
Case Report
J Mol Biol & Mol Imaging. 2016; 3(1): 1023.
Fronto-Temporal Dementia: A Case Report
Hammond ENB*, Malan N, Matentji P, Vangu MDTHW
Division of Nuclear Medicine and Molecular Imaging, Department of Radiation Services, University of the Witwatersrand and Charlotte Maxeke Johannesburg Academic Hospital, Johannesburg, South Africa
*Corresponding author: Hammond ENB, Division of Nuclear Medicine and Molecular Imaging, Department of Radiation Services, University of the Witwatersrand and Charlotte Maxeke Johannesburg Academic Hospital, Johannesburg, South Africa
Received: January 04, 2016; Accepted: February 08, 2016; Published: February 10, 2016
Abstract
Fronto-temporal dementia (FTD) is the most common form of primary degenerative dementia after Alzheimer’s disease that affects middle age with an estimated prevalence at 15 per 100,000 in the population aged between 45 – 65 years. FTD is often misdiagnosed due to its early onset, clinical and pathological heterogeneity. Neuroimaging is known to assist in early diagnosis of these disorders. We present a case of a 59 year old woman with FTD.
Keywords: Fronto-temporal dementia; Pick’s disease; Tc-99m HMPAO
Case Presentation
A 59 year old female was diagnosed with major depressive disorder and borderline personality disorder of about a year’s duration. There were continued behavioral changes, with periods of aggression and impulsiveness at times. Also, there was ongoing worsening of memory with poor concentration. She later developed irritable mood and poor appetite.
Examination on her last hospital visit showed a well groomed, alert, calm and cooperative patient. She had normal speech and normal psychomotor activity. Her mood was labile; she was however coherent and relevant showing reactive affects. She was a suicidal however objectively hallucinating.
The patient’s medications included fluoxetine 20mg daily, sodium valproate 200mg twice daily, clonazepam 0.5mg when necessary.
She underwent magnetic resonance imaging (MRI) of the brain in view of her worsening memory and concentration. This showed evidence of mild frontotemporal lobe involution (Figure 1). Incidentally, there was also an enlarged anterior pituitary gland (1.02 x 1.3 cm) seen.
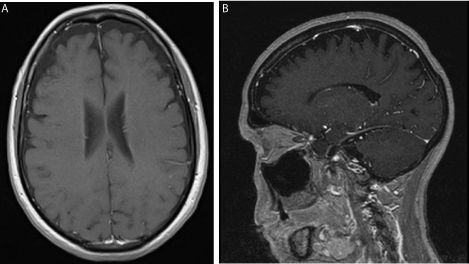
Figure 1: T1 weighted magnetic resonance images (MRI) showing mild
frontotemporal involution; (a) transverse (b) sagittal.
She was also referred to the nuclear medicine department for brain perfusion imaging – this forms part of molecular imaging and was done byacquiring SPECT images of the brain using Tc-99m HMPAO (Exametazime) injected into a pre-inserted IV line in a quiet dimly lit room. The scan showed a relatively decreased perfusion in the frontotemporal region, involving the anterior cingulate and anterior temporal cortices seen in Figure 2. These findings were in agreement with the findings of MRI and together with the clinical history and signs, consistent with frontotemporal dementia with a diagnosis of Pick’s disease most likely.
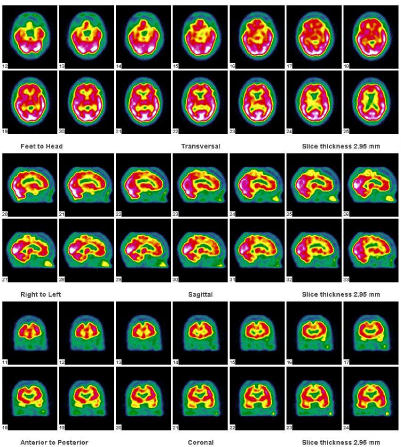
Figure 2: Transverse, sagittal and coronal reconstructed Tc-99m HMPAO
images demonstrating the reduced perfusion in the fronto-temporal areas
involving the anterior cingulate and anterior aspects of the temporal cortices.
Discussion
The concept of frontotemporal dementia includes a group of primary degenerative dementia disorders that presents with predominant frontal lobe and /or temporal lobe symptoms [1,2]. These include Pick’s disease (often referred to in broader term of frontotemporal dementia), frontotemporal lobar degeneration (FTLD) also called dementia lacking distinctive histological features (DLDH), frontotemporal lobar degeneration with motor neuron disease, corticobasal degeneration, progressive supranuclear palsy, frontotemporal dementia with parkinsonism linked to chromosome 17 and others [1].
Frontotemporal dementia (FTD), or Pick’s disease is the most common form of primary degenerative dementia after Alzheimer’s disease that affects middle age [3]. The prevalence is estimated at 15 per 100,000 in the population aged between 45 – 65 years [1,3,4] with an equal incidence in men and women. 30 – 50% of cases are hereditary positive and mean duration of disease estimated to about 7 – 9 years [1].
There is however the possibility that FTD is misdiagnosed due to its early onset, clinical (especially in behavioural form) and pathological heterogeneity [1,4]. Common misdiagnoses are affective disorders, psychosis, alcohol abuse or sometimes Alzheimer’s disease (AD), and in the presence of neurological sign a diagnosis of Parkinson’s disease or motor neuron disease. These misdiagnoses are understandable given the insidious progressive nature of FTD. It is also important to differentiate FTD from Alzheimer’s disease as treatment differs for the two conditions and anticholinesterases used in treatment of AD may show no response or even worsen symptoms in FTD [5].
The clinical diagnosis of FTD is based on operational criteria as described by Mc Khann et al. in 2001 [2]. A combination of clinical observations including status examinations, neuropsychological test, and information gained from laboratory investigations, structural brain imaging (CT or MRI), functional brain imaging (e.g. SPECT or PET), and neurophysiological investigations of the brain (EEG) are necessary to achieve a high accuracy.
The most commonly studied SPECT method uses Tc- 99mhexamethylpropyleneamine oxime (HMPAO) which crosses the blood – brain barrier and is taken up in proportion to blood flow allowing the tracking of cerebral perfusion [6] and thus providing a measure of brain activity. Hypo perfusion in the frontal and temporal regions of the brain as seen in the patient subject of this case presentation is classical for FTDs. This usually agrees with the frontotemporal atrophy expected with structural imaging such as MRI.
Functional brain imaging (perfusion SPECT imaging or metabolic PET imaging with F-18 flourodeoxyglucose (F-18 FDG) has the advantage of having a higher sensitivity for detection of FTDs, especially in early disease [1,4]. This is very important when we consider the fact that there is a high rate of misdiagnosis and delay in diagnosis from the onset of symptoms is on average 3 years with death occurring usually within 6 years of development of symptoms [3,7]. The patient presented here for example was initially being managed for major depressive disorder and border line personality disorder. The neuroimaging findings give us a benefit of hind sight as the clinical features displayed could all result from frontal lobe hypo perfusion and atrophy.
Functional imaging also has the potential utility as a marker of disease severity and to track drug effects in patient care and/or clinical trials especially in the early asymptomatic phase of disease to prevent deterioration [6,8].
Conclusion
FTDs are among the common dementia disorders and often remain misdiagnosed. Functional neuroimaging plays an important role in these conditions as they aid in early diagnosis because of their relatively higher sensitivity. One should therefore consider it as part of diagnostic armamentarium that is often omitted. The re-evaluation of clinical features and results of previous investigations often aid the diagnostic process.
References
- Sjögren M, Andersen C. Frontotemporal dementia--a brief review. Mech Ageing Dev. 2006; 127: 180-187.
- McKhann GM, Albert MS, Grossman M, Miller B, Dickson D, Trojanowski JQ, et al. Clinical and pathological diagnosis of frontotemporal dementia: report of the Work Group on Frontotemporal Dementia and Pick's Disease. Arch Neurol. 2001; 58: 1803-1809.
- Mackin P, Hamish McAllister-Williams R. Frontotemporal dementia: remembering images from the past. Neuropsychiatr Dis Treat. 2005; 1: 281-283.
- Graham A, Hodges JR. Frontotemporal dementia. Psychiatry. 2008; 7: 24-28.
- Mario F Mendez, Jill S Shapira, Aaron McMurtray, Eliot Licht. Preliminary Findings: Behavioral Worsening on Donepezil in Patients With Frontotemporal Dementia. The American Journal of Geriatric Psychiatry. 2007; 15: 84-87.
- Rohrer JD, Rosen HJ. Neuroimaging in frontotemporal dementia. Int Rev Psychiatry. 2013; 25: 221-229.
- Hodges JR, Patterson K, Oxbury S, Funnell E. Semantic dementia. Progressive fluent aphasia with temporal lobe atrophy. Brain. 1992; 115 : 1783-1806.
- Knopman DS, Boeve BS, Caselli RJ, Graff-Radford NR, Kramer JH, Mendez MF, et al. Longitudinal tracking of FTLD: toward developing clinical trial methodology. Alzheimer Dis Assoc Disord. 2007; 21: S58-63.