Abstract
Background: The corona virus disease 2019 (COVID-19) begun as an outbreak in China in December 2019 and soon became a pandemic. The disease was previously unknown to humanity, hence its clinical aspects key to adequate assessment and management are yet evolving. An understanding to these is essential to ensure optimal patient directed care amid high burden of disease. Clinicopathological factors as Liver Function Tests might be a cornerstone in predicting the severity of disease.
Objectives: To assess for changes in Liver Function Tests in COVID-19 patients and its association with disease severity.
Methods: PCR confirmed COVID-19 patients with Liver Function Tests on presentation were enrolled at Allied Hospitals of Rawalpindi, Pakistan. Demographic data and Liver Function Tests were entered and analyzed. Descriptive statistics were applied and statistical tests were run to check for association with disease severity.
Results: Patients with higher age and comorbidities are more likely to develop severe disease. Liver Function Tests are more likely to be deranged in severe patients with AST levels significantly associated with disease severity.
Conclusion: Deranged Liver Function tests particularly AST levels may act as a marker of predicting severe COVID-19.
Keywords: COVID-19; SARS COV 2; Severity; Liver Function Tests; AST; Pakistan; Rawalpindi
Introduction
The corona virus disease 2019 (COVID-19) began as an outbreak in December 2019 in Wuhan, China [1]. The disease reported outside of China on January 13, 2020 [2]. Severe Acute Respiratory Syndrome Corona virus 2 (SARS-COV-2) being highly contagious turned out to be a worldwide threat and declared Pandemic by WHO on March 11, 2020 [3]. Apart from having high infectivity, the disease also carries a significant mortality varying from 4.1% in USA to 14.6% in Belgium [4,5,6]. Europe and America are carrying a high disease burden and consequently having its adverse economic and health sector effects [7]. This puts the third world countries that are relatively spared so far into a risk of developing an even worse potential crisis.
The corona viruses are known to humanity for quite a few decades [8,9,10]. There have been outbreaks previously reported as Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS) [11,12,13,14]. However, SARSCOV- 2 is a strain completely new to mankind. Its epidemiological and clinicopathological aspects are rapidly evolving with each passing day. The initial patterns of disease were understood from case reports and a few small scale studies [15,16,17,18]. New trends are being established in the light of further studies and systematic reviews [19,20]. A considerable portion of patients who develop the disease are either asymptomatic or develop mild to moderate symptoms. These patients mainly require isolation and other precautionary measures so as to contain the spread. Indoor hospitalized care is not a necessity for most of these as they are clinically stable and no specialized monitoring or intervention has been proven to any benefit [21].
COVID-19 may be severe in around 30 % of patients calling for an observation in a health facility, with a fraction potential candidate for intensive care [22,23]. Under increasingly overwhelming COVID-19 burden on health care system ensuring patient focused quality of care based on disease severity is of utmost importance. Hence clinicopathological factors predictive of severity of disease would be a cornerstone in prioritizing resources effectively. The present study is designed to assess for accuracy of Liver Function Tests in defining the severity of COVID-19 and possible need for intensive care.
Objectives
To assess various risk factors determining severity of COVID-19, to investigate changes in Liver Function Tests and to assess its association with Severity of disease.
Materials and Methods
This was a cross sectional study conducted on patients quarantined at public sector hospitals affiliated with Rawalpindi Medical University, Rawalpindi from March 24 to April 20, 2020 after ethical approval from Institutional Research Forum, Rawalpindi Medical University. Patients were enrolled in this study via non probability consecutive sampling technique.
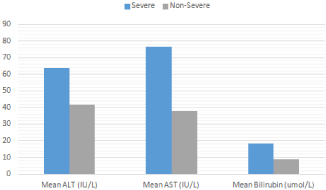
Figure 1: A comparison of Mean values of ALT, AST and Bilirubin
among ATS Severe and Non-Severe COVID-19 patients. (ALT = Alanine
Aminotransferase, AST = Aspartate Aminotransferase).
A total of 44 patients were included with PCR positive for COVID-19 based on the World Health Organization guidelines with Liver Function Tests (LFTs) including Serum Alanine Aminotransferases (ALT), Aspartate Aminotransferases (AST) and Total Bilirubin performed on presentation. Patients with preexisting Liver diseases were excluded. Data was collected using structured proforma comprising of data regarding patient gender, age, contact history, comorbidities including Diabetes and Hypertension, serum ALT, AST and Bilirubin levels. Patient anonymity was maintained. Patients were grouped into Severe and Non-Severe based on American Thoracic Society (ATS) classification of Community acquired Pneumonia [24].
Categorical variables were described as frequency and percentages, and continuous variables as mean and Standard Deviation (SD). Means for continuous variables were compared using independent group t-tests. Comparison of categorical variables was done using Fisher exact test, if the cell counts were small. Logistic Regression was applied on significant association of continuous variables with severity. Data was entered and analyzed on Intel Business Machine Statistical Package for Social Sciences (IBM SPSS) version 25.
Results
A total of 44 PCR based confirmed COVID-19 patients were included in study with Liver Function Tests (ALT, AST, Bilirubin) performed. Mean age was 50.89 ± 18.04 years. 63.6 % (N = 28) patients were Male while 36.4% (N = 16) were Female. Based on ATS Classification 18.2 % (N = 8) patients were Severe while 81.8 % (N = 36) were Non-Severe. Regarding comorbidities 68.2% (N = 30) were normotensive and normoglycemic while 31.81% (N = 14) were diabetic and/or hypertensive (Table 1).
Mean Age among Severe patients (66.625 ± 14.4611 years) was higher than that of Non-Severe patients (47.3889 ± 17.001 years) (t (42) = 2.964, p = 0.005). Gender has no significant effect in determining disease severity (p = 0.689). Patients who are Hypertensive and/or Diabetic are more likely to develop Severe disease (p = 0.001). Mean ALT, AST and Bilirubin values for Severe patients (63.75 IU/L, 76.625 IU/L, 1.0625 mg/dl) were higher as compared to that of Non-Severe patients (41.6667 IU/L, 37.9524 IU/L, 0.5194 mg/dl), the relation was significant for AST and Bilirubin (t (27) = 3.819, p = 0.001)(t (42) = 2.667 p = 0.011) (Figure 1) (Table 2). By applying Logistic Regression analysis increase in Age was significantly associated with developing Severe disease (OR = 1.077, CI = 1.015 - 1.143, p = 0.014), similarly increase in AST levels were significantly associated with Severe disease (OR = 1.060, CI = 1.006 - 1.116, p = 0.027) (Table 3).
Variable
% (N)
Gender
Male
63.6 (28)
Female
18.2 (8)
Age (years)
Mean (SD)
50.89 (± 18.04)
Comorbidities
No comorbidities
68.2 (30)
Diabetic and/or Hypertensive
31.8 (14)
Contact History
Contact with known or suspected case
65.9 (29)
No contact
34.1 (15)
ATS* Classification
Severe
18.2 (8)
Non-Severe
81.8 (36)
*ATS = American Thoracic Society
Table 1: Characteristics of Studied Patients.
Discussion
This is a Pakistan based study conducted on the very first COVID-19 patients presenting in Allied Hospitals of Rawalpindi Medical University. Various demographic factors were studies in comparison with the Severity of disease. Hepatic clinicopathological factors as ALT, AST and Bilirubin were studied as a possible link in developing severe disease. The associations were significant for age, comorbidities and AST levels.
The present study shows that COVID-19 was more common in men; however, the association was not statistically significant. A study based in China by Huang C et al. also demonstrated the prevalence of COVID-19 being more in men than women although the mechanism is still unclear [25].
Regarding age of hospitalized COVID-19 patients, the present study shows a mean age of 50.89 years. This is quite consistent with similar studies conducted in China where mean ages were 51 and 49.5 years respectively [26,27]. However, a French study showed a bit lower mean age of 45.1 years indicating relatively younger populations being affected and hospitalized in that region [28].
In our study, majority of patients were non-severe according to the community acquired pneumonia classification of American Thoracic Society (ATS). This is in accordance with a study conducted in Wuhan, China in which 68.29% patients were non-critical [25]. Majority of patients were also non-severe in another China based study [29].
A study conducted in Wuhan, China showed 32% of COVID-19 patients having comorbidities including diabetes (20%) and hypertension (15%) [25]. This seconds our findings that 31.8% patients were having comorbidities conditions. Another study showed that the most prevalent comorbidity was hypertension (16.9%), followed by diabetes (8.2%) [29].
Our study showed increasing the age, increases the risk for developing severe disease. A report by CDC United States also suggested that older age groups are more prone in progressing towards severe disease [30]. However, a study conducted in China showed that there was no statistically significant difference in age groups between severe and non- severe patients.25 Regarding gender this study showed that there was no significant effect of gender on disease severity which is quite consistent with the results of another study conducted in China [31].
Diseases such as diabetes, hypertension, respiratory system disease, cardiovascular disease and their susceptibility conditions may be linked to the pathogenesis of COVID-19. A study by Jin JM et al. significantly correlates the severity of COVID19 with the preexisting comorbid conditions which is in accordance with the results of our study [31]. Another study conducted in China showed the prevalence of hypertension and diabetes among severe patients as 30.8% which is almost equal to 31.8% comorbidities including Diabetes and Hypertension in our study [32].
Variable
Severe % (N)
Non-Severe % (N)
Total % (N)
p-value*
Gender
18.2 (8)
81.8 (36)
100 (44)
0.689**
Male
21.4 (6)
78.6 (22)
100 (28)
Female
12.5 (2)
87.5 (14)
100 (16)
Age (years)
Mean (SD)
66.63 (± 14.46)
47.38 (±17.00)
50.89 (± 18.04)
0.005***
Comorbidities
18.2 (8)
81.8 (36)
100 (44)
0.001**
No comorbidities
3.3 (1)
96.7 (29)
100 (30)
Diabetic and/or Hypertensive
50 (7)
50 (7)
100 (14)
Contact History
18.2 (8)
81.8 (36)
100 (44)
Contact with known or suspected case
13.8 (4)
86.2 (25)
100 (29)
0.414**
No contact
26.7 (4)
73.3 (11)
100 (15)
ALT (IU/L)
Mean (SD)
63.75 (± 42.52)
47.38 (± 27.56)
45.68 (±31.42)
0.072***
AST (IU/L)
Mean (SD)
76.62 (± 38.55)
37.95 (± 16.78)
48.62 (±29.70)
0.001***
Bilirubin (mg/dl)
Mean (SD)
1.0625 (± 1.12)
0.5194 (±0.28)
0.6182 (±0.56)
0.011***
*p-value considered significant below 0.05 **Fischer Exact Test applied ***Independent Sample t-test applied
Table 2: Comparison of Patient variables with Severity of COVID-19.
Variable
Odds Ratio
Confidence Interval (95%)
p-value*
Lower value
Upper value
Age
1.077
1.015
1.143
0.014
AST (IU/L)
1.06
1.006
1.116
0.027
Total Bilirubin (mg/dl)
6.595
0.622
69.969
0.117
*p-value considered significant below 0.05
Table 3: Logistic Regression analysis of Age, AST and Bilirubin levels with Severity of COVID-19
In our study, the relation of AST, ALT and total bilirubin was sought with the severity of disease. The levels of LFTs on presentation
among severe patients were higher as compared to non-severe patients with association of AST being statistically significant. A China based study showed significantly higher levels of ALT and Bilirubin among ICU patients [25]. The association of AST was also seen statistically significant in another study conducted by Guan WJ et al. in Wuhan, China [33]. However, another study by Yang et al showed no significant difference in values of liver tests among survivors and non-survivors [34]. This represents potential of AST levels in predicting the severity of COVID-19 disease so as to assess the possible pre hand need for ICU care and preparing in advance intensive care infrastructure, equipment and personals and prioritize the resources effectively to provide quality care without compromising patient wellbeing.
Conclusion
Increase in age and presence of comorbidities is significantly associated with developing Severe COVID-19. Patients with higher AST levels are at more risk to develop severe disease, rendering it as a potential marker for predicting the severity of COVID-19. The positive correlation between COVID-19 Severity and Liver damage also mandates its routine monitoring in COVID-19.
References
- World Health Organization. Novel Coronavirus-China. 2020.
- World Health Organization. WHO statement on novel coronavirus in Thailand. 2020.
- World Health Organization. WHO Timeline - COVID-19. 2020.
- Cascella M, Rajnik M, Cuomo A, Dulebohn SC, Di Napoli R. Features, evaluation and treatment coronavirus (COVID-19). InStatPearls. 2020.
- Business Insider. The US is the hardest-hit country in the coronavirus pandemic-but its 4% death rate is far lower than that of many other countries.
- Johns Hopkins University. How does mortality differ across countries?. 2020.
- WHO Health Emergency Dashboard. Coronavirus (COVID-19). 2020.
- England JJ, Frye CS, Enright EA. Negative contrast electron microscopic diagnosis of viruses of neonatal calf diarrhea. The Cornell veterinarian. 1976; 66: 172-82.
- Caul EO, Egglestone SI. Further studies on human enteric coronaviruses. Archives of Virology. 1977; 54: 107.
- Tyrrell DAJ, Myint SH. Coronaviruses. In: Baron S. (ed). Medical Microbiology. 4th edition. Galveston (TX): University of Texas Medical Branch at Galveston. 1996; 60.
- Ksiazek TG, Erdman D, Goldsmith CS, Zaki SR, Peret T, Emery S, et al. A novel coronavirus associated with severe acute respiratory syndrome. New England journal of medicine. 2003; 348: 1953-66.
- Poutanen SM, Low DE, Henry B, Finkelstein S, Rose D, Green K, et al. Identification of severe acute respiratory syndrome in Canada. New England Journal of Medicine. 2003; 348: 1995-2005.
- Zumla A, Hui DS, Perlman S. Middle East respiratory syndrome. The Lancet. 2015; 386: 995-1007.
- Assiri A, Al-Tawfiq JA, Al-Rabeeah AA, Al-Rabiah FA, Al-Hajjar S, Al-Barrak A, et al. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: a descriptive study. The Lancet. 2013; 13: 752-61.
- Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus–infected pneumonia. New England Journal of Medicine. 2020.
- Lescure FX, Bouadma L, Nguyen D, Parisey M, Wicky PH, Behillil S, et al. Clinical and virological data of the first cases of COVID-19 in Europe: a case series. The Lancet Infectious Diseases. 2020.
- Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. Jama. 2020; 323: 1061-9.
- Liu W, Zhang Q, Chen J, Xiang R, Song H, Shu S, et al. Detection of Covid-19 in children in early January 2020 in Wuhan, China. New England Journal of Medicine. 2020; 382: 1370-1.
- Rodriguez-Morales AJ, Cardona-Ospina JA, Gutiérrez-Ocampo E, Villamizar-Peña R, Holguin-Rivera Y, Escalera-Antezana JP, et al. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel medicine and infectious disease. 2020; 101623.
- Arentz M, Yim E, Klaff L, Lokhandwala S, Riedo FX, Chong M, et al. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State. Jama. 2020.
- National Health Serivce. Self-isolation if you or someone you live with has coronavirus symptoms. 2020.
- Liu Y, Yan LM, Wan L, Xiang TX, Le A, Liu JM, et al. Viral dynamics in mild and severe cases of COVID-19. The Lancet Infectious Diseases. 2020.
- Lippi G, Plebani M, Henry BM. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clinica Chimica Acta. 2020.
- Metlay JP, Waterer GW, Long AC, Anzueto A, Brozek J, Crothers K, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. American journal of respiratory and critical care medicine. 2019; 200: e45-67.
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet. 2020; 395: 497-506.
- Shi H, Han X, Jiang N, Cao Y, Alwalid O, Gu J, et al. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. The Lancet Infectious Diseases. 2020.
- Ai T, Yang Z, Hou H, Zhan C, Chen C, Lv W, et al. Correlation of chest CT and RT-PCR testing in coronavirus disease 2019 (COVID-19) in China: a report of 1014 cases. Radiology. 2020; 200642.
- Gautret P, Lagier JC, Parola P, Meddeb L, Mailhe M, Doudier B, et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. International journal of antimicrobial agents. 2020; 105949.
- Guan WJ, Liang WH, Zhao Y, Liang HR, Chen ZS, Li YM, et al. Comorbidity and its impact on 1590 patients with Covid-19 in China: A Nationwide Analysis. European Respiratory Journal. 2020.
- COVID C, Team R. Severe outcomes among patients with coronavirus disease 2019 (COVID-19)-United States, February 12-March 16, 2020. MMWR Morb Mortal Wkly Rep. 2020; 69: 343-6.
- Jin JM, Bai P, He W, Wu F, Liu XF, Han DM, et al. Gender differences in patients with COVID-19: Focus on severity and mortality. medRxiv. 2020.
- Yang J, Zheng Y, Gou X, Pu K, Chen Z, Guo Q, et al. Prevalence of comorbidities in the novel Wuhan coronavirus (COVID-19) infection: a systematic review and meta-analysis. International Journal of Infectious Diseases. 2020.
- Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical characteristics of 2019 novel coronavirus infection in China. MedRxiv. 2020.
- Yang X, Yu Y, Xu J, Shu H, Liu H, Wu Y, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. The Lancet Respiratory Medicine. 2020.
