
Case Presentation
Austin Neurosurg Open Access. 2015;2(1): 1026.
Severe Coronal Deformity Correction with a Modification of the Transpsoas Technique: Technical Note
Namath S. Hussain¹* and Rod J. Oskouian²
¹Department of Neurosurgery, Penn State Hershey Medical Center, USA
²Department of Neurosurgery, Swedish Medical Center, USA
*Corresponding author: Namath S. Hussain,Department of Neurosurgery, Penn State Hershey Medical Center, 30 Hope Drive, EC110, Hershey, PA 17033, USA
Received: January 13, 2015; Accepted: June 12, 2015; Published: June 15, 2015
Abstract
Objective: To review the proper patient selection and the rationale for the transpoas technique and when it should be applied as a useful approach in spine surgery. Our study aims to describe a technical variance with regard to the lateral transpoas interbody access and fusion technique.
Methods: Using two descriptive cases, we aim to illustrate how this technical variance can be utilized to the spine surgeon’s advantage in cases where the patient’s anatomy brings along additional challenges to the traditional technique. Emphasis was placed on examining and describing coronal pathology and how it can be corrected.
Results: Preoperative films along with intraoperative pearls are reviewed along with postoperative film results such that the reader will gain a balanced overview of the transpsoas technique and how its versatility can be applied for coronal pathology.
Conclusion: The transpoas technique can be modified to provide the spine surgeon with the ability to correct severe coronal deformity in a safe manner.
Keywords: Lateral interbody fusion; Retroperitoneal; Transpsoas; Minimally invasive spine surgery; Spine fusion
Introduction
Coronal deformity correction continues to be one of the most difficult challenges for scoliosis surgeons [1]. The minimally invasive lateral retroperitoneal transpsoas approach has become an increasingly popular corridor to correct coronal deformity. Scoliosis is a common cause of axial back pain with attendant signs and symptoms of radiculopathy, most commonly ipsilateral to the concavity of the coronal deformity [1,2]. Neurogenic claudication or instability along with varying degrees of spondylolisthesis are often encountered [3]. Abnormal spinal biomechanics due to these unbalanced forces can lead to paraspinal muscle fatigue and pain with associated degrees of Degenerative Disc Disease (DDD) which many patients undergo surgical intervention for. Chronic compression from foraminal stenosis and DDD can lead to extremity weakness, sensory disturbances, and pain [4,5]. Many different surgical approaches for the treatment of pain caused by severe coronal deformity have been developed and can range from the simple limited decompression to multi-level fusion procedures [3,6]. Prior to choosing a particular surgical technique, the goals of surgery must be clearly defined. The approach must be selected based on these goals, tempered with patient-specific anatomical and physiological factors that affect related risks and complications [4,7,8]. Neuromonitoring and other adjuncts must be utilized when needed to improve safety [4]. Open posterior approaches necessitate paraspinal muscle dissection and ligament removal which may lead to postoperative kyphotic or coronal deformity due to loss of facets and other stabilizing structures [2,6,9-11].
The minimally invasive Lateral Transpsoas Interbody Fusion (LTIF) has become an increasingly popular means of fusion because it obviates the need to disrupt the paraspinal corridor that the posterior and transforaminal approaches require [5,12,13]. Many authors have also cited the advantage of being able to indirectly decompress the foramina through the placement of a large graft that can increase disc space height [4,5,14] and improve fusion rates [12]. The attendant risks of lumbar plexus injuries from this approach can be decreased with a comprehensive understanding of patient-specific regional anatomy; especially when there is a degree of coronal deformity which can lead to stretching of the psoas over a rigid convex spine. Utilization of directional intraoperative Electromyography (EMG) is an important key to identifying the location of the femoral and other nerves [4,15].
LTIF is a new minimally invasive technique to approach the spine from a lateral corridor that has been in used for the past six years. As more and more data are collected with regard to anatomy, technique, and complications [4,13], clinicians are becoming more comfortable with utilizing the technique for a wide variety of indications and pathologies. As surgeons become more skilled at using the technique for DDD and other more common pathologies [12], they have ventured into more complex cases with unique anatomic challenges [5,16]. Deformity correction is an area where industry-surgeon collaborations have brought new products into the market that have improved surgeons’ abilities to gain access to the disc space in a safe manner and correct fixed deformities without the attendant risks associated with open, more invasive approaches. We detail in our report the use of a new technique variance of the transpsoas approach for deformity correction utilizing angled instrumentation to gain access to severely coronally offset disk spaces and a new set of instrumentation to gain access safely to correct deformity and gain an adequate fusion. This new technique has been used at our institution for the past three years with excellent results but has not been reported in the literature as of yet.
Methods
We examined the preoperative and intraoperative films of two patients who underwent elective LTIF. We have used this technique in over ten cases over the past two years. We have performed over 200 LTIF procedures over the past two years. Fusion status, deformity correction, and construct integrity were assessed with dynamic radiographs. Both patients underwent LTIF with the companybranded version known as an extreme lateral interbody fusion (XLIF; NuVasive, San Diego, CA, USA) with an 18-26 mm × 8-10 mm × 50-60 mm cage. The vertebral body below was used to determine the docking landmark for the transpsoas retractor. Directional EMG was utilized to ensure docking of the retractor was anterior to the femoral nerve. Disc height restoration with the large interbody graft improved forminal cross-sectional area and with partial correction of the coronal deformity.
Cases
There were no anesthesia-related intraoperative complications or surgical adverse events. Both patients experienced transient postoperative anterior thigh numbness that resolved by 3 months. There were no permanent neurological deficits. There were no hardware failures or pseudoarthrosis noted during follow up.
Case presentation 1
Our first patient presented with severe axial low back pain and right lower extremity radiculopathy without any lower extremity weakness. Radiographs and CT imaging of the lumbar spine (Figure 1), (Figure 2) revealed severe degenerative disease with massive osteophyte buildup leaving the patient with severe right foraminal stenosis at multiple lumbar levels. The patient was taken to the operating room for LTIF at the L2-3 and L3-4 levels. Preoperative films (Figure 1), (Figure 2), intraoperative films with slides inserted (Figure 3), and postoperative films (Figure 4), (Figure 5) are shown.
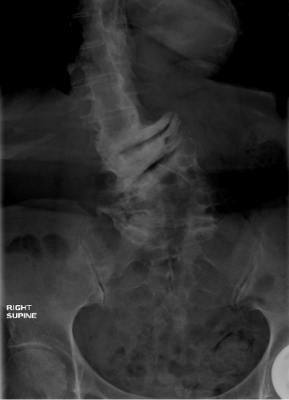
Figure 1: Standing full cassette films of the thoracolumbar spine showing the
severe degree of preoperative coronal and rotatory scoliotic deformity.
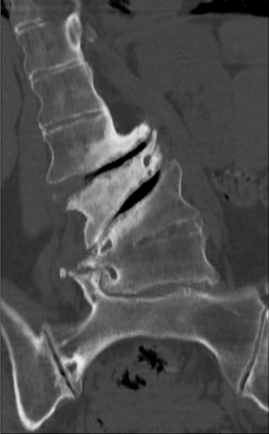
Figure 2: CT imaging of the thoracolumbar spine showing the severe degree
of osteophyte buildup along the convexity of the deformity leading to highgrade
foraminal stenosis.
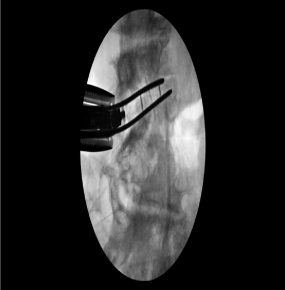
Figure 3: Intraoperative films with slides inserted in preparation for interbody
graft placement.
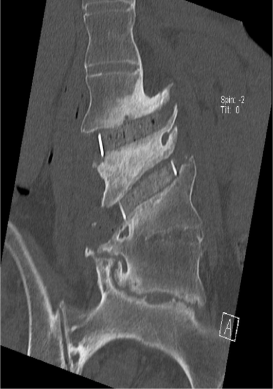
Figure 4: Coronal postoperative CT images with interbody graft in place.
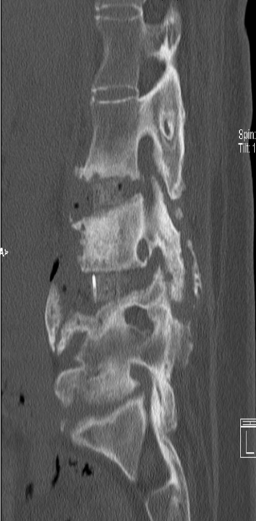
Figure 5: Sagittal postoperative CT images with interbody graft in place.
Case presentation 2
The second patient had a similar presentation except he also had bilateral lower extremity radiculopathy and neurogenic claudication and underwent a similar procedure at the L2-3, L3-4, and L4-5 levels. Preoperative and postoperative films are shown in Figure 6.
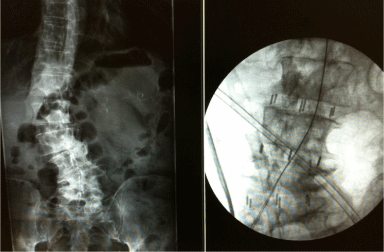
Figure 6: Preoperative and postoperative radiographs showing the degree of
coronal deformity correction achieved with interbody graft placement at the
L2-3, L3-4, and L4-5 levels.
Surgical Technique
The patient is typically positioned in the lateral decubitus position with the left side up. This is due to the position of the aorta relative to the spine and vena cava. With the aorta on the left side of the spine, it is the structure that is most likely to be injured versus the vena cava. Due to its more muscular nature, the aorta is more amenable to primary repair in the event of inadvertent laceration. Preoperative CT imaging in all patients is critical to making this procedural decision. Another consideration is the apex of the deformity and surgical planning with regard to the number of skin incisions required and the need for psoas retraction. At times, it is advantageous to approach the spine from the convex side because it is easier to enter the disc space. However, the disadvantage of this approach is the need for multiple skin incisions at times to reach multiples disk levels and the need to retract against the psoas while it is already stretched taught over the lumbar convexity. At other times, it is advantageous to approach from the concave side because it is easier to treat multiple disc spaces through the concave size but osteophytes and bony overgrowth over the space may make entering the actual disc space difficult. The last variable to consider is that the psoas is not stretched in this position and may be fatter. This means that there is a greater thickness of psoas that needs to be dissected through to enter the disk space making nerve injury more likely. There are many variables to consider when choosing which side to make the approach on during a transpsoas approach such that surgeon experience and knowledge of the patients’ pathology and preoperative anatomy are of critical importance to ensure good outcomes and cure of the operative pathology.
After the patient is positioned in the lateral decubitus position, the patient’s hip and legs are taped down to the bed and the table is broken at the iliac crest to open up the area between the iliac crest and ribs. X-rays are taken to make sure that the disc space is in a perfect lateral trajectory on AP fluoroscopic imaging. Lateral films are taken to ensure that there is no rotation and that pedicles can be seen. The patient is then taped down more firmly at the hip and at the shoulders. Neuromonitoring leads are placed at this time for use intraoperatively to avoid damage to the lumbar plexus during dilation through the psoas muscle.
After the 3cm skin incision is made, the anterior abdominal wall and oblique musculature are separated with peons. No muscle incision or cautery should be required at this step. Once the fascia is encountered, a blunt peon is used to puncture through the fascia into the retroperitoneal space which is confirmed when retroperitoneal fat is seen. Sequential dilators are placed with intraoperative monitoring to ensure that no nerves are in close proximity. The dilators are navigated down to the disk space under strict fluoroscopic guidance. When nerves are detected with intraoperative stimulation, the dilator is repositioned. Once full dilation is achieved, the retractor is placed over the largest dilator and opened. Pressure should be placed on the retractor downward against the disk space to ensure that all of the psoas fibers are behind the retractor blades and are separated. Once the disk space is visualized, the disk annulus is cut and disk material is removed with ronguers and Kerrison punch instruments (Figure 7). After the disk space is clear, the interbody graft is placed.

Figure 7: Coronal angled instruments used for discectomy and deformity
correction.
In cases of severe deformity, cleaning out the disk space and placing the interbody graft can be quite challenging. Angled currettes and rasps can be useful at this step. It is also critical to release the contralateral annulus with an angled Cobb elevator to release the fixed deformity. Placement of the actual interbody graft can be assisted with slides that protect the endplates and provide a semirigid pathway for the graft to enter the disk space (Figure 3). Once the slides are placed, the interbody graft is attached onto an angled inserted and malletted down in between the slides into the disk space under fluoroscopic guidance. Frequent AP and lateral images are taken to ensure that the graft is entering the disk space and not being placed too anteriorly into the peritoneal space or into the great vessels or too posteriorly into the spinal canal. X-rays also ensure that the graft is not being hammered into the vertebral body endplate. After the interbody graft is in proper position, the slides are removed and repeat radiographs are taken to ensure that the graft has not moved during slide removal.
Discussion
There have been several previous reports detailing the transpsoas approach for lateral interbody fusion. Isaacs et al review some of the current literature in their recent report along with complication rates [12]. The largest review of transpsoas interbody fusion in the literature is a paper by Rodgers et al that reviews 600 cases for a total of 741 levels treated by the approach [17]. They found an average hemoglobin change of 1.38 and average hospital stay of 1.21 days. The incidence of complications was 6.2%, with 1.5% being in-hospital surgery-related events, 2.8% being in-hospital medical events, 1.0% being out-of-hospital surgery-related events, and 0.8% being out-ofhospital medical events. 0.7% of patients had transient postoperative neurologic deficits, the most common being psoas weakness, which had all resolved within three months.
The goal of surgical intervention in patients with coronal deformity includes not only deformity correction but also stabilization of the motion segment and indirect neural decompression of spinal nerves exiting along the concavity [5]. Challenges with traditional posterior approaches to the spine can be placement of the interbody cage [1,3,9,18], which is critical especially when percutaneous pedicle screw fixation is used posteriorly because the interbody construct is responsible for a greater degree of the postoperative stability versus if an open posterolateral fusion was also completed. Transient sensory deficits when they do occur most likely represent mild neuropraxia which usually resolves [4,19].
The importance of a detailed understanding of the patient-specific regional anatomy is critical due to the fact that coronal malalignment can produce multiple other concomitant anatomical changes in the development of the psoas musculature, the transverse processes, and surrounding ligamentous structures. Directional real-time EMG monitoring is an important tool for determining the trajectory of the lumbar plexus motor nerves when using minimally invasive lateral transpsoas access to the spine [4,12,14,15]. Depending on the degree of scoliotic deformity, the radiographic docking region may be more anterior or posterior which again highlights the importance of realtime directional EMG in these cases. Over the concavity, the psoas muscle is usually more lax; however, not to a great degree because of the fact that there may be developmental shortening of the muscle itself. The psoas with its attendant nerves over the convexity are usually more taught and can be more susceptible to injury during docking and opening of the retractor blades.
Transient sensory deficits resolved within three months without motor involvement. In both patients the indirect decompression and correction of the coronal deformity resolved preoperative lower extremity radicular symptoms. The patients did not experience weakness or foot drop related to the deformity correction.
Surgeons should understand that there is a significant learning curve associated with the minimally invasive transpsoas approach to the spine [4]. Reliance on radiographs and EMG places the surgeon at somewhat of a disadvantage when considering traditional approaches that they may be currently more comfortable with. Cadaveric dissections along with instrumentation courses can give surgeons the experience they need to tackle these cases while minimizing patient risk.
Tormenti et al. reported a series of eight patients with scoliosis who underwent transpsoas interbody fusion with a coronal Cobb angle improvement from an average of 38.5 degrees to 10 degrees [15]. In their study, two patients suffered from postoperative motor radiculopathies while six patients experienced postoperative thigh dysesthesias. Other reports have indicated an incidence of postoperative psoas hematoma of 2.1% - 9.1% [1,10].
In their review of 58 patients, Knight et al report two permanent motor deficits and four cases that were aborted due to neural element proximity [8]. All four cases were at the L4-5 level. This rate is much higher than that reported by others in the literature [16]. The most devastating complications associated with open anterior lumbar procedures are vascular and visceral injuries [4,7,10,20]. The rates of these injuries in most large series are about three percent [21]. Other more rare complications include pneumothorax, hemothorax, chyloretroperitoneum [8], and ureter or bladder injury. In order to decrease complications, new techniques have been developed to provide less invasive approaches to the disk space. Minimallyinvasive techniques have provided newer approaches to the spine that have led to better outcomes [4] and are now the preferred method for spinal surgery.
Conclusion
Transpsoas techniques to the lumbar spine provide an excellent new minimally invasive access point for interbody fusion. This approach provides a large corridor to place a larger graft into the disk space, which is useful to reduce large coronal deformities. At times, due to severe deformities, entering the disk space or placing the interbody graft can be quite challenging. Many authors have reported complications such as damaging the vertebral body endplate or damaging the lumbar plexus causing motor weakness or postoperative parasthesias or dysesthesias [4,13,22]. To decrease the risk of these and other complications, we have developed this new technique with the use of angled currettes, rasps, and other disk space cleaning instruments along with angled inserters for the interbody graft and slides to guide the graft into proper position to assist the surgeon in achieving good disk space preparation and interbody graft placement while decreasing the patient’s disk of postoperative complications associated with placement of straight instruments into a coronally deformed disk space.
LTIF is an effective adjunct to the scoliosis surgeon’s armamentarium which may be particularly helpful when confronted with severe coronal deformity. This new minimally invasive technique can help treat patients without undue approach-related morbidity in a safe manner and with good results.
References
- Cho KJ, Suk SI, Park SR, Kim JH, Kim SS, Choi WK, et al. Complications in posterior fusion and instrumentation for degenerative lumbar scoliosis. Spine (Phila Pa 1976). 2007; 32: 2232-2237.
- Carpenter CT, Dietz JW, Leung KY, Hanscom DA, Wagner TA. Repair of a pseudarthrosis of the lumbar spine. A functional outcome study. J Bone Joint Surg Am. 1996; 78: 712-720.
- Mardjetko SM, Connolly PJ, Shott S. Degenerative lumbar spondylolisthesis. A meta-analysis of literature 1970-1993. Spine (Phila Pa 1976). 1994; 19: 2256S-2265S.
- Hussain NS, Perez-Cruet MJ. Complication management with minimally invasive spine procedures. Neurosurg Focus. 2011; 31: E2.
- Pimenta L, Oliveira L, Schaffa T, Coutinho E, Marchi L. Lumbar total disc replacement from an extreme lateral approach: clinical experience with a minimum of 2 years' follow-up. J Neurosurg Spine. 2011; 14: 38-45.
- Rousseau MA, Lazennec JY, Bass EC, Saillant G. Predictors of outcomes after posterior decompression and fusion in degenerative spondylolisthesis. Eur Spine J. 2005; 14: 55-60.
- Asha MJ, Choksey MS, Shad A, Roberts P, Imray C. The role of the vascular surgeon in anterior lumbar spine surgery. Br J Neurosurg. 2012.
- Hussain NS, Hanscom D, Oskouian RJ. Chyloretroperitoneum following anterior spinal surgery. J Neurosurg Spine. 2012.
- Cassinelli EH, Wallach C, Hanscom B, Vogt M, Kang JD. Prospective clinical outcomes of revision fusion surgery in patients with pseudarthrosis after posterior lumbar interbody fusions using stand-alone metallic cages. Spine J. 2006; 6: 428-434.
- Scaduto A, Gamradt SC, Yu WD, Huang J, Delamarter RB, Wang JC. Perioperative complications of threaded cylindrical lumbar interbody fusion devices: anterior versus posterior approach. J Spinal Disord Tech. 2003; 16: 502-507.
- Tormenti MJ, Maserati MB, Bonfield CM, Gerszten PC, Moossy JJ, Kanter AS, et al. Perioperative surgical complications of transforaminal lumbar interbody fusion: a single-center experience. J Neurosurg Spine. 2012; 16: 44-50.
- Isaacs RE, Hyde J, Goodrich JA, Rodgers WB, Phillips FM. A prospective, nonrandomized, multicenter evaluation of extreme lateral interbody fusion for the treatment of adult degenerative scoliosis: perioperative outcomes and complications. Spine (Phila Pa 1976). 2010; 35: S322-S330.
- Rodgers WB, Cox CS, Gerber EJ. Experience and early results with a minimally invasive technique for anterior column support through extreme lateral interbody fusion: XLIF. US Musculoskeletal Review. 2007; 1: 28-32.
- Knight RQ, Schwaegler P, Hanscom D, Roh J. Direct lateral lumbar interbody fusion for degenerative conditions: early complication profile. J Spinal Disord Tech. 2009; 22: 34-37.
- Tormenti MJ, Maserati MB, Bonfield CM, Okonkwo DO, Kanter AS. Complications and radiographic correction in adult scoliosis following combined transpsoas extreme lateral interbody fusion and posterior pedicle screw instrumentation. Neurosurg Focus. 2010; 28: E7.
- Rodgers WB, Cox CS, Gerber EJ. Early complications of extreme lateral interbody fusion (XLIF) in the obese. J Spinal Disorder Tech. 2010; 23: 393-397.
- Rodgers WB, Gerber EJ, Patterson J. Intraoperative and early postoperative complications in extreme lateral interbody fusion: an analysis of 600 cases. Spine (Phila Pa 1976). 2011; 36: 26-32.
- Perez-Cruet MJ, Welsh RJ, Hussain NS, Begun EM, Lin J, Park P. Use of the da vinci minimally invasive robotic system for resection of a complicated paraspinal schwannoma with thoracic extension: case report. Neurosurgery. 2012; 71: 209-214.
- Ozgur BM, Aryan HE, Pimenta L, Taylor WR. Extreme lateral interbody fusion (XLIF): a novel surgical technique for anterior lumbar interbody fusion. Spine J. 2006; 26: 435-43.
- Oskouian RJ, Johnson JP. Vascular complications in anterior thoracolumbar spinal reconstruction. J Neurosurg. 2002; 96: 1-5.
- Sasso RC, Best NM, Mummaneni PV, Reilly TM, Hussain SM. Analysis of operative complications in a series of 471 anterior lumbar interbody fusion procedures. Spine (Phila Pa 1976). 2005; 30: 670-674.
- McAfee PC, Phillips FM, Andersson G, Buvenenadran A, Kim CW, Lauryssen C, et al. Minimally invasive spine surgery. Spine (Phila Pa 1976). 2010; 35: S271-S273.