
Research Article
J Bacteriol Mycol. 2025; 12(1): 1226.
Diagnostic Accuracy between TrueNat and GeneXpert for Diagnosis of Pulmonary Tuberculosis in southern India - A Comparative Study
Muthaiah M1, Venkatachalam K1, Brammacharry U2*, Ramachandra V2, Palavesam S2, Mani MR3 and VidyaRaj CK4
1Department of Microbiology, State TB Training and Demonstration Centre, Intermediate Reference Laboratory, Government Hospital for Chest Diseases, Puducherry, India
2Institute of Basic Medical Sciences, University of Madras, Chennai, Tamil Nadu, India
3Department of Biochemistry, Queen Mary’s College, Madras, Tamil Nadu, India
4Department of Microbiology, Balaji Vidyapeeth (Deemed to be University), Puduchery, India
*Corresponding author: Dr. Usharani B, Institute of Basic Medical Sciences, University of Madras, Chennai, Tamil Nadu, India Email: cdirlpd@gmail.com
Received: July 26, 2025 Accepted: August 11, 2025 Published: August 14, 2025
Abstract
Enhancing access to reliable and accurate diagnosis of tuberculosis (TB) is a priority for global TB control. Molbio Diagnostics has developed the Truenat point-of-care molecular assays for detecting TB and resistance to rifampicin (RIF). This study aimed to compare the sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of GeneXpert with TrueNat, using the MGIT test as the gold standard. A cross-sectional comparative study involved 350 patients with suspected pulmonary TB at the Intermediate Reference Laboratory in the Government Hospital for Chest Diseases, Puducherry, India. The sensitivity, specificity, PPV, NPV, and diagnostic accuracy for tuberculosis diagnosis were calculated for TrueNat and GeneXpert and compared to the MGIT results. Data analysis was performed using MedCalc Software Ltd., specifically the Odds Ratio Calculator (Version 23.1.6. Among the 350 patients, 296 (84.57%) tested positive for TB by GeneXpert, while 280 (80.00%) tested positive with the TrueNat assay. For the GeneXpert assay in detecting pulmonary TB in sputum samples, the sensitivity was 98.66%, specificity was 96.15%, PPV was 99.32%, NPV was 92.59%, and accuracy was 98.29%. In comparison, the TrueNat technique had a sensitivity of 93.22%, specificity of 90.91%, PPV of 98.21%, NPV of 71.43%, and accuracy of 92.86%. The agreement between the TrueNat assay and GeneXpert with the BACTEC-MGIT-960 methods for detecting Mycobacterium tuberculosis in pulmonary samples was significant, with Kappa values of 0.76 (S.E.: 0.05) for TrueNat and 0.93 (S.E.: 0.03) for GeneXpert. Overall, the GeneXpert assay demonstrated greater sensitivity than TrueNat in respiratory samples. It is advisable to interpret TrueNat negative results cautiously and to correlate them closely with the patient’s clinical and treatment history.
Keywords: Mycobacterium tuberculosis; TrueNat; Sensitivity; Specificity; Accuracy
Abbreviations
DR-TB: Drug-resistant Tuberculosis; TB: Tuberculosis; DST: Drug Susceptibility Test; LTFU: Loss to follow-up; OR: Odds Ratio; CI: Confidence Interval; WHO: World Health Organization; RIF: Rifampicin; MTB: Mycobacterium Tuberculosis; ECT: Elute Collection Tube.
Introduction
Tuberculosis (TB) is a major global health challenge. In 2023, an estimated 10.8 million people fell ill with the disease, leading to approximately 1.61 million deaths. According to the WHO Global Tuberculosis Report 2024 [1], around 2.7 million individuals with TB went undiagnosed or unreported worldwide. This indicates a significant gap in the total number of cases, as only 8.2 million people were newly diagnosed. The report also emphasizes that while treatment coverage has improved, there are still notable gaps, particularly concerning multidrug-resistant TB (MDR-TB). Effective control of the tuberculosis (TB) epidemic requires rapid diagnosis and prompt treatment. Traditional culture and drug susceptibility testing methods take weeks or months for results, leading to prolonged ineffective therapy and ongoing transmission. Many high-burden countries lack the resources for these methods and rely on sputum smear microscopy. It is crucial to develop efficient point-of-care diagnostic tests for TB and drug resistance, especially in low-resource settings with high rates of pre-treatment loss to follow-up.
The currently recommended methods for diagnosing tuberculosis (TB) include molecular-based tests that can detect both TB and drugresistant forms of the disease. The World Health Organization (WHO) recommended the Xpert MTB/RIF assay [2] (developed by Cepheid, USA) in 2010 as a rapid molecular diagnostic tool for TB. In 2017, the WHO introduced the Xpert MTB/RIF Ultra assay, which offers increased sensitivity. The GeneXpert system, which utilizes both the Xpert and Ultra assays, has been widely implemented. However, the adoption of these tests in some areas has been limited due to several challenges, including the relatively high cost of the tests, insufficient infrastructure required for the equipment, and technical difficulties in maintaining the instruments. Until 2020, the Xpert system was the only WHO-recommended option for rapid molecular detection of both TB and rifampin-resistant TB. Nonetheless, it is advantageous for users to have access to various diagnostic testing options.
The TrueNat MTB Plus assay, developed by Molbio Diagnostics in India, is a newly available molecular test that operates on a portable, battery-powered Truenat platform. Its primary use has been in India. In 2020, the World Health Organization (WHO) recommended the TrueNat MTB and MTB Plus assays [3] as the preferred initial diagnostic tests for tuberculosis (TB), replacing smear microscopy and culture. Additionally, the WHO advised using the Truenat MTBRIF Dx test to detect rifampicin resistance in individuals who test positive with the TrueNat MTB or MTB Plus assays. This testing uses chip-based real-time micro PCR technology to detect M. tuberculosis. The primary TrueNat assay targets the nrdB gene, while the MTB Plus assay focuses on the nrdZ gene and multicopy targets. The MTB-RIF Dx reflex test detects rifampicin resistance by targeting the rpoB gene. All assays utilize the same DNA extract from the Trueprep DNA extraction device, which can process 0.5 mL of sputum in under 20 minutes. The micro PCR device, Truelab, detects M. tuberculosis DNA in approximately 40 minutes. If M. tuberculosis is detected, the reflex test can be performed using the same DNA sample. Both testing devices are portable, battery-operated, and function well at temperatures up to 40°C and humidity levels up to 80%. Our aim to evaluate the Truenat MTB Plus and Xpert MTB-RIF Dx assays using culture and phenotypic drug susceptibility testing as the reference standard.
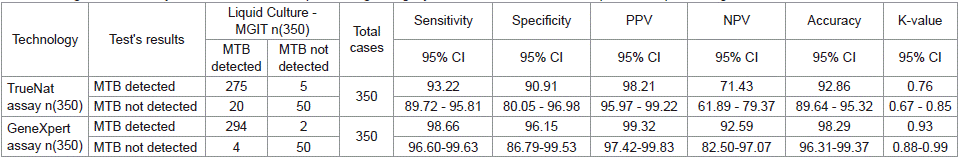
Table 1: Diagnostic accuracy of TrueNat and GeneXpert in diagnosing Mycobacterium tuberculosis in sputum samples taking MGIT as a reference standard.
Methods
Study Design and Settings
This study was a prospective diagnostic accuracy investigation. The primary objective was to evaluate the sensitivity and specificity of the Truenat MTB Plus test in detecting the M. tuberculosis complex, using tuberculosis (TB) culture as the reference standard. The secondary objective was to compare the sensitivity and specificity of the Truenat MTB Plus assay with those of the Xpert MTB/RIF assay. The Indira Gandhi Government General Hospital and Postgraduate Institute Institutional Review Board approved this study (Approval No. GHIEC/2023/102, dated March 8, 2023), which waived the requirement for informed consent. The research adhered to the ethical principles outlined in the Helsinki Declaration, and all data collected were kept confidential.
Sample Collection and Transportation
All participants were instructed to produce two sputum specimens, each with a minimum volume of 4 mL. They were asked to collect the sputum in pre-sterilized 50-mL tubes until this volume was reached. In cases where the sputum volume was insufficient, the specimens were still processed, and testing results were provided to clinicians as usual; however, the participant was considered an early exclusion, and their data were not included in the analysis. In some instances, the first specimen was produced on-site, while the second specimen was produced early the next morning. If a participant was unable to expectorate a spot specimen, then a first-morning specimen would be collected, and if possible, a second specimen would be obtained on the spot or as a second morning specimen. The samples were packed in standard triple packaging with an ice pack to maintain a temperature of 2-8°C and were transported to the Intermediate Reference Laboratory along with a request form.
Sample Reception and Processing
The Intermediate Reference Laboratory at the Government Hospital for Chest Diseases received sputum samples along with their corresponding test request forms. We verified the accuracy of the forms, checked the Nikshay number, labelled the sample tubes, and ensured there were no leaks. Accepted samples were assigned a unique laboratory number and placed in a clean rack for processing. All acceptable samples underwent decontamination using the N-acetyl-L-cysteine-sodium citrate-NaOH (NALC-NaOH) method. Afterward, the homogenized sputum was aliquoted for various tests: 50 mL for direct microscopy, 1.25 mL for culture processing, 0.5 mL for Truenat testing, and 0.5 mL for Xpert testing. The remainder of the sample was stored at -80°C as a backup for any additional testing that might be needed.
MGIT 960 Culture and Drug Susceptibility Testing (MGITDST)
The BACTEC-MGIT-960 test is used to detect the growth of M. tuberculosis in drug-containing and drug-free tubes. The BACTEC MGIT 960 instrument monitors changes in fluorescence to automatically determine susceptibility results, indicated by a green light for no growth and a red light for growth. To confirm the culture for M. tuberculosis, methods such as BHI agar, the Ziehl-Neelsen stain, and the MPT 64 antigen test are employed before conducting Drug Susceptibility Testing (DST) against first-line drugs. The testing process begins by aseptically adding 800 μl of BACTEC MGIT SIRE supplement to five labelled 7 ml MGIT tubes. Next, 100 μl of drug solution is added to four of the tubes, with concentrations as follows: S: 1.0 μg/ml, I: 0.1 μg/ml, R: 1.0 μg/ml, and E: 5.0 μg/ml. In the drugfree tube, 500 μl of Growth Control suspension is added. Each drugcontaining tube then receives 500 μl of a working culture suspension, which is mixed gently. After verifying the order of the tubes, they are loaded into the BACTEC MGIT instrument. The instrument analyses the results, determining if the strains are resistant or susceptible once the growth control reaches 400 units. Afterward, the tubes are removed, and results are manually interpreted as fully susceptible, mono-resistant, poly-resistant, or multidrug-resistant. H37RV controls are included in the process to ensure test quality [4,5].
GeneXpert MTB/RIF Assay
Using a sterile disposable pipette, add GeneXpert sample reagent to each sputum specimen in a 2:1 (v/v) ratio. Shake the container 10 to 20 times, then incubate it at room temperature for 15 minutes, shaking the container once during this period. The sample should be fully liquefied with no visible clumps remaining. Next, label each GeneXpert MTB/RIF cartridge with the corresponding lab accession number. Use the transfer pipette to draw the liquefied sample and transfer it into the open port of the cartridge, ensuring that the cartridge number matches the sputum cup number. Scan the preprinted barcode on the cartridges while the system is powered on and connected. Load the cartridge into the GeneXpert instrument according to the instructions provided. A green light will indicate that the test is in progress. The light will turn off once the test is complete, and the results will print automatically. Wait for the system to release the door lock, remove the cartridge and dispose of it in the biohazard waste container [6,7].
Truenat testing
The Truenat MTB test was conducted following the manufacturer's instructions. All samples were processed according to the Molbio sputum sample pre-treatment protocol. After centrifugation, the supernatant was discarded, and 0.5 mL of the sediment was transferred to the Lysis buffer tube. The homogenized tissue samples and pus aspirates were treated with a liquefaction buffer for 5 to 10 minutes before being transferred to the Lysis buffer tube. The tube was then vortexed and incubated for five minutes. DNA was extracted from the samples using the Trueprep AUTO Universal Cartridge-Based Sample Prep kit and device. The pre-treated sample was transferred to the cartridge's sample chamber and placed in the device, from which, the elute was then aspirated into the Elute Collection Tube (ECT). For the Truenat MTB Real-Time PCR, 6 μL of purified DNA from the ECT was added to a microtube containing freeze-dried PCR reagents. This mixture was then added to the Truenat MTB microchip that contained the lyophilized master mix, and real-time PCR was performed using a pre-programmed profile on the True Lab Analyser [8,9].
Ethical Consideration
The Ethics and Scientific Review Committee at the General Hospital Institute, part of the Directorate of Health and Family Welfare Services in Puducherry, approved this study (No. GHIEC/2023/144, dated 28-96-2023) and granted a waiver for informed consent. All methods were carried out in accordance with the guidelines and regulations set forth by the World Health Organization (WHO) and the National Tuberculosis Elimination Program.
Statistics
The sensitivity, specificity, positive predictive value, negative predictive value, accuracy, and Kappa Value of TrueNat and Xpert assays were calculated using MedCalc for Windows, version 19.4 (MedCalc Software, Ostend, Belgium) [10]. Statistical tests were deemed significant with a p-value of less than 0.05.
Results
Three hundred fifty pulmonary samples were tested using the GeneXpert and TrueNat assays, with the MGIT culture serving as the gold standard. Among the 350 samples tested with the GeneXpert assay, 296 (84.57%) were positive, while 54 (15.43%) were negative. For the TrueNat assay, 280 (80.0%) samples were positive, and 70 (20.0%) were negative. The sensitivity and specificity of the TrueNat test were found to be 89.29% (95% Confidence Interval [CI]: 85.06- 92.65) and 71.43% (156 out of 159; 95% CI: 59.38-81.60), respectively. The positive predictive value was 92.59% (95% CI: 89.60-94.78), while the negative predictive value was 62.5% (95% CI: 53.54-70.68). The overall accuracy of the TrueNat test was 85.71% (95% CI: 81.60- 89.21). In contrast, the GeneXpert test demonstrated a sensitivity of 98.67% (95% CI: 96.60-99.63) and a specificity of 96.15% (95% CI: 86.79-99.53). The positive predictive value for GeneXpert was 99.32% (95% CI: 97.42-99.83), and the negative predictive value was 92.59% (95% CI: 82.50-97.07). The test accuracy of GeneXpert was 98.29% (95% CI: 96.31-99.37). Furthermore, the agreement between the TrueNat assay and GeneXpert compared to the BACTEC-MGIT-960 method for detecting M. tuberculosis in pulmonary samples was excellent, with Kappa values of 0.76 (Standard Error [S.E.]: 0.05) for TrueNat and 0.93 (S.E.: 0.03) for GeneXpert.
Discussion
Drug-resistant tuberculosis (DR-TB) presents a significant challenge to TB control programs worldwide, particularly in low- and middle-income countries. The misuse of anti-TB medications increases the likelihood of developing drug resistance, complicating effective prevention and treatment efforts. Timely and accurate diagnosis of drug-resistant TB is crucial for identifying all infected patients and starting treatment as soon as possible. This approach leads to better health outcomes and helps prevent further disease transmission. The World Health Organization (WHO) [11] emphasizes that determining the most effective treatment regimen for tuberculosis depends on accurate susceptibility testing of Mycobacterium tuberculosis to anti- TB drugs. However, the accuracy of these susceptibility test results can vary based on the specific drug being tested and the method used for testing. In the current study, we evaluated the diagnostic accuracy of the TrueNat assay for detecting Mycobacterium tuberculosis in pulmonary specimens and compared it with the GeneXpert assay.
The sensitivity of GeneXpert was found to be 98.66%, while that of TrueNat was 93.22%, according to the MGIT gold standard. In this study, the sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy of TrueNat for detecting sputum specimens were reported as 93.22%, 90.91%, 98.21%, 71.43%, and 92.86%, respectively. Ngangue YR et al. [12] reported sensitivity and specificity rates for TrueNat in hospitalized patients with pulmonary tuberculosis as 91% and 96%, respectively, indicating lower sensitivity but higher specificity compared to our findings. Similarly, Penn-Nicholson A et al. [9] reported sensitivity and specificity rates of 84% and 95%, respectively, for TrueNat in primary healthcare, again showing lower sensitivity and higher specificity than our study. Urvashi B.S et al. [13] reported even lower sensitivity and specificity rates for TrueNat in primary healthcare at 58.7% and 87.5%, respectively. For GeneXpert, the study found sensitivity, specificity, PPV, NPV, and accuracy for sputum specimens to be 98.66%, 96.15%, 99.32%, 92.59%, and 98.28%, respectively. Rimal et al. [14] reported sensitivity, specificity, PPV, and NPV of the GeneXpert MTB/RIF assay for pulmonary sputum samples as 74.3%, 96.6%, 86.7%, and 92%, respectively. While their report showed lower sensitivity than ours, the specificity aligned closely with our results. In our previous study [6], the GeneXpert MTB/RIF assay for pulmonary tuberculosis had a sensitivity of 99.87%, specificity of 99.92%, PPV of 99.71%, NPV of 99.97%, disease prevalence of 21.38%, and accuracy of 99.91%. Elbrolosy et al. [15] indicated that the sensitivity and specificity for the GeneXpert MTB/RIF assay among pulmonary samples were 72.1% and 81.3%, respectively, which were lower than our findings. The MGIT culture technique, although a gold standard, is comparatively slow and complex, requiring specialized laboratories and skilled staff. Mycobacterium bacilli can take days to grow in liquid media and 4 to 8 weeks in solid media [6].
The Xpert MTB/RIF and TrueNat tests are straightforward and rapid techniques that require minimal training, with results available within a few hours [16]. Setting up an automated liquid culture system for tuberculosis (TB) is less expensive than installing the GeneXpert and TrueNat systems. While GeneXpert is commonly used to identify pulmonary TB from the sputum or bronchoalveolar lavage (BAL) samples, various studies have shown that it can also effectively identify mycobacteria in other body fluids, such as cerebrospinal fluid (CSF), pleural fluid, and ascitic fluid, indicating broader future applications [17]. The current study demonstrates that the performance of TrueNat MTB assays is comparable to that of the GeneXpert MTB/RIF assay for pulmonary samples. Due to the two tests' equivalence and similar performance results, the findings from other GeneXpert MTB/RIF studies can also be applied here. Additionally, the manufacturers provide integrated online connectivity, which could facilitate remote monitoring.
Ethical Approval and Consent to Participate
This retrospective study received approval from the Ethics and Scientific Review Committee of the Indira Gandhi Government General Hospital and Postgraduate Institute, Pondicherry (Approval No. GHIEC/2023/102, dated March 8, 2023), which waived informed consent. The research followed the ethical principles of the Helsinki Declaration, and all data were kept confidential.
Availability of Data and Material
All primary and secondary data are accessible through the corresponding author and the Nikshay portal of the Government of India. While these data are not publicly available, they can be requested from the corresponding author.
Contact no: +91 9944737597.
Email.ID: drmuthurajm@gmail.com
Contributors
All authors contributed to the conception and design of the study. MM, UB, VR, SP, and RMB, all participated in data analysis and interpretation. MM drafted the manuscript, and all authors contributed to revisions and approved the final version.
Acknowledgments
We thank the staff at the Intermediate Reference Laboratory in Puducherry and the medical officers at the TB treatment and primary health centres for their invaluable support during data collection.
References
- World Health Organization. Global tuberculosis report. 2024.
- Automated real-time nucleic acid amplification technology for rapid and simultaneous detection of tuberculosis and rifampicin resistance: Xpert MTB/ RIF assay for the diagnosis of pulmonary and extra pulmonary TB in adults and children: policy update.
- Ssengooba W, Katamba A, Sserubiri J, Semugenze D, Nyombi A, Byaruhanga R, et al. Performance evaluation of Truenat MTB and Truenat MTB-RIF DX assays in comparison to gene XPERT MTB/RIF ultra for the diagnosis of pulmonary tuberculosis in Uganda. BMC Infect Dis. 2024; 24: 190.
- Muthukumar A, Blasundaram RM, Pradhabane G, Kapalamurthy VC, Brammacha¬rry U, et al. Access The Diagnostic Accuracy of Genotypic Assays for The Rapid Detection of Drug–Resistant Mycobacterium Tuberculosis. J Bacteriol Mycol. 2024; 11: 1220.
- Yu H, Kim TY, Kim G, Shim HJ, Kang OK, Kim S, et al. Performance Evaluation of the BACTEC MGIT 960 System for Rifampin Drug–Susceptibility Testing of Mycobacterium tuberculosis Using the Current WHO Critical Concentration. J Clin Microbiol. 2023; 61: e01086–1022.
- Ramachandra V, Brammacharry U, Muralidhar A, Muthukumar A, Mani R, Muthaiah M, et al. Assess the Diagnostic Accuracy of GeneXpert to Detect Mycobacterium tuberculosis and Rifampicin–Resistant Tuberculosis among Presumptive Tuberculosis and Presumptive Drug–Resistant Tuberculosis Patients. Microbiol Res. 2024; 15: 91–108.
- Pradhabane G, Muthukumar A, Blasundaram RM, Prakash V, Vadakunnel MJ, et al. Impact of GeneXpert MTB/RIF Assay for the Detection of Pulmonary and Extrapulmonary Tuberculosis from Clinical Samples. Austin J Public Health Epidemiol. 2025; 12: 1173.
- Inbaraj LR, Daniel J, Rajendran P, Bhaskar A, Srinivasalu VA, Narayanan MKS, et al. Truenat MTB assays for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev. 2023; 1: CD015543.
- Penn–Nicholson A, Gomathi SN, Ugarte–Gil C, Meaza A, Lavu E, Patel P, et al. A prospective multicentre diagnostic accuracy study for the Truenat tuberculosis assays. Eur Respir J. 2021; 58: 2100526.
- MedCalc: MedCalc’s Relative risk calculator. MedCalc Software Ltd, 2024.
- WHO operational handbook on tuberculosis, second edition, Geneva, World Health Organization 2024.
- Ngangue YR, Mbuli C, Neh A, Nshom E, Koudjou A, Palmer D, et al. Diagnostic Accuracy of the Truenat MTB Plus Assay and Comparison with the Xpert MTB/RIF Assay to Detect Tuberculosis among Hospital Outpatients in Cameroon. J Clin Microbiol. 2022; 60: e0015522.
- Ioannidis P, Papaventsis D, Karabela S, Nikolaou S, Panagia M, Raftopoulou E, et al. Cepheid GeneXpert MTB/RIF assay for Mycobacterium tuberculosis detection and rifampin resistance identification in patients with substantial clinical indications of tuberculosis and smear–negative microscopy results. J Clin Microbiol. 2011; 49: 3068–3070.
- Rimal R, Shrestha D, Pyakurel S, et al. Diagnostic performance of GeneXpert MTB/RIF in detecting MTB in smear–negative presumptive TB patients. BMC Infect Dis. 2022; 22: 321.
- Elbrolosy AM, El Helbawy, RH, Mansour OM, et al. Diagnostic utility of GeneXpert MTB/RIF assay versus conventional methods for diagnosis of pulmonary and extra–pulmonary tuberculosis. BMC Microbiol. 2021; 21: 144.
- Lee DJ, Kumarasamy N, Resch SC, Sivaramakrishnan GN, Mayer KH, Tripathy S, et al. Rapid, point–of–care diagnosis of tuberculosis with novel Truenat assay: Cost effectiveness analysis for India’s public sector. PLoS ONE. 2019; 14: e0218890.
- Singh UB, Singh M, Rodrigues C, et al. Multi–centric evaluation of Truenat MTB and MTB–RIF Dx assays for diagnosis of extrapulmonary tuberculosis. Sci Rep. 2024; 14: 15680.