Case Report
Austin J Pharmacol Ther. 2014; 2 (10).1052
Morphine Overdose in a 6 ½ Week-Old Infant: A Case Report
Eagerton DH1*, Goodbar NH2, Dansby MC3, Abel SN4 and Bell WC5
Department of Pharmaceutical and Administrative Sciences, Presbyterian College School of Pharmacy, USA
Department of Pharmacy Practice, Presbyterian College School of Pharmacy, USA
Walgreen’s Pharmacy, USA
Department of Pharmacy, Madigan Army Medical Center, USA
Forensic Services, South Carolina Law Enforcement Division, USA
*Corresponding author: : Eagerton DH, Department of Pharmaceutical and Administrative Sciences, Presbyterian College School of Pharmacy, 307 N Broad St, Clinton, SC 29325, USA
Received: July 11, 2014; Accepted: October 13, 2014; Published: October 14, 2014
Abstract
Objective: To report a case of a lethal morphine overdose in a breastfed infant secondary to medication transfer into breast milk.
Case Report: The mother of a 6 ½ week old breastfed infant contacted emergency services secondary to her child being unresponsive. The infant was pronounced dead on arrival of medical personnel to the scene, and the mother reported taking numerous medications including morphine sulfate. Autopsy of the infant revealed lethal levels of morphine in samples of her tissue. The mother was charged with homicide, which is the first case of homicide secondary to medication transfer into breastmilk.
Discussion: Medication transfer into breastmilk is dependent on many factors, including size and molecular weight, lipid solubility, protein binding, and pH. Infant response to the presence of medications in breastmilk is dependent upon age-related pharmacodynamic factors that change weekly in rapidly growing and maturing infants, specifically volume of distribution, growth patterns, organ maturation, and digestive abilities. Infants also have a lower fat concentration, therefore exposure to fat-soluble drugs like morphine have enhanced blood-brain barrier penetration and ultimate accumulation following prolonged exposure.
Conclusion: Enhanced awareness of medication exposure to infants via breastmilk is vital to prevent morbidity and mortality. An understanding of the mechanism of medication transport into breastmilk, as well as differences in infant response to medications based on pharmacodynamics, are crucial in the medical management of a breastfeeding mother. In order to minimize toxicity to the breastfeeding infant, utilizing the lowest effective dose for a minimal amount of time is the cornerstone recommendation.
Keywords: Pharmacodynamics; Lethal morphine; Paracellular; Breastmilk
Introduction
In recent years the use of opioid analgesics, including morphine has increased among the general population. The CDC reported in 2013 that for every woman that dies of a prescription painkiller overdose, 30 go to the emergency department for painkiller abuse or misuse [1]. Women of childbearing age are the population most likely to abuse or misuse prescription analgesics [1]. Abuse of prescription painkillers by pregnant and lactating women can put infants at risk, including neonatal abstinence syndrome and potential overdose. Between 2000 and 2009 reports of neonatal abstinence syndrome grew almost 300% in the United States [1].
Unfortunately there have not been many studies done on the concentrations of drugs found in the plasma and their relative transfer into the breast milk of a lactating mother. While the pharmacokinetics of morphine are known, there are individual characteristics of the mother and infant that ultimately effect the total concentration of drug. The concentration of morphine that is lethal to an infant has not been well documented and can differ significantly due to age, body weight, fat distribution, and renal and hepatic enzyme development. The mother’s metabolism of the drug is another factor that influences amount transferred into breast milk. The American Academy of Pediatrics (AAP) does consider morphine to be compatible with breastfeeding [2,3]. However, the AAP also states that an infant may have measurable blood concentrations of morphine following long-term repeated exposure [4].
We report a case of a lethal morphine overdose in a breastfed infant whose mother had prescriptions for morphine sulfate, along with numerous other medications. To our knowledge, this is the first case of a mother charged with homicide secondary to medication transfer via breast milk.
Case Report
Emergency services were contacted about a 6 ½ week-old infant girl who was reportedly unresponsive. Dispatcher instructed mother on CPR. Emergency services arrived approximately 12 minutes after 911 were called. Child was pronounced dead on arrival, and the coroner was requested at the scene. Interview with the coroner revealed that the mother went to bed breastfeeding the child around 11 pm the previous night. The father of the infant woke at 5 am the following morning to find the child cold and unresponsive with fixed pupils. The mother stated she last breastfed the child around 2 am. The mother also reported that a few days before, the child was sleeping more than usual and was lethargic with a decreased appetite. Two days prior to the death, parents called the pediatrician regarding congestion and “cold” symptoms. Since the child was not running a fever they did not visit the office but instead were instructed to use a chest rub and vaporizer. The mother also reported that she gave the infant 0.8 mL of 150 mg/5 mL acetaminophen to the child the night before. She said she alternated the acetaminophen with ibuprofen, but the exact dosage of ibuprofen given, if any, was not documented.
During the interview with the coroner the mother was asked about medications she was currently taking, and she reported that she was currently prescribed levetiracetam, baclofen, amlodipine, desvenlafaxine, clonazepam and hydrocodone/ibuprofen. The prescribed dosages, date of last fill, and tablets remaining in the bottles are listed in Table 1. Upon search of the residence, prescription bottles for milancipran, carisoprodol, gabapentin, ropinirole, morphine sulfate extended release, and caffeine tablets were found. Details of these medications can be found in Table 2. All prescription medications found in the residence were prescribed to the mother. The medication most pertinent to this case is the morphine sulfate extended release, which she did not admit to taking upon her interview with the coroner. This medication was originally prescribed to her in 1998 after a serious car accident that left her with a diagnosis of chronic pain. Further investigation revealed that the prescriber of the morphine was unaware that the patient was pregnant and subsequently breastfeeding an infant. Alternatively, the obstetrician and pediatrician were unaware that the mother was taking morphine.
Medication
Dose Prescribed
Last Filled
Quantity filled / Quantity remaining
Hydrocodone 7.5 mg � Ibuprofen 200 mg
1 PO TID
8 days prior
90 / 42
Clonazepam 2mg
1 PO TID
8 days prior
90 / 77
levetiracetam 500 mg
1 PO BID
9 months prior
60 / 41
levetiracetam 500 mg
1 PO BID
6 months prior
60 / 27
Baclofen 10 mg
½ to 1 PO TID
8 days prior
90 / 46
Amlodipine Besylate 5 mg
1 QD
3 days prior
30 / 21
Desvenlafaxine
Unknown � no bottle found
Table 1: Medications Mother admitted to taking.
Medication
Dose Prescribed
Last Filled
Quantity filled / Quantity remaining
Ropirinole HCl 1mg
1PO QHS
8 days prior
90 / 75
Gabapentin 800 mg
1 PO BID
14 days prior
60 / 14
Milnacipran� 50 mg
1 PO BID
5 months prior
60 / 55
Carisoprodol 350 mg
1-2 PO BID
1 month and 15 days prior
360 /59
Morphine Sulfate ER 30 mg
1 PO Q8hr PRN
23 days prior
90 / 12
Caffeine Tablets 200 mg
N/A
N/A
16 / 6
Table 2: Additional Medications found at scene prescribed to Mother.
Within 24 hours of the death, an autopsy of the infant was performed and tissue samples including heart blood, liver, and brain were taken for toxicological analysis. Tissue samples were prepared as homogenates prior to analysis. Morphine was extracted from biological samples using solid phase extraction. Extracts were derivatized and analyzed for free morphine using full scan, gas chromatography mass spectrometry. The toxicology findings are listed in Table 3. The post-mortem examination revealed diffuse acute passive congestion with minor acute intra-alveolar hemorrhage bilaterally, diffuse multifocal subarachnoid congestion of the brain, and cerebral edema. Based on the history, autopsy and toxicological findings, it was determined that the death was the result of respiratory insufficiency secondary to synergistic drug intoxication.
Blood:
Analyte:
Levels:
Results:
Acetaminophen
Therapeutic: 10-20 mg/L
Toxic: 150 mg/L
Lethal: > 160 mg/L
19 mg/L
Diphenhydramine
Therapeutic: 0.025-0.112mg/L
Toxic: 5 mg/L
Lethal: > 8 mg/L
0.03 mg/L
Chlorpheniramine
Therapeutic: 0.01 - 0.017 mg/L
Lethal: 0.5 � 1.7 mg/L
0.06 mg/L
Methorphan
Therapeutic: 0.15 - 0.4 mg/L
Lethal: > 3mg/L
0.03 mg/L
Methorphinan
Therapeutic: 0.007-0.021 mg/L
Lethal: 0.8-2.7 mg/L
0.04 mg/L
Morphine
Therapeutic: 0.1 � 0.3 mg/L
Lethal: 0.1 � 4 mg/L
0.52 mg/L
Clonazepam
Therapeutic: 0.007 � 0.028 mg/L
Toxic: 0.46 mg/L
0.01 mg/L
Table 3: Toxicology report from blood, brain tissue and liver tissue samples of the infant.Blood:
Analyte:
Result:
Morphine
0.64 mg/Kg
Brain:
Analyte:
Result:
Morphine
0.56 mg/Kg
Lethal: 0.4 � 18 mg/Kg (average 3.0 mg/Kg)
The mother was charged with homicide by child abuse, involuntary manslaughter and unlawful conduct towards a child. She was tried and convicted of all three charges and subsequently sentenced to 20 years in prison, the maximum amount allowable by law for homicide by child abuse. This case has set precedence for future cases, as it was the first to ever prosecute a mother for killing her child by substances transferred through breast milk.
Discussion
Due to the rise in analgesic use among women and the potential for infant harm, a discussion on medication transfer into breast milk and infant response to medications is warranted. Milk formation is complex, however in order to understand the transportation of drugs into breast milk it is essential to have a basic understanding of how milk is produced. Breast physiology is markedly changed during pregnancy due to increases in estrogen and progesterone and in the post partum period by an elevation in prolactin [5-7]. With the increase in hormones, there is an increase in the size and number of alveolar units which are responsible for the production of milk [5,6]. The transfer of drug into breast milk can occur via passive diffusion, carrier mediated transport and paracellular pathway [6]. Immediately postpartum, drugs are able to diffuse into the alveolus through the paracellular pathway due to incomplete closure of junctions between the mammary alveolar cells [6]. After several days the junctions close and the paracellular pathway is impeded thereby facilitating passive diffusion and carrier-mediated transport more readily [6].
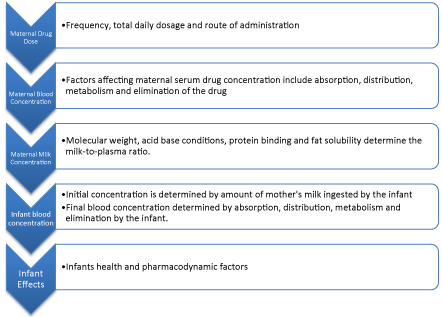
There are several factors that affect the transport of drug into the breast milk. These factors include size and molecular weight, lipid solubility, protein binding and acid base conditions [5,6]. (Figure 1) Molecules of lower weight will transfer more readily into the breast milk [5,6]. Human breast milk contains approximately 4% fat, which is significantly higher than blood fat concentration (<1%) [5,6]. Due to the heavier concentration of fat in the milk, lipid soluble drugs may diffuse and become trapped allowing for higher milk-to-plasma ratios.5,6 Protein binding has a two-fold mechanism. Drugs with high maternal protein binding will accumulate in the maternal plasma and are less likely to transfer into breast milk [5,6]. Alternatively drugs with low maternal protein binding may more readily diffuse into breast milk and bind to milk proteins thereby accumulating in the breast milk.5,6 Human milk has a lower pH (7) than serum (7.35-7.45) [5,6]. Due to milk’s lower pH, ion trapping can occur with weak bases allowing for higher concentrations in breast milk [5,6].
Figure 1: Drug Concentration Cascade from Mother to Infant.
Individual maternal pharmacodynamic factors can also play a large role in the concentration of drug available in the breast milk. Absorption, distribution, metabolism and elimination can vary from woman to woman depending on genetic, nutritional and health related factors. Pharmacodynamic factors in infants vary widely from week to week secondary to rapidly changing volume of distribution, growth patterns, organ maturation, and digestive abilities. Absorption in infants is generally slower than that of an adult [6]. Variables such as gastric pH and intestinal flora are responsible for changes in absorption [6]. Along with physical variables, the bioavailability of the drug administered will play a significant role in its absorption [7]. Body composition affects the distribution of drugs, and when comparing newborns, infants and adults, they all have vastly different fat compositions [6]. As aging occurs the percentage of fat in total body composition increases. Administration of a fat-soluble drug, such as morphine, to an individual with a lower fat concentration will enhance accumulation in the brain secondary to increased penetration of the blood-brain barrier [6,8]. This property could potentially lead to adverse CNS effects.6,8 Hepatic enzymes such as UGT2B7,which are responsible for morphine metabolism, are not fully active until several months of age. The glomerular filtration rate in full term newborns is approximately 25% of that in adults not reaching full potential until 3-5 months of age [6,9]. Due to decreases in hepatic metabolism and renal excretion, drugs such as morphine that form glucuronides are subject to longer half-lives in infants (3-14 hours) than their adult counterparts (2-3 hours). In this case only the free morphine was measured, therefore the contribution from the glucuronide metabolites is unknown.
The therapeutic, toxic, and lethal ranges reported in Table 3 represent a composite of ranges that have been reported in the literature. Although there is limited data concerning morphine exposure in neonates, Robieux et. al. report a case where a neonate (< 2 week) was exposed to morphine via breastmilk. The neonate developed clinically significant symptoms with a level of 0.004 mg/L (4 ng/ml) morphine [2]. A 10 month old boy who died following an exposure to ethylmorphine had postmortem levels of 0.054 mg/L (54 ng/ml) of ethylmorphine and 0.026 mg/L (26 ng/mL) morphine [10]. Additionally, there is a reported case where a 13 day-old neonate succumbed to a level of 70 ng/mL (0.07 mg/L) of morphine [11] and another report of an 8 year-old child who died as a result of an accidental oral ingestion of morphine sulfate with a resulting level of 0.128 mg/L (128 ng/mL), [12]. The range associated with compounds such as morphine also reflects variances due to tolerance. This range encompasses both the naÏve user (lower end of the range) and the opioid tolerant user (upper end of the range). Since the frequency and dose exposure of this infant to morphine is unknown, the level of tolerance cannot be determined. However, the level of morphine in the case presented in this paper represents a level that is well above the levels that have been reported to cause death in adults, children, and other infants.
One final concern in interpreting the levels of morphine is the phenomenon of post-mortem redistribution. The heart/femoral blood concentration ratio for morphine has been reported to be an average of 1.2 to 2.2 [13]. Again, the level of morphine in this case is well above the levels that have been associated with death from morphine intoxication even when considering any redistribution, and analysis of brain and liver tissue confirm that the infant was exposed to a lethal amount of morphine.
Conclusion
This was an unfortunate case of morphine toxicity that was exacerbated by the combination of other drugs with synergistic activity leading to CNS and respiratory depression. The bioavailability of morphine is 17-33% following oral administration. This may explain why few cases of toxicity have been reported. However in conjunction with decreased hepatic metabolism and renal excretion in the infant, morphine concentration could have accumulated. With the infant’s body composition and repeated exposure, CNS effects were markedly increased due to the diffusion across the blood-brain barrier. Additionally, the dose of morphine that this infant received likely represents a much larger dose than what is usually seen in infants who are breastfeeding from mothers prescribed opioids post-partum. In this case, the chronic misuse of extended release morphine by the breastfeeding mother led to the toxicity seen in this infant.
When considering analgesic treatment for a lactating mother one must weigh all potential risks to the infant. As always, using the lowest effective dose for a minimal amount of time is recommended as treatment with any medication in order to minimize the risk of toxicity to the infant.
References
- Prescription Painkiller Overdoses: A growing epidemic, especially among women. CDC Vital Signs. 2013.
- Robieux I, Koren G, Vandenbergh H, Schneiderman J. Morphine excretion in breast milk and resultant exposure of a nursing infant. J Toxicol Clin Toxicol. 1990; 28: 365-370.
- Bar-Oz B, Bulkowstein M, Benyamini L, Greenberg R, Soriano I, Zimmerman D, et al. Use of antibiotic and analgesic drugs during lactation. Drug Saf. 2003; 26: 925-935.
- American Academy of Pediatrics Committee on Drugs. Transfer of drugs and other chemicals into human milk. Pediatrics. 2001; 108: 776-789.
- D'Apolito K. Breastfeeding and substance abuse. Clin Obstet Gynecol. 2013; 56: 202-211.
- Hendrickson RG, McKeown NJ. Is maternal opioid use hazardous to breast-fed infants? Clin Toxicol (Phila). 2012; 50: 1-14.
- Borgelt LM, O’Connell MB, Smith JA, et al. Women’s Health Across the Lifespan: A harmacotherapeutic Approach. Bethesda, MD: American Society of Health-Systems Pharmacists. 2010: 362-364.
- Young M. Morphine in breast milk. Can Fam Physician. 2007; 53: 1005.
- Spigset O, Hägg S. Analgesics and breast-feeding: safety considerations. Paediatr Drugs. 2000; 2: 223-238.
- Helland A, Isaksen CV, Slørdal L. Death of a 10-month-old boy after exposure to ethylmorphine. J Forensic Sci. 2010; 55: 551-553.
- Koren G, Cairns J, Chitayat D, Gaedigk A, Leeder SJ. Pharmacogenetics of morphine poisoning in a breastfed neonate of a codeine-prescribed mother. Lancet. 2006; 368: 704.
- Poklis A, Edinboro LE, Wohler AS, Presswalla F, Barron D. Fatal morphine poisoning in a child due to accidental oral ingestion. Forensic Sci Int. 1995; 76: 55-59.
- Baselt R. Disposition of Toxic Drugs and Chemicals in Man. 10th edn. Biomedical Publications, Seal Beach, CA. 2014.