
Editorial
J Cardiovasc Disord. 2014;1(1): 3.
Healthy Harvest, Hale Heart
Arthur J Chu*
Adjunct Professor in Biology, Manhattanville College, Purchase, NY, USA
*Corresponding author: Arthur J Chu, Adjunct Professor in Biology, Manhattanville College, Purchase, NY, USA.
Received: September 27, 2014; Accepted: September 29, 2014; Published: September 30, 2014
Cardiovascular Disease (CVD), a non-communicable disease, presents a group of disorders of the heart and blood vessels; the heterogeneity makes CVD complicated, chronic, and progressive. Common CVD risks (Figure 1 in red) include oxidative stress, hyperlipidemia (e.g., hypercholesterolemia, hypertriglyceridemia), EC dysfunction (e.g., elevated ET-1, AT-II, and TxA2, reduced NO and PGI2), thrombosis (e.g., hypercoagulation, platelet activation/ aggregation, hypofibrinolysis), hypertension, hyperhomocysteinemia, inflammation, diabetes, and obesity. Genetic factors (not shown) often worsen CVD risk.
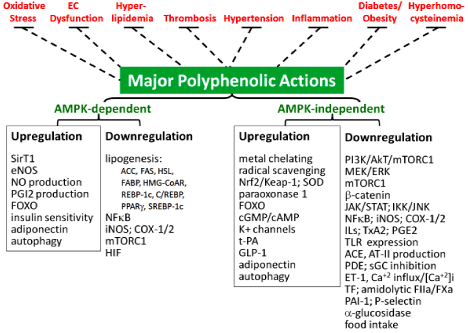
Figure 1: Polyphenols multiply target CVD risks. Polyphenol-induced AMPK
activation leads to SirT1 activation, eNOS activation, FOXO upregulation,
and adiponectin expression accompanied by mTORC1 inhibition, suppressed
lipogenesis, and anti-inflammation. In AMPK-independent signaling,
polyphenols inhibit PI3K/AkT/mTORC1, proinflammatory kinases (IKK, JNK,
JAK/STAT, MEK/ERK), PDE, and ACE as well as blood coagulation and
platelet aggregation. PI3K/AkT inhibition leads to FOXO upregulation and β-
catenin inactivation. Autophagy is activated by either AMPK- dependent or
independent fashion. Blunted dashed lines denote antagonisms.
Changes in lifestyle including diet drastically improve modifiable CVD risks, which is the mostly recommended first line of CVD prevention. In a broader view, food could function as a hormone; nutrients deriving from food can modulate intracellular events to regulate metabolic health by directly and indirectly activating receptors and signaling pathways. Typical nutritional modifications could involve enhanced endothelial NO production (by arginine, antioxidants: CoQ10, lipoic acid, vitamin C/E, glutathione, and eNOS cofactors: B2, B3, BH4, folate), protection from LDL oxidation (by antioxidants, vitamin C/E, β-carotene), lipid lowering (by conjugated linoleic acid, n-3 FAs), and lowered plasma homocysteine (by B6, B12, folate).
Natural bioactive components (e.g., phytochemicals, antioxidants, n-3 FAs, and fibers) in functional foods are cardioprotective. For instance, n-3 FAs (e.g., EPA and DHA) reduce CVD risks and ease myocardial damages, ventricular arrhythmias, atherosclerosis, thrombosis, ischemic heart disease, Myocardial Infarction (MI), and Heart Failure (HF) processes [1]. Improving EC function could be a major mode of action. n-3 FA is able to inhibit the expression of adhesion molecules (e.g., ICAM, VCAM, E-selectin, etc.), proinflammatory cytokines (e.g., TNF, IL-1/2, etc.), thrombomodulin, von Willbrand factor, and PAI-1. By promoting NO production, n-3 FA improves vasodilation. In addition, n-3 FAs exhibit potent hypotriglyceridemic action, raising HDL-C, small reduction in blood pressure, depressed platelet aggregation, anti inflammation, competitive inhibitors for arachidonate metabolism, and reduced susceptibility to LDL oxidation.
There is a perception that dietary fibers generally delay and lower fat absorption. In addition, SCFAs (e.g., butyrate) derived from colonic microbiota (e.g., Lactobacterium) fermentation of dietary fibers are anti-inflammatory [2]. SCFAs could also be associated with anorectic incretin hormone (leptin, PYY, and GLP-1) secretion, which accounts for the roles of gut microbiota and fibers in satiety, anti-diabetes, anti-obesity, and cardioprotection. Nuts are rich in fibers, high quality proteins, and minerals, which is believed to reduce cardiovascular mortality [3]. For instance, MgCl2 is proposed as an inhibitor of HMG-CoA reductase, which could produce a similar effect as statins on cholesterol lowering.
Naturally occurring antioxidants including alkaloids, lycopene, phenolics, vitamin A/C/E, lipoic acid, etc. protect biological components (e.g., DNA, proteins, lipids, etc.) from oxidation. Oxidative stress is known for various pathogenic conditions such as cancers, neurodegeneration, inflammation, CVD, aging, metabolic syndrome, and others. The French paradox certainly underscores the impacts of polyphenolics [4] that are known as antioxidants, AMPK activator, ACE inhibitor, and bioactive phytochemicals with many other biological functions beyond cardioprotection. Lipophilic polyphenolics are classified into three major groups as (1) flavonoids (e.g., catechins (catechin, epicatechin, gallocatechin, and epigallocatechin and their gallates), iso/flavanones, flavanols, and anthocyanins), (2) non-flavonoids (e.g., resveratrol, curcumin, and coumarin), and (3) phenolic (e.g., ellagic, tannic, gallic, and caffeic) acids. They can be widely found in vegetable (green vegetables, turmeric, mustard, nettles, licorice, rhubarb, soy, lentils, etc.), fruits (oranges, grapefruits, lemons, grapes, cocoa, berries, walnuts, kiwis, pomegranates, plums, cherries, apples, mango, etc.), and teas (white, green, and black). They are proposed to generally share GI fat absorption through intestinal lymphatic pathway.
Polyphenols multiply antagonize against common CVD risks (Figure 1). Apart from its classical antioxidation (e.g., radical scavenging, metal chelating, NOX/xanthine oxidase inhibition, and upregulated antioxidant enzymes), polyphenolic cardioprotection is mediated by AMPK activation synergistic with PI3K/AkT inhibition, their diverse downstream events (e.g., SirT1/FOXO upregulation, mTORC1 inhibition), downregulation on proinflammatory kinases, and many other effects on cell signaling.
Anti-Oxidative Stress
Anti-oxidative stress is mainly achieved by the classical antioxidation, which is also ensured by ACE inhibition interrupting AT-II-induced ROS generation and by the anti-inflammatory actions blocking inflammation-oxidation axis.
Anti-Inflammation
Polyphenols target inflammatory components [5] by antioxidant potentials, AMPK activation, inhibitions on PI3K/AkT, mTORC1, IKK/JNK, and JAK/STAT, suppressed HMGB1 release, and TLR suppression. As a result, polyphenols readily lead to NFκB, AP-1, HIF, and STAT inactivation with reduced proinflammatory mediators (e.g., PGE2, cytokines, adhesion molecules, growth factors, etc.). In addition, polyphenol-induced anticoagulation (e.g., TF suppression, inhibited FVIIa/Xa amidolytic activities) and anti-platelet aggregation (e.g., COX inhibition; reduced TxA2) could arrest the coagulationthrombosis- inflammation circuit [6]. Polyphenols also sustain resolution of inflammation (e.g., SirT1 activation, eNOS activation, FOXO upregulation, PDE inhibition, and adiponectin elevation).
Hypolipidemic Actions
Polyphenol-induced AMPK activation suppresses lipogenic transcription factors (e.g., SREBP1/2, C/REBP, etc.) and enzymes (e.g., HMG-CoA reductase, acetyl-CoA carboxylase, etc.) for de novo biosyntheses of cholesterol and fatty acids and TG formation.
Anti-Thrombosis
Polyphenols' anticoagulation, anti-hypofibrinolytic (e.g., downregulated PAI-1, upregulated tPA), and anti-platelet functions readily confer anti-thrombosis. In addition, AMPK-dependent eNOS activation in turn enhances NO bioavailability for protecting platelets from activation and aggregation. The classical antioxidative potentials improve EC function and NO bioavailability. The anti-inflammatory potentials interrupt the coagulation-thrombosis-inflammation circuit [6]. NF?B inactivation and consequent COX inhibition result in TxA2 suppression that is also in line with anti-platelet.
Anti-Hypertension
ACE inhibition targets RAAS and attenuates AT-II-induced oxidative stress, sGC inhibition, and ET-1 elevation, largely presenting anti-hypertension. Improved EC function (e.g., reduced ET-1/ROS, enhanced NO bioavailability, and PGI2 production) exhibits EC-dependent relaxation, while direct K+ channel activation and PDE inhibition lead to EC-independent vasodilation.
Anti-Diabetes
Polyphenols mimic/reinforce physiological insulin action by AMPK-mediated eNOS activation that increases glucose uptake/utilization through Glut-4 translocation. PI3K/AkT inhibition accompanying with JNK inhibition leads to suppressed insulin resistance, while mTORC1 inhibition promotes glycolysis (glucose utilization). While inducing adiponectin and GLP-1 production, polyphenols inhibit α-glucosidase reducing glucose inputs from dietary carbohydrates. Recent insights reveal that flavonoids promote proliferation and anti-apoptosis of pancreatic β-cells. Polyphenols as prebiotics could alter gut microbiota, which elevates circulating GLP- 1 per se as anti-diabetic as well as cardioprotective [7] by reinforcing AMPK activation, eNOS activation, improved EC function, vasodilation, insulin secretion, natriuretic peptide secretion, satiety, etc.
Anti-Obesity
Polyphenols' hypolipidemic effects account for anti-obesity. The consequent SirT1 activation ensures the inhibitions on the genes involved in adipocyte differentiation and TG accumulation. As a consequence of mTORC1 inhibition, suppressed ADD1/SREBP-1c signals are associated with decreased levels of PPARγ as well as C/ EBP a/d mRNA levels during adipogenesis. The AMPK-mediated adiponectin elevation leads to food-intake suppression and weight loss in addition to anti-inflammation. As prebiotics, polyphenols could suppress "obese microbiota" [2].
Anti-Hyperhomocysteinemia
The diverse polyphenolic actions could afford significant counteractions against hyperhomocysteinemia consequences, although limited information is known about the direct effects on homocysteine level.
In summary, polyphenols offer a host of benefits to CVD. Improved EC function (e.g., reduced PAI-1/2, ET-1 attenuation, PDE and ACE inhibition, etc.), hypolipidemic effects, anti-oxidation, anti-inflammation, and anti- hypertensive and thrombotic events readily fight against atherosclerosis, MI, and HF. Polyphenols alleviate hypertrophy pathogenesis by FOXO upregulation (atrophic gene: atrogin-1 expression), PI3K/Akt/ mTORC1 inhibition, β- catenin inactivation, and downregulated [Ca+2]i and its consequent calcineurin-dependent NFAT activation. The exhibited ECdependent or independent VSMC relaxation and improved EC functions also attenuate hypertrophy. In addition to the classical anti-oxidative potentials, the abilities to inhibit ACE, PDE, blood coagulation, PAI-1 production, and platelet aggregation along with K+ channel activation make polyphenols capable of combating arrhythmia and its manifestation (e.g., atrial fibrillation and angina).
It remains an open question whether polyphenols could modify CVD risks through gene regulation. The effects on epigenetic (e.g., DNA methylation, acetylation, Sumoylation, etc.) modifications (e.g., readers, writers, erasers) and micro-RNA expression will provide insights into CVD protection from molecular biology viewpoints. Further confirmatory basic and clinical investigations will promote healthy food for cardioprotection in a nutraceutical era.
References
- De Caterina R. n-3 fatty acids in cardiovascular disease. N Engl J Med. 2011; 364: 2439-2450.
- Clarke SF, Murphy EF, Nilaweera K, Ross PR, Shanahan F, O'Toole PW, et al. The gut microbiota and its relationship to diet and obesity: new insights. Gut Microbes. 2012; 3: 186-202.
- Bao Y, Han J, Hu FB, Giovannucci EL, Stampfer MJ, Willett WC, et al. Association of nut consumption with total and cause-specific mortality. N Engl J Med. 2013; 369: 2001-2011.
- Mohar DS, Malik S. The Sirtuin System: The Holy Grail of Resveratrol? J Clin Exp Cardiolog. 2012; 3.
- Chu AJ. Antagonism by bioactive polyphenols against inflammation: a systematic view. Inflamm Allergy Drug Targets. 2014; 13: 34-64.
- Chu AJ. Tissue factor, blood coagulation, and beyond: an overview. Int J Inflam. 2011; 2011: 367284.
- Ussher JR, Drucker DJ. Cardiovascular actions of incretin-based therapies. Circ Res. 2014; 114: 1788-1803.