
Research Article
Austin Neurosurg Open Access. 2016; 3(1): 1045.
Large Skull Defect is Associated with the Development of Brain Herniation during Continuous Lumbar Drainage
Qian Zhouqi and Wang Ke*
Neurosurgical Department, Shanghai Tenth People’s Hospital, Tongji University, China
*Corresponding author: Wang Ke, Neurosurgical Department, Shanghai Tenth People’s Hospital, Tongji University, 301 Middle Yanchang Road, Shanghai, China
Received: November 09, 2015; Accepted: March 01, 2016; Published: March 04, 2016
Abstract
Objective: This study investigated risk factors associated with the development of brain herniation during Continuous Lumbar Drainage (CLD) in neurosurgical patients.
Methods: We reviewed medical charts of the patients who consecutively received CLD in our department between January 2010 and Dec 2013. Clinical signs combined with radiographic evidence made the diagnosis of CLD-induced brain herniation. Clinical characteristics between the patients with and without CLD-induced brain herniation were compared.
Results: A total of 108 patients were enrolled, including 80 male and 28 female patients. Among them, 8 (7.4%) patients developed CLD-induced brain herniation. There was no significant difference in patient age, gender, and surgical history before drainage between the patients with CLD-induced brain herniation and those without. Patients with CLD-induced brain herniation showed faster draining speed of Cerebrospinal Fluid (CSF) and a higher rate of large skull defect due to decompressive craniectomy than those without (12.8 ± 4.4 mL/h vs. 10.0 ± 3.3 mL/h, p=0.029; 62.5% vs. 17.0%, p=0.008, respectively). They also demonstrated lower pre-drainage Glasgow Coma Scale (GCS) scores than those without CLD-induced brain herniation although the difference was not significant (p=0.085). After adjusting the effects of the draining speed and pre-drainage GCS score by logistical regression analysis, large skull defect was independently related to a nearly nine-fold increased risk of CLD-induced brain herniation.
Conclusion: Large skull defect is a risk factor with the development of brain herniation during CLD. Altered CSF dynamics in the presence of large skull defect may play a role in the process.
Keywords: Brain herniation; Lumbar drainage; Large skull defect
Introduction
Continuous Lumbar Drainage (CLD) is a commonly used procedure in neurosurgical practice. This procedure is used to prevent cerebral vasospasm after aneurysmal Subarachnoid Hemorrhage (SAH) [1,2], to treat postoperative or posttraumatic Cerebrospinal Fluid (CSF) fistula [3,4], to manage communicating hydrocephalus after intracerebral hemorrhage [5], or even to reduce refractory intracranial hypertension in traumatic brain injury [6,7]. However, it is not a risk-free procedure. Complications of this procedure have been reported in previous studies, including headache, nerve root pain, meningitis, pneumocephalus, intracerebral hemorrhage, and brain herniation [8-10]. Among them, brain herniation is the most severe complication during CLD, which may result in brain stem dysfunction or even death [11]. Few studies have investigated risk factors associated with such a catastrophic complication in neurosurgical patients who receive CLD treatment. Here, we conducted a retrospective study to reveal its incidence and risk factors.
Patients and Methods
With a waiver of written consent from the Institutional Review Board, we retrospectively reviewed medical charts and radiographic information of the patients who consecutively received CLD in our department between January 2010 and December 2013. Patients with concomitant internal or external ventricular drainage during CLD were excluded from this study. Indications for the usage of CLD in our department included aneurysmal or traumatic SAH, CSF infection, CSF fistula, and communicating hydrocephalus.
Before CLD, We routinely measured opening pressure in lumbar cistern lumbar puncture, and patients with a pressure exceeding 26 cmH2O were excluded from CLD. Patients with coagulation disorders or clinical signs of impending brain herniation were also excluded, which included pupillary abnormalities, compression or absence of basal cisterns on Computered Tomography (CT) scans. The Medtronic Neurosurgical EDM Lumbar Drainage Kit Catheter (Medtronic, MN: USA) was used according to manufacturer’s instructions. The patient was placed in the lateral decubitus position and flexed for insertion of the draining catheter. A 14-gauge Tuohy needle was introduced into the subarachnoid space via the L4/L5 or L3/L4 interspinous space using routine sterile technique. The catheter was inserted through the needle, with the proximal end of 5 cm in the subarachnoid space. The distal end of the catheter was hooked up to a closed draining system with a drip chamber. After the procedure, the patient was strictly kept at bed rest but allowed to turn from side to side and could sit up to less than 45-degree in bed. A target draining speed of 8-10 mL/h was set and the drip chamber was adjusted according to patient’s head. Clinical and neurological conditions were evaluated hourly during the drainage.
CLD-induced brain herniation was diagnosed by clinical signs combined with radiographic evidence: (1) a decrease of Glasgow Coma Scale (GCS) score; (2) unilateral or bilateral pupillary abnormalities; (3) compression or absence of basal cisterns on CT scans, which was not ascribed to new formation or progression of intracranial mass lesions.
Patient demographic features and medical history were reviewed. Average draining speed was calculated by total CSF volume drained out divided by the duration of lumbar drainage in the patients without cerebral herniation. However, this value in those with cerebral herniation referred to the average draining speed before the occurrence of brain herniation.
Continuous data were expressed as mean ± Standard Deviation (SD), and categorical data were expressed as median (Interquartile Range, IQR) or the percentage. All statistical analyses were performed with SPSS 16.0 for windows. The p values were derived from a twotailed unpaired Student’s t-test, the Mann–Whitney test, Pearson’s chi-square test, or the Fisher’s exact test. Logistical regression analysis was performed to adjust the effects of confounders. Differences were considered significant if p value was below 0.05.
Results
A total of 108 patients, 80 male and 28 female, were enrolled, with a mean age of 53.2 years old (Table 1). Indications for CLD included SAH in 36 patients, CSF infection in 53 patients, CSF fistula in 15 patients, and communicating hydrocephalus in 4 patients. Seventyseven patients (71.2%) received brain or spine surgery before the drainage. Among them, 22 patients received decompressive frontotempero- parietal craniectomy, which left them a unilateral large supratentorial skull defect. Sixty-one patients (56.5%) showed obviously decreased mental status (GCS<14) before the drainage.
All patients (n=108)
Patients without brain herniation (n=100)
Patients with brain herniation (n=8)
P value
Age (yrs), Mean ± SD
53.2 ± 14.0
53.8 ± 13.8
45.5 ± 14.1
0.100
Gender, Male
80 (74.1%)
72 (72.0%)
8 (100%)
0.109
Indications for CLD
Aneurysmal or traumatic SAH
36 (33.3%)
31 (31.0%)
5 (62.5%)
0.114
CSF infection
53 (49.1%)
50 (50.0%)
3 (37.5%)
0.716
CSF fistula
15 (13.9%)
15 (15.0%)
0
Hydrocephalus
4 (3.7%)
4 (4.0%)
0
Pre-drainage GCS score
Median (IQR)
13.0 (9.0-15.0)
13.0 (9.0-15.0)
10.5 (7.25-12.0)
0.085
14-15
47 (43.5%)
47 (47.0%)
0
0.027
9-13
35 (32.4%)
30 (30.0%)
5 (62.5%)
0.109
3-8
26 (24.1%)
23 (23.0%)
3 (37.5%)
0.396
Craniotomy before drainage
77 (71.2%)
69 (69.0%)
8 (100%)
0.102
DC before CLD
22 (20.4%)
17 (17.0%)
5 (62.5%)
0.008
Average draining speed (mL/h)
10.2 ± 3.4
10.0 ± 3.3
12.8 ± 4.4
0.029
SAH: Subarachnoid Hemorrhage; CSF: Cerebrospinal Fluid; GCS: Glasgow Coma Scale; DC: Decompressive Craniectomy; SD: Standard Deviation; IQR: Interquartile Range.
Table 1: Demographic and clinical characteristics of the 108 patients.
The average draining speed was 10.2 ± 3.4 mL/h. Among a time period of drainage, eight (7.4%) patients developed CLD-induced brain herniation. The incidences of brain herniation in the subgroup of patients with SAH and CSF infection were 13.9% and 5.7%, respectively. None of the patients with CSF fistula or communicating hydrocephalus developed cerebral herniation after the drainage.
There was no significant difference in patient age, gender, and surgical history before drainage between patients with CLD-induced brain herniation and those without. However, patients with CLDinduced brain herniation showed faster draining speed and higher rates of a large skull defect due to decompressive craniectomy before drainage than those without (12.8 ± 4.4 mL/h vs. 10.0 ± 3.3 mL/h, p=0.029; 62.5% vs. 17.0%, p=0.008, respectively). Patients with CLDinduced brain herniation also showed a lower pre-drainage GCS score although the difference was not significant (p=0.085). After adjusting the effects of the draining speed and pre-drainage GCS score by logistical regression analysis, a large skull defect is an independent risk factor of CLD-induced brain herniation (p=0.007, Table 2). It was related to a nearly nine-fold increased risk of developing brain herniation during CLD.
Variables in equation
B
P �value
OR (95% CI)
Average draining speed
0.2
0.043
1.2 (1.0 – 1.5)
DC before drainage
2.2
0.007
8.9 (1.8 – 43.8)
DC: Decompressive Craniectomy; OR: Odds Ratio; CI: Confidence Interval.
Table 2: The result of logistic regression analysis.
Illustrative Case a 51 year-old male patient who suffered from traumatic brain injury due to falling. He had a GCS score of 7 at admission with both pupils normal-sized and responsive to light. A head CT scan on the emergency department showed an acute subdural hematoma one the right side with a mild midline shift (Figure 1A). He was transmitted to the operating room and received emergency hematoma evacuation with decompressive craniectomy (Figure 1B). After the surgery, the GCS score rose to 9, and he was transferred to the neuro-critical care unit. His neurological conditions gradually improved, and the GCS score increased to 12 on the tenth day after the surgery. The head CT scan showed the presence of basal cisterns (Figure 1C). At that time, he suffered from intermittent high fever with the peak temperature of 39°C. The result from lumbar puncture showed blood-containing CSF with opening pressure in lumbar cistern of 15 cmH2O. A continuous CSF drainage was placed to drain the blood-containing CSF at an average rate of 8 ml/h. Six hours after drainage, the GCS score decreased to 6, and both pupils became light-sluggish. An emergent CT scan showed the absence of basal cisterns (Figure 1D). The CSF drainage was immediately clipped and the patient was flat positioned. After 24 h, the GCS score returned to 12, and the pupils became light-reactive. The CT scan 24 h after discontinuation of lumbar drainage showed the reappearance of basal cistern (Figure 1E). The patient regained consciousness before being discharged.
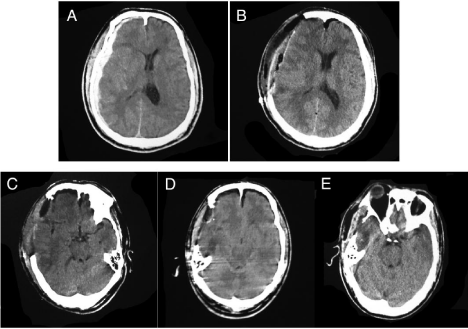
Figure 1: Head CT scans at different time points.
Discussion
Brain herniation is an acute and severe complication after lumbar puncture or during lumbar drainage. Elevated intracranial pressure and a fast draining speed are well-known risk factors of this complication following lumbar puncture [12,13]. In this study, we reviewed the patients who received CLD in our department during an over 3-year period, with emphasis on risk factors of CLD-induced brain herniation. Our study showed that a large skull defect was associated with an increased risk of this devastating event.
Brain herniation is abnormal shift of brain parenchyma driven by a pressure gradient between two compartments. Free and sufficient CSF communication between the compartments avoids the formation of such a pressure gradient. When lumbar drainage is instituted, a pressure gradient between intracranial compartment and lumbar cisterns appears. Normally, the pressure gradient drives the cephalocaudal flow of CSF. However, when the flow of CSF is blocked and the pressure gradient reaches a threshold value, the pressure gradient will drive the brain downward and result in brain herniation. A CSF pressure gradient over 10 mmHg between the intracranial compartment and the lumbar cistern may be adequate to induce transtentorial herniation [14]. Note worthily, this threshold value derived from the patients with normal cranial integrity. It remains unknown what the threshold value will be in the setting of a large skull defect. Separate case reports revealed the development of brain herniation, termed as paradoxical herniation, after lumbar puncture or drainage among patients with a large skull defect [15-17]. These studies implicated the influence of the atmosphere on CSF dynamics via the skull defect. This study showed that a large skull defect was associated with an increased risk of CLD-induced brain herniation. We hypothesize that destroyed cranium integrity such as a large skull defect makes the atmosphere interfere with CSF dynamics, which predisposes the occurrence of brain herniation.
The incidence of brain herniation in this study was 7.4%, which was higher than that in previous reports [3,18]. That is possibly ascribed to broad indications of CLD in this study, including its usage for CSF infection. Also, it is noteworthy that the incidence of brain herniation was distinct between the subgroups of patients. The incidence in the subgroup of patients with SAH was 13.9%, similar to that in a recent study conducted in patients with severe SAH (13.9% vs. 9.1%, p>0.05) [14]. None of the 15 patients with CSF fistula developed brain herniation during CLD, which is also comparable to previous studies [3,8]. These findings suggest that abnormal CSF characteristics may have an impact on the occurrence of brain herniation, possibly due to the altered CSF liquidity and dynamics.
In this study, all the eight patients who developed CLD-induced brain herniation showed impaired consciousness before the drainage. Patients who developed CLD-induced brain herniation showed a lower pre-drainage GCS score than those without although the difference did not reach a significant level. Until now, there is no evidence to support the association between impaired consciousness and the development of CLD-induced brain herniation. Therefore, future studies are warranted to elucidate the relationship between them.
Certain limitations of this study should be noted. Due to the retrospective design, this study inevitably has sources of bias and variation. Additionally, a limited number of patients were enrolled in this study, which may have weakened its statistical power. Therefore, futures studies with a larger sample size are necessary to confirm these findings. Despite these limitations, this study indicates that CLDinduced brain herniation is not a rare complication in neurosurgical practice. A large skull defect is associated with an increased risk of this event, possibly due to altered CSF dynamics.
References
- Klimo P Jr, Kestle JR, MacDonald JD, Schmidt RH. Marked reduction of cerebral vasospasm with lumbar drainage of cerebrospinal fluid after subarachnoid hemorrhage. J Neurosurg. 2004; 100: 215-224.
- Al-Tamimi YZ, Bhargava D, Feltbower RG, Hall G, Goddard AJ, Quinn AC, et al. Lumbar drainage of cerebrospinal fluid after aneurysmal subarachnoid hemorrhage: a prospective, randomized, controlled trial (LUMAS). Stroke. 2012; 43: 677-682.
- Dalgic A, Okay HO, Gezici AR, Daglioglu E, Akdag R, Ergungor MF. An effective and less invasive treatment of post-traumatic cerebrospinal fluid fistula: closed lumbar drainage system. Minim Invasive Neurosurg. 2008; 51: 154-157.
- Shapiro SA, Scully T. Closed continuous drainage of cerebrospinal fluid via a lumbar subarachnoid catheter for treatment or prevention of cranial/spinal cerebrospinal fluid fistula. Neurosurgery. 1992; 30: 241-245.
- Huttner HB, Schwab S, Bardutzky J. Lumbar drainage for communicating hydrocephalus after ICH with ventricular hemorrhage. Neurocrit Care. 2006; 5: 193-196.
- Tuettenberg J, Czabanka M, Horn P, Woitzik J, Barth M, Thomé C, et al. Clinical evaluation of the safety and efficacy of lumbar cerebrospinal fluid drainage for the treatment of refractory increased intracranial pressure. J Neurosurg. 2009; 110: 1200-1208.
- Münch EC, Bauhuf C, Horn P, Roth HR, Schmiedek P, Vajkoczy P. Therapy of malignant intracranial hypertension by controlled lumbar cerebrospinal fluid drainage. Crit Care Med. 2001; 29: 976-981.
- A?ikbas SC, Akyüz M, Kazan S, Tuncer R. Complications of closed continuous lumbar drainage of cerebrospinal fluid. Acta Neurochir (Wien). 2002; 144: 475-480.
- Saito K, Inamasu J, Kuramae T, Nakatsukasa M, Kawamura F. Tension pneumocephalus as a complication of lumbar drainage for cerebral aneurysm surgery--case report. Neurol Med Chir (Tokyo). 2009; 49: 252-254.
- Rumpf JJ, Dlaske H, Hansen N, Seiler C, Solymosi L, Muellges W. Progressive multilocal supratentorial hemorrhages as a rare complication of artificial CSF drainage after lumbar disc surgery. Clin Neuroradiol. 2012; 22: 157-159.
- Gower DJ, Baker AL, Bell WO, Ball MR. Contraindications to lumbar puncture as defined by computed cranial tomography. J Neurol Neurosurg Psychiatry. 1987; 50: 1071-1074.
- Sharp CG, Steinhart CM. Lumbar puncture in the presence of increased intracranial pressure: the real danger. Pediatr Emerg Care. 1987; 3: 39-43.
- Kapadia FN, Jha AN. Simultaneous lumbar and intraventricular manometry to evaluate the role and safety of lumbar puncture in raised intracranial pressure following subarachnoid haemorrhage. Br J Neurosurg. 1996; 10: 585-587.
- Staykov D, Speck V, Volbers B, Wagner I, Saake M, Doerfler A, et al. Early recognition of lumbar overdrainage by lumboventricular pressure gradient. Neurosurgery. 2011; 68: 1187-1191.
- Fields JD, Lansberg MG, Skirboll SL, Kurien PA, Wijman CA. "Paradoxical" transtentorial herniation due to CSF drainage in the presence of a hemicraniectomy. Neurology. 2006; 67: 1513-1514.
- Oyelese AA, Steinberg GK, Huhn SL, Wijman CA. Paradoxical cerebral herniation secondary to lumbar puncture after decompressive craniectomy for a large space-occupying hemispheric stroke: case report. Neurosurgery. 2005; 57: E594; discussion E594.
- Jung HJ, Kim DM, Kim SW. Paradoxical transtentorial herniation caused by lumbar puncture after decompressive craniectomy. J Korean Neurosurg Soc. 2012; 51: 102-104.
- A?ikbas SC, Akyüz M, Kazan S, Tuncer R. Complications of closed continuous lumbar drainage of cerebrospinal fluid. Acta Neurochir (Wien). 2002; 144: 475-480.