Research Article
Austin Alzheimers J Parkinsons Dis. 2014;1(2): 10.
CSF Tau Protein in Alzheimer’s Disease and other Neurological and Psychiatric Diseases
Rozenstein-Tsalkovich L1, Kahana E2, Shenker A2, Baitcher F1, Cohen OS3, Kahana-Merhavi S1, Chapman J3, Korczyn AD4, Aharon-Perez J5, Milo R2, Rosenberg A6, Treves T7, Wertman E8, Abramsky O1, Meiner Z1,9 and Rosenmann H1*
1Departments of Neurology, Hadassah Hebrew University Medical Center, Israel
2Department of Neurology, Barzilai Medical Center, Israel
3Department of Neurology, Sheba Medical Center, Israel
4Sieratzky Chair of Neurology, Tel Aviv University, Israel
5Department of Neurology, Rambam Medical Center, Israel
6Department of Neurology, Sorasky Medical Center, Israel
7Department of Neurology, Rabin Medical Center, Israel
8Neurology Services, Clalit HMO, Israel
9Physical Medicine and Rehabilitation, Hadassah Hebrew University Medical Center, Israel
*Corresponding author: Rosenmann H, Departments of Neurology, Hadassah Hebrew University Medical Center, Jerusalem 91120, Israel.
Received: September 08, 2014; Accepted: October 14, 2014; Published: October 15, 2014
Abstract
Total tau (T-tau) in the Cerebrospinal Fluid (CSF) is among the most reliable markers for the diagnosis of Alzheimer’s disease (AD). However, the high inter-center variation in biomarker concentrations points to the need for setting up specific diagnostic cut-offs for each population. We analyzed the level of T-tau in the CSF of 490 patients affected by AD or other neurological and psychiatric diseases, among the Israeli population. The T-tau levels were significantly higher in the AD group than in the other non-AD diseases, particularly in the other common dementia Fronto Temporal Dementia (FTD), as well as in psychiatric diseases. Receiver-Operating-Characteristic (ROC) analysis provided a ≥240 pg/ml cut-off for discrimination between AD and other non-AD diseases {sensitivity 68%, specificity 60%, negative-PV (predictive-value) 84.4%}, and psychiatric diseases (60%, 80%, positive-PV 93.4%), and from FTD (60%, 61.1%, positive-PV 85.8%), respectively. In spite of low PVs, T-tau levels in the AD patients were also higher than in vascular-dementia, Parkinson’s-disease, epilepsy, with similar trends relative to multiple-system-atrophy, dementia with Lewy-body-disease, autoimmune and other degenerative diseases, while comparable with metabolic and acute neurological diseases. Our previously reported ≥1,000 pg/ml cut-off for Creutzfeldt–Jakob Disease (CJD) diagnosis allows discrimination from AD (sensitivity 75%, specificity 92.7%, PPV 86.56% NPV 85.8%). An inverse correlation was noticed between CSF T-tau levels and Mini-Mental-State-Examination (MMSE) scores in AD and in VD.
Using ≥240 pg/ml as a cut-off we showed here that the T-tau level in CSF could be an indicator for differentiation of AD from psychiatric diseases and from FTD in our population. While also informative at ≥1000 pg/ml for CJD, the T-tau level was less informative for discrimination of AD from other neurological diseases. Combining the T-tau level in the CSF with other parameters (additional CSF markers, as well as genetic and clinical, including imaging parameters) may provide a stronger indication for AD.
Keywords: Neurofibrillary; Olivopontocerebellar; Neurological diseases; Encephalomyopathy
Introduction
There is accumulating evidence that the major neuropathological features characteristic of the Alzheimer’s disease (AD) brain - are generally reflected in the CSF. A higher amyloid plaque burden in the brain is reflected as lower amyloid beta (Aβ) levels in the CSF [1-4]. A higher CSF tau protein level represents axonal injury and cell death {particularly reflected by T (total)-tau}, as well as a higher neurofibrillary tangle burden {represented by both T- and phosphorylated (P)-tau} [4-6]; all of these markers are considered to be the “core CSF AD markers”. Conflicting results have been reported on the association of the CSF Aβ levels with the cognitive /clinical status [7-11]. Yet, there is more evidence for an inverse association between the tau protein and the cognitive status [7,9,10,12-15].
Many studies have shown the diagnostic value of both Aβ42 and the tau protein in the CSF for discrimination between AD patients and non-demented subjects [16-33]. As for the diagnostic value of CSF markers in discrimination between AD and other dementias, there are conflicting results regarding CSF Aβ42, specifically for differentiation between AD and dementia with Lewy-Body Disease (DLB) and Vascular Dementia (VD) [34,35]; yet, there are more consistent results showing a lower level of the tau protein in the non- AD dementias relative to the AD dementia (yet with overlap) [23,35- 41].
CSF studies in AD patients and non-demented controls from different centers report different biomarker concentrations, reference ranges and diagnostic cut-offs; in some studies, CSF marker levels in AD patients even exceeded (in the case of amyloid) the levels in controls of other studies [42,43]. Several multi-center studies have been conducted {such as the DESCRIPA study by Visser et al. [44], the ADNI study by Shaw et al. [28], the European-ADNI (E-ADNI) by Buerger K et al. [45] and the multi-center study of Mattsson et al. [19]}, showing lower diagnostic accuracies than those of homogenous mono-center studies, presumably related to inter-center variations. The very wide range of cut-offs of the AD CSF markers among centers (such as for tau, ranging from 195 pg/ml to 450 pg/ml [43]), points to the need for setting up specific diagnostic cut-offs to be used in each center, and validate the diagnostic accuracy of the CSF markers in each of the studied populations.
Since tangle pathology is in good correlation with clinical dementia [46-48] and since T-tau in the CSF is associated with tangle-pathology as well as with neuronal loss, the T-tau is considered to be the most commonly used diagnostic tool in many centers. We report here the levels of the T- tau in the CSF of AD patients and of other dementias as well as of patients with other neurological or psychiatric diseases among the Israeli population referred to our laboratory, in the years 1999-2007, which is a national referral center for CSF analysis of neurodegenerative markers.
Methods
Patients
The study population included a total of 490 patients affected by:
AD: AD (n=124), mixed dementia (AD + vascular dementia (n= 14)
Non-AD dementias: VD (n=114), Fronto Tempotal Dementia (FTD) (n=36) {including tauopathies (Pick’s disease, cortico basal degeneration, progressive supranuclear pulsy (n=14)}, DLB (n=11); Parkinson’s disease with dementia (PDD) (n=6);
Acute neurological diseases: encephalitis (n=13), cerebrovascular accident (CVA) (n=17), intoxication (alcoholism, drug addiction) (n=16), brain tumor (n=5), and paraneoplastic syndromes (n=6);
Other neurological diseases: Degenerative diseases {multiple system atrophy (MSA) (n=14), PD (n=8), amyotrophic lateral sclerosis (ALS) (n=5), Huntington’s disease (HD) (n=2), spinocerebellar ataxia (n=3), cerebellar atrophy (n=2), cerebellar degeneration (n=3), and others (olivopontocerebellar degeneration cerebellar ataxia, pontocerebellar atrophy, cerebellar atrophy, Shy Drager, neuro-axonal sclerotic encephalopaty, CNS degeneration, prog bulbar palsy) (n=8)}, epilepsy (n=16), metabolic diseases {hepatic/pancreatic encephalopathies, mitochondrial encephalomyopathy, lactic acidosis and stroke (MELAS)} (n=14), autoimmune diseases (vasculitis, multiple sclerosis, Bechet) (n=19), mild cognitive impairment (MCI) (n=2), and others {dystonia, polyneuropathy, ataxia telangiectasia (ATM)} (n=4);
Psychiatric diseases: (depression, psuedodementia) (n=28). Demographic data included gender, age at onset and the time point of lumbar-puncture (LP). When available, scores of the Mini Mental State Examination (MMSE) were reported. In a small part of the study population, the number of years of education or the type of occupation (divided into 3 degrees of challenging level) was available. The CSF samples of these patients were sent to our laboratory as a routine work-up for dementia. The diagnosis, made according to medical data and follow-up using established internationally agreed criteria, was based on medical records collected from the different medical centers. The results of the CSF T-tau analysis were not included in the diagnostic criteria. In the AD group, although the diagnosis was based on international clinical criteria (clinically probable AD) - it may be assumed that the mixed pathology of AD with VD is quite common. For comparison, we also included the group affected with Creutzfeldt-Jacob disease (CJD) (n=76) patients, previously reported by us [24]. Clinical data from the patients is registered in The Israeli National Registry Database of CJD overseen by one of us (E.K) according to Israeli’s laws. Maintenance of participants’ anonymity was strictly kept during database analyses. We summarized here the results of CSF samples tested by us in 1999-2007. Clinical follow-up was performed during at least three years allowing confirmation of diagnosis.
CSF analysis
CSF samples were tested for T-Tau protein at our Laboratory of Neurogenetics, a national referral center of the Department of Neurology, Hadassah Hebrew University Medical Center. This laboratory is a national referral center for genetic and CSF analysis for neurodegenerative diseases in Israel. CSF samples were sent to us (usually within 24 to 48 hours following LP stored in plastic tubes). Tau concentrations were measured by ELISA (Innotest hTau-Ag, Innogenetics, Ghent, Belgium).
Statistical analysis
To compare continuous variables among all of the patient groups, the non-parametric Kruskal-Wallis Test was used. To compare continuous variables between two groups, the two sample t-test and the non-parametric Mann-Whitney test were applied. For multiple pair-wise analyses, the Bonferroni correction of the significance level was used, as follows: p<0.0035 for both age and tau levels; p=0.01 for MMSE scores. Comparisons were performed only in groups with n≥8 patients. The comparison of qualitative variables between two groups was carried out using the Chi-Square test. To assess linear associations between two continuous variables, the Pearson correlation coefficient was calculated. Analysis of covariance (ANCOVA) was used for simultaneously assessing the effect of several independent variables (both continuous and qualitative) on a dependent continuous variable. Sensitivity, specificity and positive and negative predicting values were calculated. Receiver Operating Characteristic curve (ROC) analysis was performed in order to find the optimal cut-off point of CSF tau for differentiation between AD patients and other patient groups {with a relatively high number of cases (n≥28): VD, FTD, psychiatric diseases, CJD}. All of the applied tests were two-tailed, and a p-value of 5% or less was considered statistically significant.
Results
Demographics
The demographic data and the MMSE scores of the study population are summarized in the Table. Comparisons were performed for groups containing at least eight subjects. The mean age of disease onset in the AD patients was 60.02±8.68 (M ± SD), and the age at lumbar puncture (LP) was 61.95±8.32. Comparing these ages with those of other diseases (two sample t-test) revealed a significantly different age (higher) only in the VD group (67.06±10.37, and 67.84±10.41, respectively, p<0.0001 for each). Some differences in ages relative to the AD group were also noticed in other groups: higher in mixed dementia (67.17±8.85, 68.74±8.81, respectively), while lower in the encephalitis group (43.52±23.11, 45.99±27.52), CVA (onset: 67.09±13.31), autoimmune diseases (48.67±15.45, and 50.75±15.18) and psychiatric patients (54.12±12.97, 55.41±12.73); however, Bonferroni correction excluded significant differences. No significant difference in the male to female ratios (M/F) was detected between the AD patients (60/64) and the other groups. Some differences in mean MMSE scores were noticed (two sample t-test) (18.17±6.49 in the AD group, while 21.53±5.89 and 23.38±5.78 was noted in the FTD and the psychiatric group, respectively), yet Bonferroni correction excluded a significant difference.
Group
No. Patients
M/F
Age at onset (ys)
Mean±SDAge at LP (ys)
Mean±SDMMSE
Mean±SD (N)Tau (pg/ml)
Mean±SDTau (pg/ml)
Median (min-max)AD:
AD
124
60/64
60.02±8.68
61.95±8.32
18.17±6.43(99)
434.64±315.43
401.5 (1-1479)
Mixed dementia
14
7/7
67.17±8.85^
68.74±8.81^
20.62±2.50(8)^
473.86±448.63
229.0 (71-1222)
Non-AD dementias:
�������������� VD
114
54/60
67.06±10.37l*a
67.84±10.41*a
18.98±7.64(43)
300.47±306.38*b
182.5 (20-1602)*a
FTD
36
19/17
57.93±12.01
59.58±12.01
21.53±5.89(21)^
266.16±275.83*c
178.0 (15-1125) *b
DLB
11
5/6
63.06±8.13
64.95±7.81
20±4.24 (2)
364.84±444.88^
157.0 (52-1295)
PDD
6
6/0
68.33±7.53
71.06±8.14
18±4.24 (2)
503.67±506.24
290.0 (75-1343)
Acute neurological diseases:
Encephalitis
13
5/8
43.52±23.11^
45.99±27.52^
28(1)
567.25±491.10
349.0 (64-1310)
CVA
17
12/5
67.09±13.31^
67.32±13.41
20.4±12.35(5)
547.41±440.37
542.0 (14-1253)
Intoxication
16
11/5
58.73±17.86
59.17±17.93
7±7.7(4)
414.25±471.82
223.0 (41-1782)
Brain tumor
5
4/1
65.75±12.33
65.95±12.41
30(1)
402.40±245.77
333.0 (131-732)
Paraneoplastic
6
3/3
54.58±14.36
54.78±24.39
10(1)
518.77±433.87
353.0 (165-1234)
Other neurological diseases:
Degenerative:
MSA
14
8/6
60.83±11.59
63.50±10.42
27.8±2.67(5)
244.07±167.31*b
193.5 (86-629)^
PD
8
4/4
69.35±7.94
54.40±10.02
25± 3.58(6
122.78±79*a
107.0 (19-261) *b
ALS
5
1/4
59.69±11.36
60.95±11.12
NA
288.20±186.81
255 (19-497)
HD
2
2/0
47.24±14.42
47.8±13.7
25.5±3.53(2)
153.50±26.16
153.5 (135-172)
Spinocerebellar
3
0/3
60±0.41
61.05±0.46
24(1)
439.00±235.56
575.0 (167-575)
Cerebellar atrophy
2
2/0
56.3±2.2
57.86±2.38
21(1)
249.05±248.83
249.05(73-425)
Cerebellar degeneration
3
2/1
46.91±17.89
49.29±22.55
26(1)
208.33±90.85
251.0(104-270)
Others
8
5/3
57.39±8.32
59.27±10.13
20.5±3.53(2)
249.88±386.91
153.0 (15-1184)^
Epilepsy
16
8/8
53.38±23.36
54.88±22.40
21.34±8.94(8)
148.64±101.54*a
140.5 (29-363)*a
Metabolic diseases
14
7/7
61.47±15.57
62.09±14.63
18.4±9.18(5)
421.48±354.93
364.5 (43-1339)
Autoimmune diseases
19
9/10
48.67±15.45^
50.75±15.18^
26.2±3.56(5)
348.55±440.25
135 (30-1458) ^
MCI
2
1/1
57.68±0.72
57.76±0.60
28(1)
339.50±249.61
339.5 (163-516)
Others
4
2/2
55.21±13.59
55.84±14.0
30(1)
116.75±42.6
126.0 (57-158)
(dystonia, polyneuropathy,ATM)
Psychiatric diseases:
28
13/15
54.12±12.97^
55.41±12.73^
23.38±5.78(13)^
172.34 ± 135.86*a
140.5 (18-574 *a
Table 1: Demographic data, MMSE-scores, and CSF- tau values of the study groups.
T-tau levels in the CSF
The mean and the median values of T-tau in the CSF of the different patient groups are presented in the Table and in Figure 1. There was a statistical significant difference between the tau levels in the different groups (p<0.0001, Kruskal-Wallis Test). Comparing the mean values of tau in the CSF of the AD group (434.64±315.43 pg/ml) with those of the other patient groups (two sample t-test) revealed : lower levels in the non-AD group (total patients with any non-AD disease, 314.66±333.83, p=0.0004), VD (300.47±306.38, p=0.001), FTD {266.16±175.83, p=0.002 (and among them the tauopathy cases 197.70±274.85, p=0.007)}, MSA (244.07±167.31, p=0.001), PD (122.76±79, p<0.0001), epilepsy (148.64±101.64, p<0.0001), and psychiatric diseases (172.34±135.86, p<0.001); lower levels in DLB were detected (364.84±444.88, however Bonferroni correction excluded a significant difference), while comparable levels were noted with the acute neurological diseases (encephalitis, CVA, intoxication), as well as with metabolic and autoimmune diseases. Similar results were obtained when we also included for comparison in the AD group patients with the diagnosis of mixed dementia (10% of the AD cases) (mean value of the wide AD group 438.61±327.36, comparable with that of the AD).
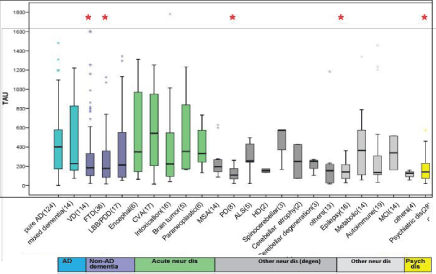
Figure 1: Box Plot of tau in the CSF of AD patients and of non-AD dementias, acute neurological diseases, other neurological diseases (degenerative and others) and psychiatric diseases (CJD patients not included). Results presented as medians, 25% and 75% quartiles, as well as outliers. Tau levels are significantly higher in AD patients than in VD (p<0.0001), FTD (p=0.001), PD (p=0.001), epilepsy (p<0.0001), and psychiatric diseases (p<0.00001).
Similar differences were noted when comparing the median values of tau in the AD (401.5 pg/ml) with those in the other groups (Mann- Whitney Test) (Figure 1): significantly lower level in the VD (182.5, p<0.0000), FTD {178.0, p=0.001 (and among them the tauopathy cases 98.0, p=0.0001)}, PD (107.0, p=0.0001), epilepsy (140.5, p=0.0001), and psychiatric diseases (140.5, p=0.0001). Lower median levels of tau relative to AD patients were also detected in MSA (193.5), other neurological degenerative diseases (153.0) and autoimmune diseases (135.0); however Bonferroni correction excluded a significant difference. Comparable levels of tau were observed between the AD patients and those with DLB, acute neurological diseases, metabolic diseases. Similar results were obtained when we also included in the AD group patients with mixed dementia (median value of the wide AD group 374 pg/ml, comparable with the 401.5 pg/ml of the AD).
As presented by us in our previous publication [24], the level of tau in the CSF of CJD patients was high, mean value (combined for sporadic and genetic): 1175.46 ± 519.84 pg/ml, median: 1249.00 (49- 3495), significantly higher than AD and any other diseases of the CNS (p<0.0001), with an age onset of 62.78± 10.37, significantly higher than in AD (60.02±8.68) (p=0.0014).
Similarly to the higher level of tau in AD relative to psychiatric diseases, the level was also relatively higher in the non-AD dementias relative to the psychiatric diseases (p=0.0003), particularly noticed in the VD group vs (p=0.001).
Correlation analysis of age of onset and sex with tau levels in the CSF
There was no significant correlation between age at onset and the tau level (Pearson correlation coefficient) (r=0.059, p=0.116) for the entire sample, as well as in the different subgroups of diseases, however, there was a borderline significance for an inverse correlation of tau with age in the patients with acute neurological diseases (r=- 0.258, p=0.08). Testing the correlation with sex revealed higher levels of tau in women than in men, again with a borderline significance (p=0.08). When applying the ANCOVA model to the tau levels, with disease, age and sex entered into the model as explanatory variables, only disease and sex had a significant effect (p<0.0001, p=0.005) whereas age was not significant (p=0.75). The ANCOVA model which included only subgroup and age had an R2 of 0.42 compared with an R2 of 0.43 when sex was included in the model as well. This finding suggests that the contribution of sex to the model is very small. Therefore it is obvious that disease effect is highly significant when correcting for age and sex.
Correlation analysis of MMSE scores with tau level in the CSF
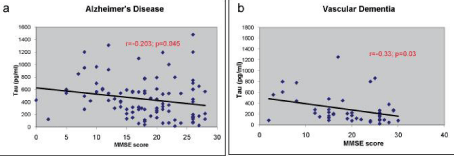
As presented in Figure 2, an inverse correlation was detected between MMSE scores and the tau levels in the CSF of the AD patients (Pearson correlation coefficient) (r=-0.203, p=0.045), with lower MMSE values being associated with higher tau levels. Such an inverse correlation was detected also in the VD group (r= - 0.33, p=0.03). A similar trend was noticed in the acute neurological diseases, however it did not reach a statistical significance (r=-0.33, p=0.3). The tau levels and the ages (at onset or LP) did not differ between patients with or without MMSE scores available.
Figure 2: Correlation between tau levels in CSF and MMSE scores. Significant inverse correlation in AD (p=0.045) (a); and in VD (p=0.03) (b).
Characterizing AD patients with low tau levels and other patients with high tau
Among the AD patients, 39 had low tau levels (<240 pg/ml). In order to characterize this population, we compared the demographic data with those of the AD patients having a high tau level (n=85, ≥240 pg/ml) in the CSF. While no difference was noticed at age of onset or of LP, sex, as well as in years of education {11.85±4.5 (n=14) and 11.48±3.73 (n=29), respectively}, and type of occupation {among 1-3 degrees of challenging occupations, 1.5±0.92, for each (n=8/group)}, we did notice a higher MMSE score in the group with a low tau as compared with that in the high tau group {21.45±5.63 (n=29) and 16.81±6.27 (n=70), respectively, p=0.00065}. This is in accord with the above mentioned significant inverse correlation we detected between tau levels and MMSE scores in the AD group.
We were also interested in characterizing the subjects who had high tau levels although not affected by dementia or other neurological diseases (such as acute or degenerative ones). For this purpose, we selected the psychiatric patients with high tau levels in the CSF. While no difference was detected at the age of onset and of LP as well as in sex, relative to the psychiatric patients with low tau levels (n=22), there was some borderline significance (p=0.057) of lower MMSE scores in the cases with high tau [19.5± 3.87 (n=4) and 25.44± 5.72 (n=8)]. Data regarding years of education and mode of occupation were not informative.
Measures of Diagnostic Accuracy (sensitivity, specificity and predictive values, ROC)
We next tested the diagnostic potential of the significant higher levels of CSF tau in AD patients relative to others. ROC analysis was performed in order to find the optimal cut-off point of CSF tau for differentiation between AD patients and the other patient groups (I: the height of p value and size of graph is not the same in the a and b parts of figure. II: Should be the same a, b, c, d in figure and legend (and not a,b,c,d)).
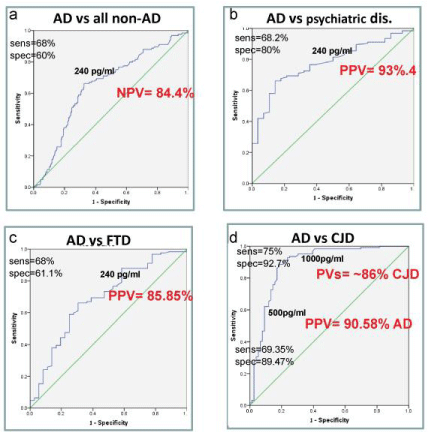
Discrimination between AD and non-AD diseases: When performing the ROC analysis, in order to test whether the level of tau in the CSF provides adequate discrimination between AD and any other neurological/psychiatric diseases (not including CJD patients), it revealed an AUC of 0.647, which, although statistically significant (p=0.000), provides only limited discrimination (while not reaching the ≥0.7 value representing an acceptable discrimination, being higher than 0.5, an AUC provides no discrimination at all) (Figure 3a). Using 240 pg/ml as a cut-off provides 68% sensitivity, 60% specificity, with PPV of 37.8%, while a NPV of 84.4%, suggesting that a <240 pg/ml tau level gives a quite high (84.8%) prediction of non-AD disease, while ≥240 pg/ml is not informative in a Differential Diagnosis (DD) of AD from non-AD diseases, and can be only considered as suspected for AD (higher cut-offs have also low PPV values).
a) ROC curves of tau in the CSF. At a cutoff of 240 pg/ml: sensitivity 68%, specificity 60% and NPV 84.4% to distinguish between AD and non-AD diseases.
(b) Sensitivity 68.2%, specificity 80% and PPV 93.4% to distinguish between AD and psychiatric diseases.
(c) Sensitivity 68%, specificity 61.1% and PPV 85.85% to distinguish between AD and FTD.
(d) At a cut-off of 1000 pg/m, sensitivity 75%, specificity 92.7% with PVs ~86% to distinguish between CJD and AD, while at a cut-off of 500 pg/ml: sensitivity 69.35%, specificity 89.47% with PPV 90.58%.
Figure 3:a) ROC curves of tau in the CSF. At a cutoff of 240 pg/ml: sensitivity 68%, specificity 60% and NPV 84.4% to distinguish between AD and non-AD diseases.
(b) Sensitivity 68.2%, specificity 80% and PPV 93.4% to distinguish between AD and psychiatric diseases.
(c) Sensitivity 68%, specificity 61.1% and PPV 85.85% to distinguish between AD and FTD.
(d) At a cut-off of 1000 pg/m, sensitivity 75%, specificity 92.7% with PVs ~86% to distinguish between CJD and AD, while at a cut-off of 500 pg/ml: sensitivity 69.35%, specificity 89.47% with PPV 90.58%.
In the clinical setting, there is a greater need for the diagnosis of AD as DD in diseases like psychiatric ones or other dementias. This was calculated here as follows:
Discrimination between AD and psychiatric diseases: ROC AUC of 0.775 (p=0.000) provides an acceptable discrimination between AD patients and psychiatric diseases (pseudodementia, depression) (Figure 3b). Using the 240 pg/ml cut-off provides a sensitivity of 68.2% with a specificity of 80%, and PPV of 93.4% while NPP of 36%, suggesting that while a ≥240 pg/ml tau level gives a high (93.4%) prediction of AD disease, a level below this cut-off is not informative in DD of AD from psychiatric diseases (lower cut-offs have also low NPP values).
Discrimination between AD and non-AD dementias: ROC AUC of 0.654, although statistically significant (p=0.000), it does not point to an adequate discrimination between the AD group and non- AD dementias, which included VD, FTD, and DLB and PDD.
The ROC curve for discrimination between AD specifically and VD patients provided an AUC of 0.651 (p<0.0001). At the 240 pg/ ml cut-off, a sensitivity of 68.2% and a specificity of 58.7% with some limited predictive values of PPV 64.39% and NPP of 63.2% (lower cut-offs did not improve the diagnostic accuracy).
More informative was the ROC curve for discrimination between AD and FTD patients: the AUC was 0.688 (p=0.001) (Figure 3c). At the 240 pg/ml cut-off, a sensitivity of 68% and a specificity of 61.1% with a PPV of 85.85% and a NPP of 36.05% were provided, suggesting that a ≥240 pg/ml tau provides a good prediction of AD as DD from FTD in our population. A cut-off of 50 pg/ml gives a better NPV of 63.63% (with a sensitivity of 96.8% while a specificity of only 21%, with a PPV of 80.53%), indicative of the non-AD dementia, FTD. This data suggest that in the DD of FTD from AD, a tau level of ≥240 pg/ ml has a high PV (85.85%) for AD, while <50 pg/ml has a high PV (80.53%) for FTD.
Discrimination between AD and CJD: Using our previously reported data of CSF T-tau levels in CJD patients and other neurological controls, using the 1,000 pg/ml cut-off [46], we performed here a ROC analysis for discrimination between AD and CJD. An AUC of 0.886 (p<0.0001) was noticed, a value which falls in the range of excellent discrimination (0.8-0.9) (Figure 3d).
Sensitivity was 75% and specificity 92.7%, PPV 86.56% of being CJD, with a NPV of 85.8% for not being CJD, but being AD when tau is lower than 1,000 pg/ml. T-tau levels of <500 pg/ml revealed a higher PV (90.58%) of AD (sensitivity 69.35%, specificity 89.47%, while NPV only 59.13% of being CJD when >500pg/ml).
Discussion
In this study, we report that the levels of tau in the CSF are significantly higher in AD patients than in patients with other diseases, particularly psychiatric diseases and FTD, VD, epilepsy and PD, with some trend of lower levels in DLB, autoimmune and other degenerative diseases, while comparable with metabolic and acute neurological diseases. These differences were also evident when we included with the AD patients those clinically diagnosed of mixed dementia (AD and VD). The diagnoses were based on clinical criteria, which are established as highly accurate exceeding 85% (as compared to brain pathology in autopsy) [49].
In an effort to use these results as biomarkers for diagnostic purposes of AD, we established a cut-off of 240 pg/ml, to be used as follows:
- <240 pg/ml gives 84.4% PV of not having AD as DD from other non-AD diseases (while higher levels being not really informative, to only be considered as suspected for AD).
- ≥240 pg/ml gives 93.4% PV of having AD as DD from psychiatric diseases (<240 can only be considered as suspected for a psychiatric disease).
- ≥240 pg/ml gives 85.85% of having AD as DD from FTD (< 50 pg/ml gives an indication (80.53%) for having FTD.
This 240 cut-off appears to provide a quite adequate predictive value for AD, particularly useful as DD from psychiatric diseases, and of the specific diagnostic relevance in the clinic for discrimination between AD and another dementia, the FTD.
As for discrimination between AD and VD, although tau levels were lower in VD, they provide a less adequate tool (≥240: PVs of 63-64%), which may be related to the overlap in the symptomatology of these two clinical conditions which may have caused difficulties in the DD of these patients.
Also informative were the tau levels when we used them for discrimination between AD and CJD: ≥1,000 pg/ml had a high PV (86.56%) for CJD, and <1,000 pg/ml had 85.8% for AD (and <500 pg/ml had even PV 90.58% for AD). These results are in agreement with other studies (reviewed in [50]). Our current results comparing CJD patients to AD showed somewhat different PV values to those reported by us previously when comparing CJD group to all other neurological diseases (PPV 67.3%, NPV 94.2%) [51], probably since the other neurological disease groups included also patients with high T-tau levels, such as acute neurological diseases.
Other diseases may have a high false positive rate of CSF T-tau test, however they are usually distinguished clinically from dementias, particularly acute neurological diseases (such as encephalitis, CVA and even tumors), autoimmune- and metabolic- diseases.
Our results showing significant higher tau levels in AD patients relative to psychiatric diseases are in accord with other studies showing that tau levels in the CSF of Major Depressive Disorder (MDD) and of healthy controls are significantly lower than in AD patients [52] and that the levels are similar in MDD patients and cognitive intact controls [53]. Also among MCI patients, the tau levels do not change when symptoms of depression, agitation, irritability or apathy are evident [54]. Our sensitivity, specificity and PPV values for differentiation between AD and psychiatric diseases using the tau levels in CSF (68%, 80%, 93.4%, respectively) are similar to the values reported for discrimination between AD patients and cognitively intact individuals (sensitivity: 57%-85%; specificity: PPV: 85.7%-90.7%) [21,24,28,41,55].
Also, our results showing significant higher T-tau levels in AD patients relative to the non-AD dementia FTD are in accord with other studies ([23,41,55-57] and reviewed in [58,59]). There are wide variations in the sensitivity (36%-92%) and specificity (25%-89%) for discrimination between FTD and AD as reported by various centers (reviewed in [50]), with our values of 68% and 61% falling within these ranges. Our quite high 86% PPV for the diagnosis of AD vs FTD is close to those of other populations (87% [55] and 94.7% [21]). There are some controversies with regard to the tau level in FTD vs non-demented controls (elevated in FTD [23,56]; normal values [57,60,61]); in our population, comparable tau levels were detected in FTD and psychiatric cases.
Also, our results showing significant higher T-tau levels in AD relative to VD patients are in accord with other reports [41,62]. Our sensitivity and specificity values (68.2%, 58.7%) fall within the wide ranges (14-100% and 53-100%, respectively), reported in other populations ([41] and reviewed in [50]).
Regarding tau levels in DLB, there are controversies as to whether T-tau levels are lower than [12,35,62,63] or comparable with [64,65] those in AD patients, and also whether higher [63,66,67] or comparable [68-70] levels with those in non-demented controls. Our results showing only some non-significant trend of lower tau median value in the DLB cases relative to AD, while comparable in DLB with psychiatric diseases, they are in line with the published studies.
Importantly, there is a wide variability in the T-tau levels (and in the CSF AD biomarkers in general) among the centers, such as in AD patients (mean value 184-960 pg/ml), VD (88-708 pg/ml), FTD (55-575 pg/ml), DLB/PDD (138-508 pg/ml) (reviewed in [50]) and psychiatric diseases (273±152 pg/ml [53] and 169 pg/ml [52]). Our results of 434.64 pg/ml, 300.47 pg/ml, 266.16 pg/ml, 413.84 pg/ml and 172 pg/ml, respectively, fall within these ranges. Subsequently, there is a wide range of cut-off values for diagnostic use among the centers as reported by Hort [43], ranging from 195 pg/ml to 450 pg/ml, with our cut-off of 240 pg/ml tau protein falling within these values, and similar to the following suggested values: 234 pg/ml [55], 275 pg/ml [23].
The inter-center different concentrations, and diagnostic cut-offs of CSF biomarkers in general and of T-tau in particular, point to the need for setting up specific diagnostic cut-offs to be used in each center, and validate the diagnostic accuracy of the CSF markers in each studied population. The higher diagnostic accuracy which have been reported in a well defined homogeneous population than in a multi-center heterogeneous population, demonstrates the potency of CSF biomarkers to identify pathological processes in AD when a stringent analytical protocol is used as well as a defined population [14]. Moreover, taking into account the relative differences between patients and controls in the different studies being generally consistent, further strengthen the diagnostic value of the CSF AD biomarkers. However, the inter-center variations make it complicated and even misleading to compare CSF biomarker levels between centers and studies. Possible reasons for such inter-center variations may be: subject selection, CSF-handling, -obtaining, and -storing, (such as type of test tubes, freeze/thaw procedures, plasma contamination, etc.) and analytical factors {different immunoassays (ELISA or multiplex techniques), batch to batch variations, and different reagents} ([45,71] and reviewed by us [72]). In addition to the setting up of diagnostic cut-offs by each center, attempts are made to overcome these inter-center variations, by the establishment of the internationally quality control program for CSF AD biomarkers run by the Alzheimer’s Association and administered from the Clinical Neurochemistry Laboratory in Molndal. Participating laboratories, including us, receive CSF samples for analysis with recommended guidelines for LP, and sample handling and storage [73,74].
One major factor affecting the inter-center variations in CSF tau levels and cut-offs is the difference in the study population, such as whether the diagnosis is autopsy confirmed or only clinically made, whether the patients present classical AD symptoms or rather uncharacteristic symptoms (which may be the direct reason for being applied to CSF tau analysis), whether in certain centers the CSF analysis is a routine work up of dementia or not (like in patients living in elderly citizens’ homes and who are less referred to LP procedure in medical centers), etc. The CSF samples tested by us for tau in this study were sent to our lab as part of a work up of patients affected by dementia. Indeed, diagnoses were not confirmed by autopsy, but rather, by the long clinical follow-up of at least three years which allowed us to have a relatively accurate diagnosis. However, since our lab is also a referral center for the analysis of CJD (both genetic analysis and CSF tau analysis), we assume that at least part of the patients were referred for tau analysis as DD of CJD, possibly pointing that this population may include rapid progressive dementia/ AD cases. Also since the tau analysis is not a routine test for the AD diagnosis (at least part of the medical centers referring to our lab), we assume that the AD cases that were sent to us were more atypical cases of AD, rather than of classical AD. The relative early age at onset of the AD patients in our population (mean value 60.02y) may support this notion. We may speculate that more classical AD cases in our population may have some lower tau levels in the CSF than atypical and particularly the rapid progressive ones, as was reported by others for higher tau levels in atypical [75] and in rapid progressive cases [15,76,77].
We noticed an inverse correlation between the levels of tau in the CSF and the MMSE scores in the AD patients. As the presence of tau in the CSF represents the tangle pathology with a positive correlation between tau in CSF and the tangle burden [4] and as the tangle pathology is the best correlate with clinical dementia [46-48] - the correlation of tau with the MMSE scores is reasonable and quite to be expected. Although the MMSE evaluation and the LP were not performed necessarily at the same time point - the correlation was still evident, probably since the tau levels in the CSF are relatively constant along AD progression [78-81]. Other studies have also shown a correlation of MMSE (or other clinical, more specific, tools for evaluating dementia) with tau in CSF [7,9,10,12-15,82], as well as with p-tau, a marker which may be even more specific for the tangle pathology [6]. Actually the presence of T-tau in the CSF is also a marker for neuronal damage or death, as well as of glial cells which also express the tau protein [83,84]. It is well accepted that once neurons are injured, their content is leaked into the CSF, and therefore the neuronal (and glial) intracellular protein is detected in the CSF. This is the case in neurodegenerative diseases like AD or VD, in acute neurological diseases (like encephalitis and CVA), after brain injury, and particularly in the severe neurological condition in CJD. This may explain also the inverse correlation of tau with MMSE detected here in the VD, and a similar trend also in the acute neurological diseases.
The origin of the tau in the CSF may not only be the neuronal death and the intracellular tangle pathology, it may also be attributed to the extracellular tau secreted by the neurons [85-87], a process taking place in the propagation of the tangle-pathology in a prion-like manner in AD, in which extracellular tangles (“ghost tangles”) are evident [88]. The contribution of the secretion of tau and its extracellular accumulation may vary in the different neurodegenerative diseases with tangle pathology, as presented in cultured cells, with tau isoforms and mutations altering extracellular tau levels, in a way that cells expressing FTD-associated tau mutations produce significantly less extracellular T-tau than cells expressing wild-type tau [89]. These different tau metabolism systems may explain, at least partially, the different tau levels of the CSF in different diseases with tau-pathology, particularly the lower tau level of the CSF in primary tauopathies (FTDP, Pick’s disease, etc, included in our FTD patient group) which are associated with mutations in tau - relative to AD, a secondary tauopathy (the tangle formation are secondary to the amyloid pathology) and is not linked to any mutation in tau. Another explanation for the lower tau level of the CSF in tauopathies, and in FTD in general, is the reduced levels of soluble brain tau in the absence of insoluble tau or fibrillary tau inclusions described in dementia lacking distinctive histopathology (DLDH), the most common pathological variant of FTD, considered as a (“tau-less”) tauopathy [90].
Of interest were the AD patients with normal/low tau in the CSF (<240), and psychiatric patients with high tau level (≥240). In both patient groups, the age and sex did not differ from those in the AD patients with high tau and psychiatric patients with low tau. It was only the MMSE score that differed, being higher in the AD cases with low tau relative to cases with high tau, and being lower in psychiatric patients with high tau relative to low tau. This is in accord with the above mentioned inverse correlation of the MMSE with tau. It is possible that the psychiatric patients with a high level of T-tau are actually affected with presymptomatic AD. Nevertheless, it can be hypothesized that the levels of amyloid and of p-tau were abnormal (low amyloid, high p-tau) in the low tau AD patients, as well as normal (high amyloid, low p-tau) in the high tau psychiatric patients, and that the ratios of amyloid and p-tau with the tau will define these “uncharacteristic cases” as “characteristic” AD and non- AD cases, accordingly. Analyzing these additional biomarkers, and particularly their ratio may also allow for a better diagnosis of AD and its discrimination from other neurodegenerative and dementia diseases, as is suggested in other studies [34,91,92]. In addition, these markers, in combination with other parameters (age, MMSE, APOE status, PET imaging or MRI [93,94]), can be more predictive of AD-like pathology.
Conclusion
Using ≥240 pg/ml as a cut-off we showed here that the T-tau level in CSF could be an indicator for differentiation of AD from psychiatric diseases and from FTD in our population. While also informative at ≥1000 pg/ml for CJD, the T-tau level was less informative for discrimination of AD from other neurological diseases. Combining the T-tau level in the CSF with other parameters (additional CSF markers, as well as genetic and clinical, including imaging parameters) may provide a stronger indication for AD.
Acknowledgment
We thank Tali Bdolach-Avram from the Statistics Unit, School of Medicine – Hadassah Hebrew University, for statistic analysis.
References
- Fagan AM, Mintun MA, Mach RH, Lee SY, Dence CS, Shah AR, et al. Inverse relation between in vivo amyloid imaging load and cerebrospinal fluid Abeta42 in humans. Ann Neurol. 2006; 59: 512-519.
- Strozyk D, Blennow K, White LR, Launer LJ. CSF Abeta 42 levels correlate with amyloid-neuropathology in a population-based autopsy study. Neurology. 2003; 60: 652-656.
- Fagan AM, Mintun MA, Shah AR, Aldea P, Roe CM, Mach RH, et al. Cerebrospinal fluid tau and ptau(181) increase with cortical amyloid deposition in cognitively normal individuals: implications for future clinical trials of Alzheimer's disease. EMBO Mol Med. 2009; 1: 371-380.
- Tapiola T, Alafuzoff I, Herukka SK, Parkkinen L, Hartikainen P, Soininen H, et al. Cerebrospinal fluid {beta}-amyloid 42 and tau proteins as biomarkers of Alzheimer-type pathologic changes in the brain. Arch Neurol. 2009; 66: 382-389.
- Tapiola T, Overmyer M, Lehtovirta M, Helisalmi S, Ramberg J, Alafuzoff I, et al. The level of cerebrospinal fluid tau correlates with neurofibrillary tangles in Alzheimer's disease. Neuroreport. 1997; 8: 3961-3963.
- Buerger K, Ewers M, Pirttilä T, Zinkowski R, Alafuzoff I, Teipel SJ, et al. CSF phosphorylated tau protein correlates with neocortical neurofibrillary pathology in Alzheimer's disease. Brain. 2006; 129: 3035-3041.
- Csernansky JG, Miller JP, McKeel D, Morris JC. Relationships among cerebrospinal fluid biomarkers in dementia of the Alzheimer type. Alzheimer Dis Assoc Disord. 2002; 16: 144-149.
- Riemenschneider M, Schmolke M, Lautenschlager N, Guder WG, Vanderstichele H, Vanmechelen E, et al. Cerebrospinal beta-amyloid ((1-42)) in early Alzheimer's disease: association with apolipoprotein E genotype and cognitive decline. Neurosci Lett. 2000; 284: 85-88.
- Lin YT, Cheng JT, Yao YC, Juo, Lo YK, Lin CH, et al. Increased total TAU but not amyloid-beta(42) in cerebrospinal fluid correlates with short-term memory impairment in Alzheimer's disease. J Alzheimers Dis. 2009; 18: 907-918.
- Ivanoiu A, Sindic CJ. Cerebrospinal fluid TAU protein and amyloid beta42 in mild cognitive impairment: prediction of progression to Alzheimer's disease and correlation with the neuropsychological examination. Neurocase. 2005; 11: 32-39.
- Mehta PD, Pirttila T, Patrick BA, Barshatzky M, Mehta SP. Amyloid beta protein 1-40 and 1-42 levels in matched cerebrospinal fluid and plasma from patients with Alzheimer disease. Neurosci Lett. 2001; 304: 102-106.
- Tato RE, Frank A, Hernanz A. Tau protein concentrations in cerebrospinal fluid of patients with dementia of the Alzheimer type. J Neurol Neurosurg Psychiatry. 1995; 59: 280-283.
- Kanai M, Matsubara E, Isoe K, Urakami K, Nakashima K, Arai H, et al. Longitudinal study of cerebrospinal fluid levels of tau, A beta1-40, and A beta1-42(43) in Alzheimer's disease: a study in Japan. Ann Neurol. 1998; 44: 17-26.
- Andersson C, Blennow K, Johansson SE, Almkvist O, Engfeldt P, Lindau M, et al. Differential CSF biomarker levels in APOE-epsilon4-positive and -negative patients with memory impairment. Dement Geriatr Cogn Disord. 2007; 23: 87-95.
- Sämgård K, Zetterberg H, Blennow K, Hansson O, Minthon L, Londos E. Cerebrospinal fluid total tau as a marker of Alzheimer's disease intensity. Int J Geriatr Psychiatry. 2010; 25: 403-410.
- Sunderland T, Linker G, Mirza N, Putnam KT, Friedman DL, Kimmel LH, et al. Decreased beta-amyloid1-42 and increased tau levels in cerebrospinal fluid of patients with Alzheimer disease. JAMA. 2003; 289: 2094-2103.
- Motter R, Vigo-Pelfrey C, Kholodenko D, Barbour R, Johnson-Wood K, Galasko D, et al. Reduction of beta-amyloid peptide42 in the cerebrospinal fluid of patients with Alzheimer's disease. Ann Neurol. 1995; 38: 643-648.
- Mehta PD, Pirttilä T, Mehta SP, Sersen EA, Aisen PS, Wisniewski HM. Plasma and cerebrospinal fluid levels of amyloid beta proteins 1-40 and 1-42 in Alzheimer disease. Arch Neurol. 2000; 57: 100-105.
- Mattsson N, Zetterberg H, Hansson O, Andreasen N, Parnetti L, Jonsson M, et al. CSF biomarkers and incipient Alzheimer disease in patients with mild cognitive impairment. JAMA. 2009; 302: 385-393.
- Mattsson N, Blennow K, Zetterberg H. CSF biomarkers: pinpointing Alzheimer pathogenesis. Ann N Y Acad Sci. 2009; 1180: 28-35.
- Hulstaert F, Blennow K, Ivanoiu A, Schoonderwaldt HC, Riemenschneider M, De Deyn PP, et al. Improved discrimination of AD patients using beta-amyloid(1-42) and tau levels in CSF. Neurology. 1999; 52: 1555-1562.
- Hampel H, Teipel SJ, Fuchsberger T, Andreasen N, Wiltfang J, Otto M, et al. Value of CSF beta-amyloid1-42 and tau as predictors of Alzheimer's disease in patients with mild cognitive impairment. Mol Psychiatry. 2004; 9: 705-710.
- Grossman M, Farmer J, Leight S, Work M, Moore P, Van Deerlin V, et al. Cerebrospinal fluid profile in frontotemporal dementia and Alzheimer's disease. Ann Neurol. 2005; 57: 721-729.
- Galasko D, Chang L, Motter R, Clark CM, Kaye J, Knopman D, et al. High cerebrospinal fluid tau and low amyloid beta42 levels in the clinical diagnosis of Alzheimer disease and relation to apolipoprotein E genotype. Arch Neurol. 1998; 55: 937-945.
- Buerger K, Zinkowski R, Teipel SJ, Arai H, DeBernardis J, Kerkman D, et al. Differentiation of geriatric major depression from Alzheimer's disease with CSF tau protein phosphorylated at threonine 231. Am J Psychiatry. 2003; 160: 376-379.
- Andreasen N, Hesse C, Davidsson P, Minthon L, Wallin A, Winblad B, et al. Cerebrospinal fluid beta-amyloid(1-42) in Alzheimer disease: differences between early- and late-onset Alzheimer disease and stability during the course of disease. Arch Neurol. 1999; 56: 673-680.
- Zetterberg H, Wahlund LO, Blennow K. Cerebrospinal fluid markers for prediction of Alzheimer's disease. Neurosci Lett. 2003; 352: 67-69.
- Shaw LM, Vanderstichele H, Knapik-Czajka M, Clark CM, Aisen PS, Petersen RC, et al. Cerebrospinal fluid biomarker signature in Alzheimer's disease neuroimaging initiative subjects. Ann Neurol. 2009; 65: 403-413.
- Riemenschneider M, Lautenschlager N, Wagenpfeil S, Diehl J, Drzezga A, et al. Cerebrospinal fluid tau and beta-amyloid 42 proteins identify Alzheimer disease in subjects with mild cognitive impairment. Arch Neurol. 2002; 59: 1729-1734.
- Herukka SK, Hallikainen M, Soininen H, Pirttilä T. CSF Abeta42 and tau or phosphorylated tau and prediction of progressive mild cognitive impairment. Neurology. 2005; 64: 1294-1297.
- Hansson O, Zetterberg H, Buchhave P, Londos E, Blennow K, Minthon L. Association between CSF biomarkers and incipient Alzheimer's disease in patients with mild cognitive impairment: a follow-up study. Lancet Neurol. 2006; 5: 228-234.
- Brys M, Pirraglia E, Rich K, Rolstad S, Mosconi L, Switalski R, et al. Prediction and longitudinal study of CSF biomarkers in mild cognitive impairment. Neurobiol Aging. 2009; 30: 682-690.
- Bouwman FH, Schoonenboom SN, van der Flier WM, van Elk EJ, Kok A, Barkhof F, Blankenstein MA. CSF biomarkers and medial temporal lobe atrophy predict dementia in mild cognitive impairment. Neurobiol Aging. 2007; 28: 1070-1074.
- Parnetti L, Lanari A, Amici S, Gallai V, Vanmechelen E, Hulstaert F; Phospho-Tau International Study Group. CSF phosphorylated tau is a possible marker for discriminating Alzheimer's disease from dementia with Lewy bodies. Phospho-Tau International Study Group. Neurol Sci. 2001; 22: 77-78.
- Gómez-Tortosa E, Gonzalo I, Fanjul S, Sainz MJ, Cantarero S, Cemillán C, et al. Cerebrospinal fluid markers in dementia with lewy bodies compared with Alzheimer disease. Arch Neurol. 2003; 60: 1218-1222.
- Verbeek MM, Pijnenburg YA, Schoonenboom NS, Kremer BP, Scheltens P. Cerebrospinal fluid tau levels in frontotemporal dementia. Ann Neurol. 2005; 58: 656-657.
- Rosso SM, van Herpen E, Pijnenburg YA, Schoonenboom NS, Scheltens P, Heutink P, et al. Total tau and phosphorylated tau 181 levels in the cerebrospinal fluid of patients with frontotemporal dementia due to P301L and G272V tau mutations. Arch Neurol. 2003; 60: 1209-1213.
- Riemenschneider M, Wagenpfeil S, Diehl J, Lautenschlager N, Theml T, Heldmann B, et al. Tau and Abeta42 protein in CSF of patients with frontotemporal degeneration. Neurology. 2002; 58: 1622-1628.
- Parnetti L, Tiraboschi P, Lanari A, Peducci M, Padiglioni C, D'Amore C, et al. Cerebrospinal fluid biomarkers in Parkinson's disease with dementia and dementia with Lewy bodies. Biol Psychiatry. 2008; 64: 850-855.
- Kasuga K, Tokutake T, Ishikawa A, Uchiyama T, Tokuda T, Onodera O, et al. Differential levels of alpha-synuclein, beta-amyloid42 and tau in CSF between patients with dementia with Lewy bodies and Alzheimer's disease. J Neurol Neurosurg Psychiatry. 2010; 81: 608-610.
- Buerger K, Zinkowski R, Teipel SJ, Tapiola T, Arai H, Blennow K, et al. Differential diagnosis of Alzheimer disease with cerebrospinal fluid levels of tau protein phosphorylated at threonine 231. Arch Neurol. 2002; 59: 1267-1272.
- Blennow K, Hampel H. CSF markers for incipient Alzheimer's disease. Lancet Neurol. 2003; 2: 605-613.
- Hort J, Bartos A, Pirttilä T, Scheltens P. Use of cerebrospinal fluid biomarkers in diagnosis of dementia across Europe. Eur J Neurol. 2010; 17: 90-96.
- Visser PJ, Verhey F, Knol DL, Scheltens P, Wahlund LO, Freund-Levi Y, et al. Prevalence and prognostic value of CSF markers of Alzheimer's disease pathology in patients with subjective cognitive impairment or mild cognitive impairment in the DESCRIPA study: a prospective cohort study. Lancet Neurol. 2009; 8: 619-627.
- Buerger K, Frisoni G, Uspenskaya O, Ewers M, Zetterberg H, Geroldi C, et al. Validation of Alzheimer's disease CSF and plasma biological markers: the multicentre reliability study of the pilot European Alzheimer's Disease Neuroimaging Initiative (E-ADNI). Exp Gerontol. 2009; 44: 579-585.
- Braak H, Braak E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991; 82: 239-259.
- Bierer LM, Hof PR, Purohit DP, Carlin L, Schmeidler J, Davis KL, et al. Neocortical neurofibrillary tangles correlate with dementia severity in Alzheimer's disease. Arch Neurol. 1995; 52: 81-88.
- Arriagada PV, Growdon JH, Hedley-Whyte ET, Hyman BT. Neurofibrillary tangles but not senile plaques parallel duration and severity of Alzheimer's disease. Neurology. 1992; 42: 631-639.
- Geldmacher DS, Whitehouse PJ Jr. Differential diagnosis of Alzheimer's disease. Neurology. 1997; 48: S2-9.
- van Harten AC, Kester MI, Visser PJ, Blankenstein MA, Pijnenburg YA, van der Flier WM, et al. Tau and p-tau as CSF biomarkers in dementia: a meta-analysis. Clin Chem Lab Med. 2011; 49: 353-366.
- Meiner Z, Kahana E, Baitcher F, Korczyn AD, Chapman J, Cohen OS, et al. Tau and 14-3-3 of genetic and sporadic Creutzfeldt-Jakob disease patients in Israel. J Neurol. 2011; 258: 255-262.
- Reis T, Brand&aTilde;o CO, Freire Coutinho ES, Engelhardt E, Laks J. Cerebrospinal fluid biomarkers in Alzheimer's disease and geriatric depression: preliminary findings from Brazil. CNS Neurosci Ther. 2012; 18: 524-529.
- Pomara N, Bruno D, Sarreal AS, Hernando RT, Nierenberg J, Petkova E, et al. Lower CSF amyloid beta peptides and higher F2-isoprostanes in cognitively intact elderly individuals with major depressive disorder. Am J Psychiatry. 2012; 169: 523-530.
- Ramakers IH, Verhey FR, Scheltens P, Hampel H, Soininen H, Aalten P, et al. Anxiety is related to Alzheimer cerebrospinal fluid markers in subjects with mild cognitive impairment. Psychol Med. 2013; 43: 911-920.
- Clark CM, Xie S, Chittams J, Ewbank D, Peskind E, Galasko D, et al. Cerebrospinal fluid tau and beta-amyloid: how well do these biomarkers reflect autopsy-confirmed dementia diagnoses? Arch Neurol. 2003; 60: 1696-1702.
- Blennow K, Wallin A, Agren H, Spenger C, Siegfried J, Vanmechelen E. Tau protein in cerebrospinal fluid: a biochemical marker for axonal degeneration in Alzheimer disease? Mol Chem Neuropathol. 1995; 26: 231-245.
- Mecocci P, Cherubini A, Bregnocchi M, Chionne F, Cecchetti R, Lowenthal DT, et al. Tau protein in cerebrospinal fluid: a new diagnostic and prognostic marker in Alzheimer disease? Alzheimer Dis Assoc Disord. 1998; 12: 211-214.
- Vanmechelen E, Vanderstichele H, Hulstaert F, Andreasen N, Minthon L, Winblad B, et al. Cerebrospinal fluid tau and beta-amyloid(1-42) in dementia disorders. Mech Ageing Dev. 2001; 122: 2005-2011.
- Irwin DJ, Trojanowski JQ, Grossman M. Cerebrospinal fluid biomarkers for differentiation of frontotemporal lobar degeneration from Alzheimer's disease. Front Aging Neurosci. 2013; 5: 6.
- Toledo JB, Brettschneider J, Grossman M, Arnold SE, Hu WT, Xie SX, et al. CSF biomarkers cutoffs: the importance of coincident neuropathological diseases. Acta Neuropathol. 2012; 124: 23-35.
- Bian H, Van Swieten JC, Leight S, Massimo L, Wood E, Forman M, et al. CSF biomarkers in frontotemporal lobar degeneration with known pathology. Neurology. 2008; 70: 1827-1835.
- Herbert MK, Aerts MB, Kuiperij HB, Claassen JA, Spies PE, Esselink RA, et al. Addition of MHPG to Alzheimer's disease biomarkers improves differentiation of dementia with Lewy bodies from Alzheimer's disease but not other dementias. Alzheimers Dement. 2014; 10: 448-455.
- Mollenhauer B, Bibl M, Wiltfang J, Steinacker P, Ciesielczyk B, Neubert K, et al. Total tau protein, phosphorylated tau (181p) protein, beta-amyloid(1-42), and beta-amyloid(1-40) in cerebrospinal fluid of patients with dementia with Lewy bodies. Clin Chem Lab Med. 2006; 44: 192-195.
- Vranová HP, Hényková E, Kaiserová M, Menšíková K, Vaštík M, Mareš J, et al. Tau protein, beta-amyloid1?42 and clusterin CSF levels in the differential diagnosis of Parkinsonian syndrome with dementia. J Neurol Sci. 2014; 343: 120-124.
- Kaerst L, Kuhlmann A, Wedekind D, Stoeck K, Lange P, Zerr I. Using cerebrospinal fluid marker profiles in clinical diagnosis of dementia with Lewy bodies, Parkinson's disease, and Alzheimer's disease. J Alzheimers Dis 2014; 38: 63-73.
- Arai H, Morikawa Y, Higuchi M, Matsui T, Clark CM, Miura M, et al. Cerebrospinal fluid tau levels in neurodegenerative diseases with distinct tau-related pathology. Biochem Biophys Res Commun. 1997; 236: 262-264.
- Mollenhauer B, Trenkwalder C, von Ahsen N, Bibl M, Steinacker P, Brechlin P, et al. Beta-amlyoid 1-42 and tau-protein in cerebrospinal fluid of patients with Parkinson's disease dementia. Dement Geriatr Cogn Disord. 2006; 22: 200-208.
- Molina L, Touchon J, Herpé M, Lefranc D, Duplan L, Cristol JP, et al. Tau and apo E in CSF: potential aid for discriminating Alzheimer's disease from other dementias. Neuroreport. 1999; 10: 3491-3495.
- Kanemaru K, Kameda N, Yamanouchi H. Decreased CSF amyloid beta42 and normal tau levels in dementia with Lewy bodies. Neurology. 2000; 54: 1875-1876.
- Andreasen N, Minthon L, Davidsson P, Vanmechelen E, Vanderstichele H, Winblad B, et al. Evaluation of CSF-tau and CSF-Abeta42 as diagnostic markers for Alzheimer disease in clinical practice. Arch Neurol. 2001; 58: 373-379.
- Bjerke M, Portelius E, Minthon L, Wallin A, Anckarsäter H, Anckarsäter R, et al. Confounding factors influencing amyloid Beta concentration in cerebrospinal fluid. Int J Alzheimers Dis. 2010; 2010.
- Rosenmann H. CSF biomarkers for amyloid and tau pathology in Alzheimer's disease. J Mol Neurosci. 2012; 47: 1-14.
- Vanderstichele H, Bibl M, Engelborghs S, Le Bastard N, Lewczuk P, Molinuevo JL, et al. Standardization of preanalytical aspects of cerebrospinal fluid biomarker testing for Alzheimer's disease diagnosis: a consensus paper from the Alzheimer's Biomarkers Standardization Initiative. Alzheimers Dement 2012; 8: 65-73.
- Mattsson N, Andreasson U, Persson S, Carrillo MC, Collins S, Chalbot S, et al. CSF biomarker variability in the Alzheimer's Association quality control program. Alzheimers Dement. 2013; 9: 251-261.
- Koric L, Felician O, Guedj E, Hubert AM, Mancini J, Boucraut J, et al. Could clinical profile influence CSF biomarkers in early-onset Alzheimer disease? Alzheimer Dis Assoc Disord. 2010; 24: 278-283.
- Schmidt C, Wolff M, Weitz M, Bartlau T, Korth C, Zerr I. Rapidly progressive Alzheimer disease. Arch Neurol. 2011; 68: 1124-1130.
- Wallin AK, Blennow K, Zetterberg H, Londos E, Minthon L, Hansson O. CSF biomarkers predict a more malignant outcome in Alzheimer disease. Neurology. 2010; 74: 1531-1537.
- Andreasen N, Minthon L, Vanmechelen E, Vanderstichele H, Davidsson P, Winblad B, et al. Cerebrospinal fluid tau and Abeta42 as predictors of development of Alzheimer's disease in patients with mild cognitive impairment. Neurosci Lett. 1999; 273: 5-8.
- Blennow K, Zetterberg H, Minthon L, Lannfelt L, Strid S, Annas P, et al. Longitudinal stability of CSF biomarkers in Alzheimer's disease. Neurosci Lett. 2007; 419: 18-22.
- Mollenhauer B, Bibl M, Trenkwalder C, Stiens G, Cepek L, Steinacker P, et al. Follow-up investigations in cerebrospinal fluid of patients with dementia with Lewy bodies and Alzheimer's disease. J Neural Transm. 2005; 112: 933-948.
- Andreasen N, Minthon L, Clarberg A, Davidsson P, Gottfries J, Vanmechelen E, et al. Sensitivity, specificity, and stability of CSF-tau in AD in a community-based patient sample. Neurology. 1999; 53: 1488-1494.
- Wallin AK, Blennow K, Andreasen N, Minthon L. CSF biomarkers for Alzheimer's Disease: levels of beta-amyloid, tau, phosphorylated tau relate to clinical symptoms and survival. Dement Geriatr Cogn Disord. 2006; 21: 131-138.
- LoPresti P. Regulation and differential expression of tau mRNA isoforms as oligodendrocytes mature in vivo: implications for myelination. Glia. 2002; 37: 250-257.
- Iwasaki Y, Yoshida M, Hattori M, Goto A, Aiba I, Hashizume Y, et al. Distribution of tuft-shaped astrocytes in the cerebral cortex in progressive supranuclear palsy. Acta Neuropathol. 2004; 108: 399-405.
- Frost B, Jacks RL, Diamond MI. Propagation of tau misfolding from the outside to the inside of a cell. J Biol Chem. 2009; 284: 12845-12852.
- Clavaguera F, Bolmont T, Crowther RA, Abramowski D, Frank S, Probst A, et al. Transmission and spreading of tauopathy in transgenic mouse brain. Nat Cell Biol. 2009; 11: 909-913.
- Kfoury N, Holmes BB, Jiang H, Holtzman DM, Diamond MI. Trans-cellular propagation of Tau aggregation by fibrillar species. J Biol Chem. 2012; 287: 19440-19451.
- Schmidt ML, Gur RE, Gur RC, Trojanowski JQ. Intraneuronal and extracellular neurofibrillary tangles exhibit mutually exclusive cytoskeletal antigens. Ann Neurol. 1988; 23: 184-189.
- Karch CM, Jeng AT, Goate AM. Extracellular Tau levels are influenced by variability in Tau that is associated with tauopathies. J Biol Chem. 2012; 287: 42751-42762.
- Zhukareva V, Sundarraj S, Mann D, Sjogren M, Blenow K, Clark CM, et al. Selective reduction of soluble tau proteins in sporadic and familial frontotemporal dementias: an international follow-up study. Acta Neuropathol. 2003; 105: 469-476.
- Vanderstichele H, De Vreese K, Blennow K, Andreasen N, Sindic C, Ivanoiu A, et al. Analytical performance and clinical utility of the INNOTEST PHOSPHO-TAU181P assay for discrimination between Alzheimer's disease and dementia with Lewy bodies. Clin Chem Lab Med. 2006; 44: 1472-1480.
- De Riva V, Galloni E, Marcon M, Di Dionisio L, Deluca C, Meligrana L, et al. Analysis of combined CSF biomarkers in AD diagnosis. Clin Lab. 2014; 60: 629-634.
- Marcus C, Mena E, Subramaniam RM. Brain PET in the Diagnosis of Alzheimer's Disease. Clin Nucl Med. 2014; 39: e413-426.
- Suri S, Topiwala A, Mackay CE, Ebmeier KP, Filippini N. Using structural and diffusion magnetic resonance imaging to differentiate the dementias. Curr Neurol Neurosci Rep. 2014; 14: 475.