
Research Article
Austin J Musculoskelet Disord. 2014;1(1): 1005.
Diversity of Femoral Neck and Spine Bone Mineral Density - Surrogate Marker of Aortic Calcification in Postmenopausal Women
Petar JA1*, Maja PA2, Miroslav ZL3 and Aleksandar S4
1Department of Internal medicine, JZU Clinical Hospital “D-r Trifun Panovski”
2University clinic of Obstetrics and Gynecology, “Ss. Cyril and Methodius University” Skopje, Republic of Macedonia
3Department of Internal medicine, JZU City General Hospital, Republic of Macedonia
4University clinic of Nephrology, “Ss. Cyril and Methodius University”, Republic of Macedonia
*Corresponding author: Docent D-r Petar Avramovski, Primarius, department of Internal medicine, JZU “Clinical Hospital D-r Trifun Panovski”, 7000 Bitola, ul. Ivan Milutinovik 37/4-26, Republic of Macedonia
Received: August 03, 2014; Accepted: August 22, 2014; Published: August 23, 2014
Abstract
Background/Aims: Osteoporosis and Abdominal Aortic Calcification (AAC) are major causes of morbidity and mortality in postmenopausal women. The aim of this study was to determine the accuracy of Anterior-Posterior (AP) Dual-energy X-ray Absortiometry (DXA) in detecting and scoring the AAC compared with x-ray Lateral Lumbar Radiography (LLR).
Methods: We estimated femoral neck and lumbar spine Bone Mineral Density (BMD) by AP DXA and AAC by x-ray LLR in 55 postmenopausal female-aged 59.01 ± 9.27 years. We hypothesized that subtracted femoral neck BMD (BMDFN) from lumbar spine BMD (BMDLS) presented as ΔBMD = BMDLS – BMDFN would have predictable diagnostic value in detection of abdominal vascular calcification.
Results: The mean BMDFN was 0.744 ± 0.184 g /cm2 and the mean BMDLS was 0.833 ± 0.157 g /cm2, P < 0.0001; the mean ΔBMD was 0.089 ± 0.077 g / cm2 and the mean AAC score was 2.182 ± 1.982. Bivariate Pearson’s revealed significant positive correlation between AAC and ΔBMD (r = 0.449, p = 0.0006); by linear regression analysis: R2 = 0.2019, coefficients β: b0 = 1.151 (P = 0.003) and b1 = 11.5049 (P = 0.0006) and by multiple regression analysis: βst = 13.5244 (P < 0.0001). We found sensitivity of 64.3% and specificity of 82.9% by receiver operating characteristic (AUC = 0.759) in prediction of AAC by ΔBMD.
Conclusions: This AP subtracting BMD DXA method provides a useful tool for detecting and scoring subclinical and extensive AAC in postmenopausal women, using simple, semiquantitative, accuracy scoring system, with minimal radiation exposure and low cost.
Keywords: Osteoporosis; Aortic calcification; Postmenopausal women; Dual-energy x-ray absortiometry; Lateral lumbar radiography
Abbreviations
AAC: Abdominal Aortic Calcification; AP: Anterior-Posterior; DXA: Dual Energy X-Ray Absortiometry; LLR: Lateral Lumbar Radiograph; BMD: Bone Mineral Density; BMDFN: Femoral Neck Bone Mineral Density; BMDLS: Lumbar Spine Bone Mineral Density; ΔBMD: Delta Bone Mineral Density; Std. error: Standard Error; AUC: Area Under Curve; BMI: Body Mass Index; SD: Standard Deviation; WHO: World Health Organization; CI: Confidence Interval; ROC: Receiver Operating Characteristics; CHP: Chronic Hemodialysis Patients; GPP: General Population Patients
Introduction
Osteoporosis and atherosclerosis are major causes of morbidity and mortality in postmenopausal women [1]. Calcification is a common feature of atherosclerotic plaques and is regulated in a way similar to bone mineralization [2]. There are not enough studies that examined whether presence of atherosclerotic calcification is associated with bone loss.
The term osteoporosis is used to define a group of clinical disorders characterized by reduced bone mass without defect in mineralization. Osteoporosis occurs when bones lose an excessive amount of their protein and mineral content (calcium). Bone is living tissue that is constantly being renewed in two-stage process (resorption and formation) that occurs throughout life. After mid- 30s, bone mass is lost at a faster pace than it is formed, so the bone mineral density in the skeleton begins to slowly decline. Most cases of osteoporosis occur as an acceleration of this normal aging process, which is referred to as primary osteoporosis [3].
Bone mineral loss is most often in older people and in women after menopause. They lose bone mineral mass more rapidly after menopause (usually around age 50), when they stop producing a bone-protecting hormone called estrogen. Seven years following menopause, women can lose more than of 20% of their bone mineral mass. Women are about five times more likely to be affected than men to develop osteoporosis [4].
Vascular calcification and osteoporosis are common age-related processes. AAC is displayed on routine lateral lumbar spine radiographs as dense calcium mineral deposits of the aorta that lies adjacent to vertebrae. The two processes may represent independent age-related phenomena, or mobilization of calcium in developing atherosclerotic plaque. It means that, vascular compromise due to aortic calcification might, in itself, results in bone loss [5]. Atherosclerosis calcification has long been considered a late stage, unregulated sequel of atherosclerotic process. Aortic calcification occurs more early with rapid progress and arterial narrowing. Recent studies implicated several possible metabolic linkages between aortic calcification and bone mineral density loss, including estrogen, vitamin D and K, lipid oxidation products and osteoprotegerin (protein that regulates osteoclast activity and proliferation).
Our hypothesis was that the value of subtracted femoral neck BMD from lumbar spine BMD (ΔBMD) should be greatest in those individuals with more vascular calcification of the abdominal aorta. The aims of this study were:
- To find association between AAC and femoral neck BMD; between AAC and spine BMD; AAC and ΔBMD;
- To determine the accuracy of AP DXA scan in detecting and scoring AAC compared with detected AAC by LLR.
Materials and Methods
Patients
This cross-sectional study was conducted from October to December 2013. The study group included volunteer sample of 55 white postmenopausal women with mean age of 59.01 ± 9.27 years, their mean Body Mass Index (BMI) of 27.7 ± 3.65 kg/m2. Fourteen women were smokers, 12 were diabetic, and 30 were hypertensive. Exclusion criteria were chronic renal disease, insulin-dependent diabetes, malignancy, rheumatoid arthritis, liver disease, or any chronic disease that might affect the skeleton. They signed an informed consent and the Ethics Committees of our institution approved the study. Menopausal state was assessed by a self-administered questionnaire that asked whether the menses had stopped. Women were classified as postmenopausal once they had experience at least 12 consecutive months of amenorrhea.
Demographic and clinical data were collected from the patient’s chart and included age, weight, height, history of diabetes mellitus, smoking habit, hypertension, and above mentioned disease that might affect the bone mass. BMD of the femoral neck and the lumbar spine was assessed by Dual Energy X-Ray Absorptiometry (DXA). Lateral Lumbar Radiography (LLR) of the abdominal aorta was used to determine the overall Abdominal Aortic Calcification (AAC) score.
Assesment
Bone mineral density
Bone density scanning, also called Dual-Energy X-Ray Absorptiometry (DXA) or bone densitometry, is an enhanced form of X-ray technology that is used to measure bone density. DXA is today’s established standard for measuring BMD [6].
We conducted BMD testing using DXA by Hologic QDR4500SL system (Hologic Inc., Bedford, MA, USA). BMD was measured by DXA in the lumbar spine and femoral neck. Two X-ray beams with differing energy were used for measurement of BMD. The BMD was determined based on the absorption of each beam by bone after subtraction of the absorption of soft tissue. For assessment of the spine, the patient’s legs were supported on a padded box to flatten the pelvis and lower the (lumbar) spine. For assessment of the femoral neck, the patient’s foot was placed in a brace that rotates the hip inward. In both cases, the detector was passed slowly over the area, generating images on a computer monitor [7].
Absolute BMD values and T-scores (number of SDs below the BMD of a young reference group) of the lumbar spine and femoral neck were recorded as BMD (g/cm2) and T-score (for femoral neck, total and L1 to L4 region). The WHO (World health organization) defined the following categories based on bone density in Caucasian females: normal bone, T-score greater than -1; osteopenia, T-score between -1 and -2.5; osteoporosis, T-score less than -2.5.
Abdominal aortic calcification
We performed lateral lumbar radiographs to determine AAC in the standing position using standard radiographic equipment (Shimadzu RADSpeed 324-DK, Nishinokyo-Kuwabarachou. Nakagyo-ku. Kyoto 604-8511. Japan. The film distance was 1 m and estimated dose of radiation was no more than 15 mGy. Abdominal aortic calcification is often seen as linear thin-film tracks at the anterior or posterior wall of the abdominal aorta with linear edge corresponding to the aortic wall beside lumbar vertebral segments L1 to L4.
We estimated aortic score using a previously validated system [6- 8]. The measure for the unit AAC score is the linear length of aortic calcification compared with 1/3 of aortic longitudinal wall projected near the vertebral segment beside it: score 0 – no calcific deposits in front of the vertebra; score 1 – small scattered calcific deposits filling less than 1/3 of the longitudinal wall of the aorta; score 2 – 1/3 or more, but less than 2/3 of the longitudinal wall of the aorta calcified; score 3 – 2/3 or more of the wall calcified. The scores were summarized using the composite score for anterior and posterior wall severity (range score 0–3), where the scores of individual aortic segment calcifications, both for the anterior and posterior walls (max. 2 x 12) were summed (maximum score 24) [8,9].
Statistical analysis
The data were analyzed using MedCalc version 13.0.6.0 (Acacialaan 22, 8400 Ostend, Belgium). Results were expressed as mean ± SD or percentage. Student’s t test for paired data was used to compare the femoral neck BMD and lumbar spine BMD. Pearson’s correlations were calculated to explore the relationship between femoral neck BMD, spine BMD and ΔBMD and other variables, as appropriate. Simple linear regression analysis was performed to assess the associations between dependent and independent variables and to create the equation of linear regression. We conducted a multiple backward regression analysis to determine the effect on the dependent variable (AAC) of variations in one of the independent variables (femoral neck BMD, diabetes, hypertension, spine BMD, smoking, age and BMI) while the other independent variables were fixed. All tests were two-sided. A value of p < 0.005 was considered to indicate a significant difference.
Results
During the 3-month period from October to December 2013, DXA and Lateral lumbar X-ray radiography measurements and other demographic examinations were successfully conducted on 55 postmenopausal female participants aged 59.01 ± 9.27 years and body mass index 27.7 ± 3.65 kg/m2. The demographic and clinical characteristics of the patients studied are presented in Table 1.
Characteristic
mean ± SD, n(%)
95%CI
Range
Age, Yr
59.01 ± 9.27
54.0-62.0
46-79
Height, cm
161.8 ± 7.37
160.0-164.0
150-182
Weight, Kg
72.61 ± 10.55
69.76-74.0
50-101
BMI, Kg/m2
27.7 ± 3.65
27.135-28.130
22.2-35.3
Hypertension
30(54.5)
/
/
Diabetes
12(21.8)
/
/
Smokers
14(25.4)
/
/
BMD F. neck, g/cm2
0.744 ± 0.184
0.702-0.811
0.223-1.056
BMD Spine, g/cm2
0.833 ± 0.157
0.775-0.858
0.443-1.101
ΔBMD,g/cm2
0.089 ± 0.077
0.044-0.100
0.004-0.306
Aortic calcification
2.182 ± 1.982
1.0-3.0
0.00-8.00
Table 1: Demographic and clinical characteristics of the patients studied.
The mean BMD of the femoral neck was 0.744 ± 0.184 g /cm2 and the mean BMD of the lumbar spine was slightly greater 0.833 ± 0.157 g /cm2. The results from Paired t test between femoral neck and lumbar spine BMD were: mean difference (-0.0896), test statistic t (-8.583), degrees of freedom (DF, 54) and two-tailed probability (P < 0.0001). The mean difference of lumbar spine and femoral neck BMD, presented as ΔBMD, was 0.089 ± 0.077 g /cm2. The mean aortic calcification was 2.182 ± 1.982.
Fourteen (25.4%) patients were smokers, 12 (21.8%) were diabetic, and 30 (54.5%) were hypertensive, their mean BMI was 27.7 ± 3.65 kg/m2.
In the same table, besides the column of mean ± SD, the columns of 95% confidence interval and range are also presented.
The notched box-and-whisker bars for BMD’s tissue biomarkers are presented in Figure 1. Their mean, 95% CI of the mean, range, median, 25th and 75th percentiles present lumbar spine BMD, femoral neck BMD and lumbar spine minus femoral neck BMD (Δ).
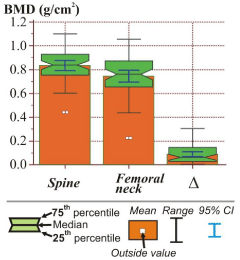
Figure 1: Box plots of the mean, range, median, 25th and 75th percentiles for tissue biomarkers.
The results of the bivariate Pearson’s correlation analysis of demographic characteristic with BMD and aortic calcification are presented as (r) indexes and (p) values. The positive value of Pearson product-moment correlation coefficient (r), as measure of the strength of linear dependence between two variables (one in the measured tissue markers in top horizontal row and one in the demographic and tissue markers in vertical column) indicated significant positive correlation between: aortic calcification and hypertension (r = 0.268, p = 0.047), aortic calcification and smoking (r = 0.352, p = 0.008) and aortic calcification and ΔBMD (r = 0.449, p = 0.0006); BMD and BMI (r = 0.278, p = 0.041) and BMI and femoral neck BMD (r = 0.291, p = 0.031). Pearson’s revealed significant inverse correlation between: age and both femoral neck and lumbar spine BMD (r = -0.325, p = 0.015 and r = -0.356, p = 0.007 respectively), femoral neck BMD and smoking (r = -0.286, p = 0.034) and lumbar spine BMD and smoking (r = -0.323, p = 0.016).
The results of linear regression which are an approach for modeling the relationship between a scalar dependent variable Y (aortic calcification) and a explanatory variable denoted X (ΔBMD, g/ cm2) are presented in table 3.
Coefficient of determination R2 (0.2019) is showing that 20.19% from the total variability is explained with the linear relation between aortic calcification and ΔBMD or that 20.19% from aortic calcification is dependent of the ΔBMD. Only 20.19% from the changes in aortic calcification are result of the ΔBMD value changes and the rest 79.81% from the total variability between them are not explained (79.81% of aortic calcifications are dependent of other factors, which are not covered with the regression model). This model was used as criterion for best regression equation choice, so the greater its value is, the better the model of approximation will be.
The regression parameter bo = 1.151 is showing the expected theoretical value of aortic calcification in case if ΔBMD would have value equal to zero. This parameter also shows the point of the y-axis (dependent variable axis, aortic calcification) through which the regression line passes across. The regression parameter b1 = 11.5049 signifies that with at each increasing of one unit (g/cm2) in ΔBMD, aortic calcification score increases for 11.5049. The equation of simple linear regression y = 1.1510 + 11.5049 · X shows the average coordination of aortic calcification and ΔBMD variations. With this equation, we get the evaluated (theoretical) aortic calcification values in opposition to its empirical values.
A figure 2 shows a scatter plot of aortic calcification and ΔBMD. There is a positive association between these variables. The data from each one of 55 patients is displayed as a collection of colored point (red square, blue circle and white circle) determining the bone strength presented by T-score. Each point has the value of one variable determining the position on the horizontal axis and the value of the other variable determining the position on the vertical axis. Linear regression lines computed by data acquired from different BMD patient’s status (normal, osteopenia and osteoporosis) are plotted and shown by different color and line style (orange solid line, brown dashed line and blue dash-dot line). Linear regression line plotted with double-colored line (red-purple) shows a positive correlation between aortic calcification and ΔBMD in all examined female group independent of their bone strength status (BMD).
Assessments (standardized coefficient β [βst], standard error of βst, t and p value) of independent predictor (ΔBMD) or determinants (femoral neck BMD, diabetes and hypertension) for increasing of abdominal aortic calcification in postmenopausal women after backward multiple regression analysis are shown in table 4. P values followed the order of statistical significance: ΔBMD (< 0.0001), diabetes (0.0091) and femoral neck BMD (0.0241). There are no statistical significance of βst coefficients expressed by P-value for hypertension (0.0560) and spine BMD, smoking, BMI and age with P > 0.1. Coefficient of determination R2 (0.4758) is showing that 47.58% from the total variability is explained with the linear relation between aortic calcification and ΔBMD accompanied by other determinants, or that 47.58% from aortic calcification is dependent of the ΔBMD as predictor and other determinants (femoral neck BMD, diabetes and hypertension). There is an inverse correlation (negative βst coefficient, βst = -3.1871) between the femoral neck BMD and the AAC, only. That means any reduction of the femoral neck BMD results with increased abdominal aortic calcification.
We used discrimination, the ability of a model (estimation of cutoff point) to distinguish patients with or without calcification. We assessed them by Receiver Operating Characteristic (ROC) curve analysis. Receiver operating characteristics curves for ΔBMD as a prognostic diagnostic marker associated with anterior-posterior DXA predicting the presence of AAC as detected by LLR, sensitivity, specificity, area under curve (AUC), 95% CI for sensitivity and specificity, Z statistic, criterion value of ΔBMD variable and P-value are shown in figure 3.
Each point on the ROC curve represents a sensitivity/specificity pair corresponding to a particular threshold (ΔBMD in detection of AC). The results we got by ROC curve analysis were: area under curve (0.759), z statistic (3.524), significance level (p = 0.0004), sensitivity (64.3%) and specificity (82.9%). The ΔBMD cutoff point where the pars of sensitivity/specificity points were highest was 0.094 g/cm2.
Discussion
To our knowledge, this is the first cross-sectional study that investigates the relationship between ΔBMD and AAC in postmenopausal women. Several studies detect AAC by Computed Tomography (CT). We know that CT is currently gold standard of AAC measuring, but it is limited by high radiation dose exposure. The study of Marina C. 2013 determines the accuracy of lateral-DXA scan in detecting AAC compared to CT in healthy women [9]. In our study, we determined the accuracy of anterior-posterior DXA in detecting AAC compared with LLR (at a subtracted BMDH from BMDLS).
The lumbar spine BMD (0.833 ± 0.157 g/cm2) was greater than the femoral neck BMD (0.744 ± 0.184 g/cm2). This difference was statistically significant (p < 0.0001). Is it possible to get such a big difference in bone density at two different anatomical sites during an identical process of bone loss in the same individual? The reason for the greater BMD in the spine than the femoral neck may lie in the fact that DXA relies on measurement of the relative absorption of dual energy X-ray beams projected blindly through the body. Densely calcified aorta rather than the spine absorb the X-ray, causing a falsely elevated BMD reading [11,12]. The patients with higher score of aortic calcification results with more x-ray absorption, expressed with elevated spine BMD value. Vertebral BMD is usually measured in the AP plane, though this method may give falsely high values in the presence of lumbar spondylosis or osteoarthritis, especially when associated with osteophytes and aortic calcification in the same time.
Sclerosis and joint narrowing had little effect on BMD at lumbar spine or hip. Indirect effects of osteoarthritis on BMD were small and inconsistent across genders. Multiple regression analysis, including weight, age, and vertebral calcification scores, demonstrated a small but significant effect of osteophyte score on lumbar BMD (partial r2 = 0.04; P = 0.012) [13].
An advantage of our study is the fact that association between aortic calcification and bone mineral density was estimated in postmenopausal women, the period from which the prevalence of atherosclerosis and osteoporosis increases. Bone loss during menopause may results from a common etiologic factor, such as estrogen deficiency. Arteries and bones are target organs for estrogen. Estrogen receptors have been demonstrated on vascular endothelial and smooth cells, osteoblasts and osteoclasts, suggesting a direct effect of estrogen on vascular and bone cells [14]. Estrogen deficiency may have indirect effects on arteries and bone by the production of inflammatory agents, such as interleukin-1 and -6 and tumor necrosis factor, which are involved in atherogenesis and contribute to accelerated bone resorption [15]. There are a whole lot of circulating biomarkers that contribute in accelerated bone resorption and atherosclerosis: calcium-regulating hormones, vitamin D deficiency, serum calcium, calcium-phosphorus product and plasma homocysteine.
The aim of our study was not investigation of their effect on bone resorption and atherosclerosis, but only to find an association between them. We found (by bivariate Pearson correlation) significant positive correlation between aortic calcification and ΔBMD (p = 0.0006), aortic calcification and hypertension (p = 0.47), aortic calcification and smoking status (0.008), but negative correlation between femoral neck BMD and age (0.015), femoral neck BMD and BMI (0.031) (Table 2). We found a positive correlation between aortic calcification as a dependent variable and ΔBMD as an independent variable (by linear regression analysis, p = 0.0006, Table 3). The predictable power of subtracted BMDH from BMDLS for aortic calcification detection we expressed by linear regression equation and its β coefficients. Each increase of one ΔBMD unit, results with elevated percent of detected aortic calcification by LLR, or aortic calcification score increases for 11.5049 for each one single increase of ΔBMD. The predictable power of different stage of bone strength we presented by three linear regression line for normal bone, osteopenia and osteoporosis, and fourth, for common predictable line for all postmenopausal women, independent of their bone mineralization stage (Figure 2). Osteoporosis line has the greatest angle of ascent and presents the ascendant power in predicting of aortic calcification, because the greatest subtracted value of bone matrix in different anatomical site, lumbar spine and femoral neck. It has superior power in predicting AAC than osteopenia or normal line.
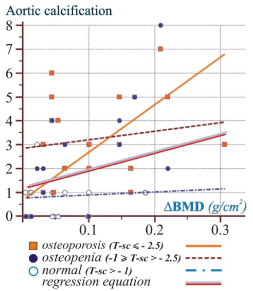
Figure 2: Scatter plot of ΔBMD and aortic calcification.
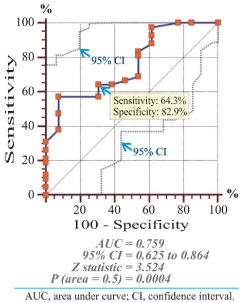
Figure 3: Receiver Operating Characteristics (ROC) curves for ΔBMD as a prognostic diagnostic marker for AAC and Area Under Curve (AUC).
Characteristic
Aortic Calcification
BMD F.neck, g/cm2
BMD spine, g/cm2
ΔBMD, g/cm2
r
p
r
p
r
p
r
p
Age, Yr
0.118
0.391
-0.325
0.015
-0.356
0.007
0.197
0.149
BMI, Kg/m2
0.135
0.324
0.291
0.031
0.204
0.135
0.278
0.041
Hypertension
0.268
0.047
-0.062
0.654
-0.039
0.775
0.032
0.817
Diabetes
0.116
0.398
0.235
0.084
0.231
0.091
0.081
0.556
Smokers
0.352
0.008
-0.286
0.034
-0.323
0.016
0.187
0.171
BMD F. neck, g/cm2
-0.241
0.076
/
/
0.214
0.116
0.131
0.324
BMD Spine, g/cm2
-0.178
0.193
0.214
0.116
/
/
0.235
0.084
ΔBMD,g/cm2
0.449
0.0006
0.131
0.324
0.235
0.084
/
/
Aortic calcification
/
/
-0.241
0.076
-0.178
0.193
0.449
0.0006
Table 2: Bivariate Pearson’s correlation analysis of demographic characteristic with BMD and aortic calcification.
Regression
Dependent Y
Aortic Calcification
Independent X
ΔBMD, g/cm2
��
Sample size
55
Coefficient of determination R2
0.2019
Residual standard deviation
1.7875
��
Regression Equation
Y=1.1510+11.5049.X
parameter
Coefficient �
Std. Error
95%CI
t
P
Intercept b0
1.151
0.3706
0.4077 to 1.8943
3.1058
0.003
Slope b1
11.5049
3.1418
5.2033 to 17.8065
3.6619
0.0006
Table 3: Linear regression analysis of aortic calcification and ΔBMD.
In multiple regression analysis, we found an independent predictor (ΔBMD, p < 0.0001) for aortic calcifications (Table 4). Routine LLR for detection of aortic calcification of all women is not feasible for most populations; hence, identification of high-risk subset women by DXA will be an important element of effective preventive strategies for bone resorption and atherosclerosis. By multiple regression analysis, we find the diabetes as a determinant for increasing of abdominal aortic calcification; and femoral neck BMD as a determinant with inverse correlation with aortic calcification. The abnormal metabolic state accompanying diabetes results in changes in the state of arterial structure and function. Most patients with diabetes, including those with vascular disease, demonstrate abnormalities of endothelial function and vascular regulation. Local increases in these proinflammatory factors, together with the loss of normal nitric oxide function are associated with increased leukocyte chemotaxis, adhesion, transmigration, and transformation into foam cells. This latter process is further augmented by increased local oxidative stress. Foam cell transformation is the earliest precursor of atheroma formation and calcification [16,17]. There is strong correlation between ΔBMD and AAC: about 47.58% from total variability is explained with the linear positive correlation between above-mentioned covariates.
Multiple Regression
Dependent Y
Aortic Calcification
Method
Backward
Enter Variable if:
P<0.05
Remove variable if: p>0.1
Sample Size
55
Coefficient of determination R2
0.4758
Residual standard deviation
1.5067
Regression Equation
Independent variables
Coefficient �st
Std.Error
t
P
BMD, g/cm2
13.5244
2.7833
4.859
<0.0001
BMD F.Neck, g/cm2
-3.1871
1.369
-2.328
0.0241
Diabetes
1.7008
0.6266
2.715
0.0091
Hypertension
0.8546
0.4366
1.957
0.056
Table 4: Multiple backward regression analysis of determinants of aortic calcification.
AP DXA imaging may therefore provide an important low-radiation tool for detecting patients at increased risk of large artery stiffening, isolated systolic hypertension, and cardiovascular events. Cardiovascular disease remains the leading cause of death in women, with approximately 30 % of cardiovascular events unexplained by conventional risk factors [18]. Our previous comparative DXA study in Chronic Hemodialysis Patients (CHP) and General Population Patients (GPP), confirms the spine and the femoral neck BMD difference dependent of AAC. The difference (0.049 g/cm2) in bone density between the spine (0.924 g/cm2) and femoral neck (0.875 g/ cm2) was less evident in the GPP group than in the CHP group (0.886 g/cm2 - 0.759 g/cm2 = 0.127 g/cm2); likely due to the smaller degree of aortic calcification in the GPP resulting in low absorption of the X-ray beam, leading to a decreased BMD value [19]. During last six months, we used a figure 2 as nomogram, (statistical predictive model that can provide the aortic calcification score [y-axis] based of the subtracted BMDH from BMDLS value) which we plot from DXA results. Example: in postmenopausal osteoporotic woman with ΔBMD of 0.2 g/cm2 after reflexion on line for osteoporosis we got 4.5 AAC score units on y-axis. After LLR x-ray radiography in this woman, we found AAC score five, with minimal error of 11.1%. In this way, we discover patients who have shown an increased risk for AAC and we send for further verification of aortic calcification by x-ray LLR or CT.
AP DXA scans therefore provide a low-radiation method (only 0.001 mSv for DXA compared to 8 mSv for abdominal CT and 0.3 mSv for LLR) [20] with high sensitivity (64.3%) and specificity (82.9%) to detect initial or extensive aortic calcification in postmenopausal women. This subtracting BMD DXA method provides a useful tool for detecting subclinical AAC compared to LLR using simple, semiquantitative accuracy scoring system, with minimal radiation exposure dose and low cost.
Limitations
The first limitation of this study was the small number of patients sampled. Recruiting male and female patients in sufficient numbers ultimately proved to be impossible. Due to the limitation of current imaging techniques, we were unable to distinguish between intimal and medial aortic calcification. Future prospective studies will be required to define the clinical implications of aortic calcification as detected by AP DXA. The main limitations of this study include the need for validation of the results in broader trial general populations.
Lumbar spine radiographs is x-ray method used to identify osteophyte formation, facet joint osteoarthritis, vertebral fracture, sclerosis, joint space narrowing and aortic calcification. The last limitation in our study because we did not evaluate the results of lumbar spine osteoarthritis on the available LLR in order to check its effects on the spine BMD results. Stepwise multiple regression analysis indicated that osteoarthritis (formation of osteophytes and joint space narrowing) explained 16.6% of variation in lumbar spine BMD in elderly women. Lumbar spine ostoephytes affect most subjects over the age of 60 years (mean age in our participants was 59.01 ± 9.27 years) and contribute substantially to lumbar spine BMD measured in the AP DXA [21]. In our study, by multiple regressions analysis we proved that 47.58% from the total variability is explained with the linear relation between aortic calcification and ΔBMD.
Conclusion
This AP subtracting BMD DXA method provides a useful proven tool for detecting and scoring subclinical and extensive AAC in postmenopausal women, using simple, semiquantitative, accuracy scoring system, with minimal radiation exposure and low cost.
References
- Hak AE, Pols HA, van Hemert AM, Hofman A, Witteman JC. Progression of aortic calcification is associated with metacarpal bone loss during menopause: a population-based longitudinal study. Arterioscler Thromb Vasc Biol. 2000; 20: 1926-1931.
- Demer LL. Vascular calcification and osteoporosis: inflammatory responses to oxidized lipids. Int J Epidemiol. 2002; 31: 737-741.
- Donna O, Christine J, Karen B. Gale Encyclopedia of Medicine 5th edn. Emerald Group Publishing Limited. 2008.
- Elliott, William T. HRT, estrogen, and postmenopausal Women: Year-old WHI Study Continues to Raise Questions. Critical Care Alert. 2003; 7: 1.
- Kiel DP, Kauppila LI, Cupples LA, Hannan MT, O'Donnell CJ, Wilson PW. Bone loss and the progression of abdominal aortic calcification over a 25 year period: the Framingham Heart Study. Calcif Tissue Int. 2001; 68: 271-276.
- Honkanen E, Kauppila L, Wikström B, Rensma PL, Krzesinski JM, Aasarod K, et al. Abdominal aortic calcification in dialysis patients: results of the CORD study. Nephrol Dial Transplant. 2008; 23: 4009-4015.
- Avramovski P, Janakievska P, Koneska M, Sotiroski K, Sikole A. Associations between Pulse Wave Velocity, Vascular Calcifiaction, and Bone Mineral Density in Chronic Hemodialysis Patients and General Population. ISRN Vasc Medicine 2013; 10: 1-9.
- El Maghraoui A, Roux C. DXA scanning in clinical practice. QJM. 2008; 101: 605-617.
- Toussaint ND, Lau KK, Strauss BJ, Polkinghorne KR, Kerr PG. Determination and validation of aortic calcification measurement from lateral bone densitometry in dialysis patients. Clin J Am Soc Nephrol. 2009; 4: 119-127.
- Martina C, Michele LF, Tim DS, Phil C. Abdominal Aortic Calcification Detection Using Dual-Energy X-Ray Absortiometry: Validation Study in Healthy Women Compared to Computed Tomography. Calcif Tissue Int. 2013; 2: 7-13.
- Toussaint ND, Lau KK, Strauss BJ, Polkinghorne KR, Kerr PG. Associations between vascular calcification, arterial stiffness and bone mineral density in chronic kidney disease. Nephrol Dial Transplant. 2008; 23: 586-593.
- Adler RA. Osteoporosis: Pathophysiology and Clinical Management. 2nd edn. Totowa: Humana Press; 2010.
- Reid IR, Evans MC, Ames R, Wattie DJ. The influence of osteophytes and aortic calcification on spinal mineral density in postmenopausal women. J Clin Endocrinol Metab. 1991; 72: 1372-1374.
- Mendelsohn ME, Karas RH. The protective effects of estrogen on the cardiovascular system. N Engl J Med. 1999; 340: 1801-1811.
- Ross R. The pathogenesis of atherosclerosis: a perspective for the 1990s. Nature. 1993; 362: 801-809.
- Wildner M, Peters A, Raghuvanshi VS, Hohnloser J, Siebert U. Superiority of age and weight as variables in predicting osteoporosis in postmenopausal white women. Osteoporos Int. 2003; 14: 950-956.
- Tsao PS, Wang B, Buitrago R, Shyy JY, Cooke JP. Nitric oxide regulates monocyte chemotactic protein-1. Circulation. 1997; 96: 934-940.
- Kauppila LI, Polak JF, Cupples LA, Hannan MT, Kiel DP, Wilson PW. New indices to classify location, severity and progression of calcific lesions in the abdominal aorta: a 25-year follow-up study. Atherosclerosis. 1997; 132: 245-250.
- Avramovski P, Sikole A. The progression of bone mineral density loss in dialysis patients compared with the general population. Korean J Intern Med. 2012; 27: 436-442.
- Wall BF, Hart D. Revised radiation doses for typical X-ray examinations. Report on a recent review of doses to patients from medical X-ray examinations in the UK by NRPB. National Radiological Protection Board. Br J Radiol. 1997; 70: 437-439.
- Liu G, Peacock M, Eilam O, Dorulla G, Braunstein E, Johnston CC. Effect of osteoarthritis in the lumbar spine and hip on bone mineral density and diagnosis of osteoporosis in elderly men and women. Osteoporos Int. 1997; 7: 564-569.