There was no evidence of t(15:17), t(8:21) or inversion 16. The sibling was also diagnosed ALL at the age of 5 years. Twelve year back the child developed rash over face and body with vomiting and loose stools. The clinical examination was without any finding. On kidney biopsy the glomeruli show mesengial expansion with increase in mesengial cellularity and segmental basement membrane thickening. Blood vessels show mild medial thickening. Interstitial shows focal space inflammatory infiltrate. There was no segmental or diffuse proliferation or crescent formation or tuft necrosis of the glomeruli. The mentioned histopathology is suggestive of lupus nephitis (WHO class II). Antinuclear antibody (ANA) and anti-double stranded DNA (anti dsDNA) were both positive. The patient was on prednisone for SLE from initially but after deliberation of disease the steroid was stopped 6 months back.
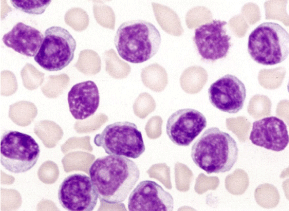
Figure 1: Peripheral blood film analysis suggests acute lymphoblastic
leukemia.
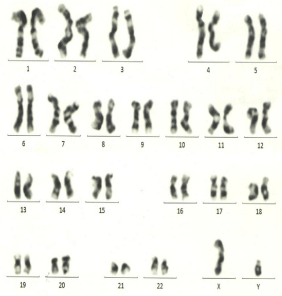
Figure 2: Karyotype of child without any structural abnormality.
Discussion
Systemic lupus erythematosus (SLE) is an autoimmune disease of unknown origin. The immune system mistakenly attacks the body’s tissues itself and causes inflammation (swelling) in the skin, kidneys, lungs, nervous system, and other organs [1]. Although it can occur at any age, SLE becomes more frequent after five years of age and is increasingly prevalent after the first decade of life [2]. SLE affects children in the same ways as adults. The main difference being the difference in care of children from the adults as treatment can have a great impact on a child’s physical and psychological growth and development. Early damage is mostly related to disease whereas late damage-namely infections, atherosclerosis, and malignanciesis usually related to complications of longstanding disease and treatment related. SLE may occasionally present with high fever and lymphadenopathy simulating lymphoid or hematological malignancy. Hematological malignancies (particularly non- Hodgkin’s lymphoma (NHL) and rarely lymphoproliferative disease) and cervical and lung cancer occur more commonly in SLE compared to the general population. Most of the data show that the malignancy is detected following the diagnosis and treatment of SLE [3]. NHL is associated with SLE with the most commonly identified histologic subtype being diffuse large B cell lymphoma, which usually runs an aggressive course. Researchers found that people with lupus were 3.2 times more likely to develop lymphomas than the general population and 3.4 times more likely to develop non-Hodgkin’s lymphoma and were 1.7 times greater risk of developing leukemia [4]. Bernatsky et al. reported an increased risk of all hematologic malignancies among an international cohort of 9,547 SLE patients. Only a few patients with coexistent chronic lymphatic leukemia/chronic myeloid leukemia and concomitant SLE have been reported. Patients were followed for eight years and the calculated leukemia standardized incidence ratio was 1.89 (95% CI 0.76–3.88) [5]. The risk for hematological malignancies may increase after exposure to immunosuppressive medications, particularly after a period of 5 years following cessation of therapy. The renal involvement is a dreaded complication of SLE and the main cause of death is irreversible renal damage [6]. Prednisone is one of the most effective treatments for controlling inflammation in lupus and has to be continued for many years. After careful deliberation, the maintenance steroids can be stopped.
References
- Maheshwari A, Pandey M, Rath B, Chandra J, Singh S, Sharma S. Clinical and laboratory observation systemic lupus erythematosus and acute lymphocytic leukemia: An unusual case. Indian J Med Paediatr. Oncol 2011; 32: 154-56.
- Font J, Cervera R, Espinosa G, Pallares L, Casals MR, Jimenez S, et al. Systemic lupus erythematosus in childhood: analysis of clinical and immunological findings in 34 patients and comparison with SLE characteristics in adults. Ann Rheum Dis. 1998; 57: 456-59.
- Vasquez S, Kavanaugh AF, Schneider NR, Wacholtz MC, Lipsky PE. Acute nonlymphocytic leukemia after treatment of systemic lupus erythematosus with immunosuppressive agents. J Rheumatol. 1992; 19: 1625-27.
- https://www.rheumatology.org/about/newsroom/2010/2010_people_with_lupus_might_be_at_a_greater_risk_for_cancer.asp
- Bernatsky S, Ramsey-Goldman R, Clarke A. Exploring the links between systemic lupus erythematosus and cancer. Rheum Dis Clin North Am. 2005; 31: 387–402.
- Singh S,Devidayal, Kumar L, Joshi K. Mortality Patterns in Childhood Lupus – 10 Years’ Experience in a Developing Country. Clin Rheumatol. 2002; 21: 462-65.