Review Article
Austin Neurosurg Open Access. 2014;1(2): 1008.
Anatomic Aspects of the Carpal Tunnel Release Surgery
Bartosz Godlewski*, Ryszard Czepko, Tomasz Gierula and Grzegorz Klauz
Department of Neurosurgery, Hospital of Saint Raphael, Poland
*Corresponding author: Bartosz Godlewski, Department of Neurosurgery, Scanmed - St. Raphael Hospital, 12 Bochenka Street, 30-693 Cracow, Poland
Received: March 05, 2014; Accepted: May 07, 2014; Published: May 08, 2014
Abstract
Of all upper limb nerve entrapment syndromes, the carpal tunnel syndrome is most often treated surgically. Despite apparent technical simplicity of the procedure, failure to identify the precise anatomical relations within the operative site may result in complications and/or lack of post–operative improvement. The article presents anatomic aspects of the carpal tunnel syndrome that are important for the operating surgeon.
Keywords : Carpal tunnel syndrome; Carpal tunnel anatomy; Median nerve
Introduction
Of all upper limb nerve entrapment syndromes, the carpal tunnel syndrome (CTS) is most frequently treated surgically. In carpal tunnel syndrome, the median nerve is compressed within the carpal tunnel distally – above the distal carpal line. CTS chiefly affect individuals between 40 and 60 years of age, with a female–to–male ratio of 4:1. It is bilateral in more than 50% of patients and tends to be more severe in the dominant limb. The most characteristic symptoms are tingling and numbness in the hand and fingers in the median nerve distribution. The symptoms usually occur during the night and often wake the patient up. The pain becomes more intense on raising the limb or flexing the wrist. Lowering the limb brings relief. Over time, precise grip becomes difficult and there may be atrophy of thenar muscles.
The treatment of this syndrome is initially conservative with nonsteroidal anti–inflammatory drugs, rest, neutral position splints and steroid injection into the tunnel. Surgical treatment is indicated in patients who did not improve with conservative treatment, patients with thenar atrophy or electrophysiological evidence of denervation [1–4].
Median Nerve Topography in the Carpal Area
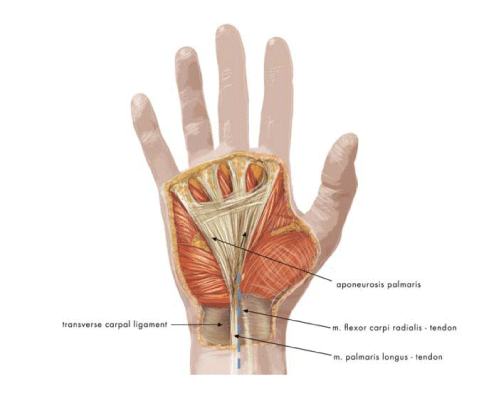
In the region of the carpal line, the median nerve projects medially to the tendon of the radial flexor muscle of the wrist (flexor carpi radialis, FCL) and slightly laterally and inferiorly to the tendon of the long palmar muscle (palmaris longus). In some individuals, the palmaris longus tendon and the FCL sheath, which is situated more deeply (under the transverse carpal ligament), are palpable and sometimes visible under the skin. The palmaris longus tendon lies over the transverse carpal ligament, and the FCL tendon lies under it. The median nerve can be expected to be found between these two structures, the nerve projecting as an “extension” of the 3rd finger. The median nerve courses under the transverse carpal ligament and enters the carpal tunnel. The palmaris longus tendon becomes the palmar aponeurosis. The transverse carpal ligament is continuous with the antebrachial fascia proximally and with the flexor retinaculum of the hand distally (Figure 1). The junction between the transverse carpal ligament and the flexor retinaculum is approximately 3 cm above the distal carpal line. Approximately 10–20% of the population lack a palmaris longus tendon. In these individuals, the palmar aponeurosis fuses only with the flexor retinaculum [1,5–7].
Figure 1: Anatomic structures of the palmar surface of the hand that is important during carpal tunnel release surgery. The course of the median nerve is marked with a dashed blue line.
Sensory and Motor Branches of the Median Nerve
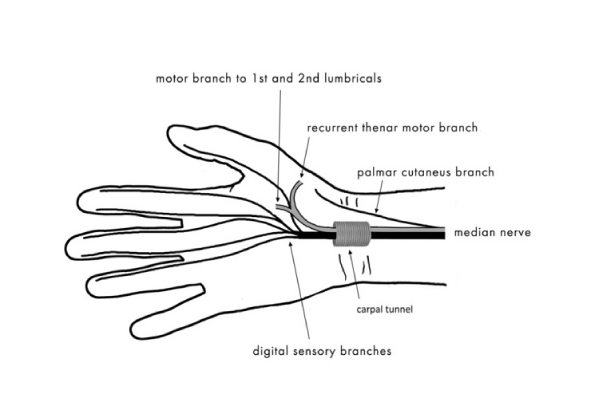
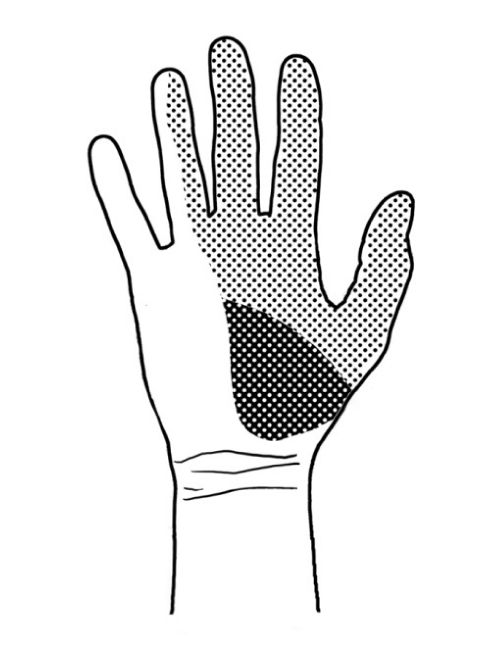
Distal to the carpal tunnel, the median nerve divides into a motor branch and sensory branches of the fingers. The motor branch supplies the first and second lumbrical muscles (of the index and middle fingers, respectively) and three muscles of the thenar prominence: the opponens pollicis, abductor pollicis brevis and flexor pollicis brevis. The sensory branches of the fingers supply the index and middle fingers and, partly, the thumb and the radial aspect of the fourth finger. There is a separate sensory ramus arising proximally to the carpal tunnel. This is the palmar cutaneous branch, which arises from the median nerve on the radial (lateral) side under the tenosynovia of the superficial finger flexors, at the level of the middle (third) finger. It originates approximately 5 cm proximal to the styloid process of the radial bone, crosses the wrist over the transverse carpal ligament and provides sensory innervation to the base of the thenar eminence (Figure 2). Injury to the sensory branch of the median nerve may cause pain associated with the formation of a traumatic neurinoma and sensory deficit in the area it supplies (Figure 3) [5,7–9].
Figure 2: The motor and sensory branches of the median nerve.
Figure 3: The area supplied by the sensory branch of the median nerve in the palmar surface of the hand. The sensory branch (palmar cutaneous branch) is marked with a darker colour.
Carpal Tunnel
The carpal tunnel is bounded on the ulnar side by the ulnar carpal prominence, formed by the pisiform bone and the hook of thehamate bone, and on the radial side by the radial carpal prominence, formed by the tubercles of the navicular bone of the hand and of the trapezium bone. Dorsally, it is bounded by the carpal sulcus, formed by both rows of the carpal bones, while the flexor retinaculum bounds it on the palmar side. The carpal tunnel contains the median nerve, the tenosynovium of the flexor pollicis longus and the tenosynovia of eight finger flexors (superficial and deep). Lying between these bony confines is also the sheath of the flexor radialis carpi tendon, which, anatomically, is not regarded as contained within the carpal tunnel (Figure 4) [6,9–11].
Figure 4: The anatomic structures of the carpal tunnel.
Anatomy and Surgery
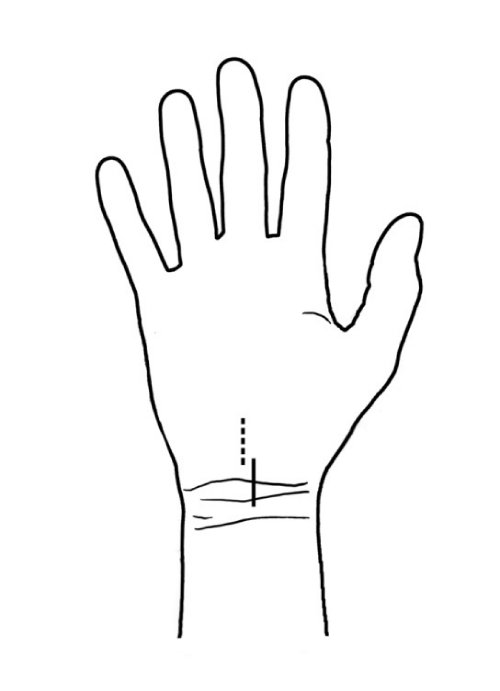
Nowadays, most surgeons prefer a longitudinal skin incision above the course of the median nerve in the carpal region. The incision may be performed directly above the distal wrist line and extended somewhat towards the palmar surface of the hand. With this incision, the nerve can be easily located, which is the first stage of the procedure. Next, the median nerve is followed so that the flexor retinaculum can be reached and then severed, thus decompressing the nerve. An advantage of this surgical approach is that is affords relatively easy and direct access to the median nerve. In order to enable better intraoperative exposure, surgeons are advised to place a small “sandbag” under the wrist for the hand to rest on during the procedure. This “sandbag” allows for slight “hyperextension” of the wrist that affords better visual exposure of the surgical site. With this technique, it is not necessary to extend the incision into the palmar surface of the hand since there is good palmar exposure and the retinaculum can be cut to release the compressed median nerve. When the skin incision is made in the region of the carpal line, damage to the palmar cutaneous branch can be prevented by choosinga more “ulnar” site for the incision relative to the anatomic course of the nerve. This reduces the risk of palmar cutaneous nerve injury and the resulting pain associated with the formation of a traumatic neurinoma [1,7,8,12]. A transverse or Z–shaped incision was mainly performed historically to prevent tensioning of the skin around the surgical wound during movements of the wrist and improve wound healing conditions. A similar effect can be achieved with a longitudinal incision if it is slightly curved (S–shaped) [11,13,14].
Another common surgical approach involves an incision distal to the carpal line on the palmar surface of the hand. This approach helps avoid problems with surgical wound healing associated with movements of the wrist, but special care needs to be taken when exposing the median nerve since this involves cutting the flexor retinaculum, which overlies the nerve, adhering to and compressing it. After the median nerve has been identified, it is released distally towards the fingers and proximally towards the wrist.
Recently there has been a trend to treat CTS by the endoscopic release of the transverse carpal ligament [15,16]. Endoscopic carpal tunnel release is claimed to be associated with minimal pain and scarring due to small incision, a shortened recovery period and a high level of patient satisfaction [17]. The disadvantages of this technique are the higher cost of surgery and the highest learning curve of the surgeon, compared to the open technique [4,17,18]. Current literature suggests that the long–term results of endoscopic carpal tunnel release are the same as those of open carpal tunnel release [19].
An issue that is sometimes mentioned and discussed in descriptions of median nerve decompression procedures is whether the palmar aponeurosis is dissected during the operation. Most often, it is only the transverse carpal ligament and the flexor retinaculum that are cut. The palmar aponeurosis, which is a continuation of the palmaris longus tendon, lies above these structures and does not need to be dissected to enable decompression of the nerve, especially when the skin incision is made in the area of the wrist line and extends into the palmar surface of the hand [5,6]. When the palmaris longus tendon is missing (10–20%), the palmar aponeurosis fuses directly with the flexor retinaculum [1,7]. In such cases, the aponeurosis may be partially dissected during the decompression procedure. When the incision is made more distally — on the palmar surface of the hand - the palmar aponeurosis is sometimes traversed and partly incised in order to reach the flexor retinaculum. Figure 5 shows the two common incisions for median nerve decompression surgery in patients with carpal tunnel syndrome described above.
Figure 5: The two most common skin incisions in carpal tunnel release surgery.
The authors recommend using a grooved probe or small spatula to protect the median nerve from above during the dissection of the flexor retinaculum. Decompression proceeds until the nerve has been released along its course. However, it should be remembered that when the release is proceeding towards the fingers, the anatomic location of the superficial arterial arch should not be crossed as this may cause additional bleeding. Roughly, the line that should not be crossed runs transversely as a continuation of the base of the thumb (Figure 6) [5,6,12].
Figure 6: Anatomic variants of the superficial arterial arch. The dashed line roughly indicates the border of nerve decompression towards the fingers which should not be crossed so as to avoid damage to the superficial arterial arch.
Possible complications of carpal tunnel surgery include: pain due to neuroma formation following transection of palmar cutaneous branch of median nerve, injury to recurrent thenar (motor) branch of median nerve, direct injury to the main trunk of the median nerve, hypertrophic scar causing compression of median nerve, injury to superficial arterial palmar arch , infection, hematoma [1,8].
Conclusion
Awareness of the fine anatomic and topographic details of the surgical site helps avoid complications and/or lack of improvement following median nerve decompression surgery. The use of correct anatomic nomenclature facilitates the exchange of views and discussions about methods of treatment and surgical outcomes among surgeons who carry out these procedures.
References
- Greenberg MS. Peripheral nerves. Peripheral neuropathies. Greenberg MS, editors. Handbook of Neurosurgery. 6th edn. New York: Thieme. 2006; 565-569.
- Shapiro BE, Preston DC. Entrapment and compressive neuropathies. Med Clin North Am. 2009; 93: 285-315, vii.
- Wessel LE, Fufa DT, Boyer MI, Calfee RP. Epidemiology of carpal tunnel syndrome in patients with single versus multiple trigger digits. J Hand Surg Am. 2013; 38: 49-55.
- Okamura A, Meirelles LM, Fernandes CH, Raduan Neto J, Dos Santos JB, Faloppa F. Evaluation of patients with carpal tunnel syndrome treated by endoscopic technique. Acta Ortop Bras. 2014; 22: 29-33.
- Bochenek A, Reicher M. Brachial plexus. Median nerve. Lasinski W, editors. In: Human anatomy. 4th edn. Warszawa: Wydawnictwo Lekarskie PZWL. 1998: 51-61.
- Sobotta J. Head, neck, upper limb. Putz R, Pabst R, editors. In: Human anatomy. 1st edn. Wroclaw; Urban and Partner. 1994.
- Demircay E, Civelek E, Cansever T, Kabatas S, Yilmaz C. Anatomic variations of the median nerve in the carpal tunnel: a brief review of the literature. Turk Neurosurg. 2011; 21: 388-396.
- Louis DS, Greene TL, Noellert RC. Complications of carpal tunnel surgery. J Neurosurg. 1985; 62: 352-356.
- Vargas A, Chiapas-Gasca K, Hernandez-Diaz C, Canoso JJ, Saavedra MA, Navarro-Zarza JE, et al. Clinical anatomy of the hand. Reumatol Clin. 2012; 8: 25-32.
- Waloch J, Skawina A, Gorczyca J. Upper limb. Carpal tunnel. Skawina A, editors. In: Human anatomy. Cracow, Jagiellonian University Press. 2002: 65.
- Eversman WW. Entrapment and compression neuropathies. Green DP, editors. Operative hand surgery. 3rd edn. New York: Churchil Livingstone. 1993; 1356-1365.
- Phalen GS. The carpal-tunnel syndrome. Clinical evaluation of 598 hands. Clin Orthop Relat Res. 1972; 83: 29-40.
- Zyluk A, Puchalski P. A comparison of the results of carpal tunnel release in patients in different age groups. Neurol Neurochir Pol. 2013; 47: 241-246.
- Wilbourn AJ, Gilliatt RW. Double-crush syndrome: a critical analysis. Neurology. 1997; 49: 21-29.
- Agee JM, McCarroll HR Jr, Tortosa RD, Berry DA, Szabo RM, Peimer CA. Endoscopic release of the carpal tunnel: a randomized prospective multicenter study. J Hand Surg Am. 1992; 17: 987-995.
- Chow JC. Endoscopic release of the carpal ligament for carpal tunnel syndrome: 22-month clinical result. Arthroscopy. 1990; 6: 288-296.
- Trumble TE, Diao E, Abrams RA, Gilbert-Anderson MM. Single-portal endoscopic carpal tunnel release compared with open release : a prospective, randomized trial. J Bone Joint Surg Am. 2002; 84-84A: 1107-15.
- Ferdinand RD, MacLean JG. Endoscopic versus open carpal tunnel release in bilateral carpal tunnel syndrome. A prospective, randomised, blinded assessment. J Bone Joint Surg Br. 2002; 84: 375-379.
- Brief R, Brief LP. Endoscopic carpal tunnel release: report of 146 cases. Mt Sinai J Med. 2000; 67: 274-277.