
Research Article
Austin J Clin Cardiolog. 2022; 8(3): 1098.
Serum Inflammatory Cytokines and Peritoneal Dialysis in Infants with Acute Renal Failure Following Open Heart Surgery
Liu Y, Mao Y, Ma Y, Zhai M, Li L, Jin P, and Yang J*
Department of Cardiovascular Surgery, Xijing Hospital, Air Force Medical University, Xi’an, China
*Corresponding author: Jian Yang, Department of Cardiovascular Surgery, Xijing Hospital, Air Force Medical University, 127 Changle West Road, Xi’an 710032, Shaanxi, China
Received: October 12, 2022; Accepted: November 11, 2022; Published: November 18, 2022
Abstract
Background: Acute Renal Failure (ARF) is a common complication after open heart surgery with Cardiopulmonary Bypass (CPB) because of the capillary leak syndrome. Peritoneal Dialysis (PD) has been widely used to treat ARF after CPB. In this study, the clearance of inflammatory cytokines of PD in infants with ARF following open heart surgery was investigated.
Methods: Twenty-nine infants with ARF following open heart surgery who underwent PD were divided into survival and nonsurvival groups. Clinical records were reviewed to document clinical features, operative procedures, and perioperative courses. The serum inflammatory cytokine levels, including those of Tumor Necrosis Factor (TNF)-a, interleukin (IL)-6, IL-8, and IL-10, were measured perioperatively.
Results: There was no difference in the preoperative characters between the 2 groups. Longer CPB time, duration of mechanical ventilation, duration from surgery to the beginning of PD, and higher central venous pressure appeared in the nonsurvival group. The serum concentrations of IL-6 and IL-8 were significantly higher in the nonsurvival group than in the survival group before PD and on PD 1 day, PD 4 days, and PD 7 days, respectively. The serum concentrations of IL-10 were higher in the nonsurvival group than in the survival group on PD day 4 and PD day 7, respectively. There were no significant differences of TNF-a between the 2 groups during the perioperative period and PD.
Conclusion: PD could eliminate inflammatory cytokines effectively in patients with mild ARF after open heart surgery.
Keywords: Inflammatory cytokines; Peritoneal dialysis; Acute renal failure; Cardiopulmonary bypass; Infant
Bulleted Clinical Perspectives
Acute renal failure is a common complication after open heart surgery with cardiopulmonary bypass.
In the group with mild acute renal failure, for example, the patients in the survival group, adequate amounts of the inflammatory cytokines could be removed cytokines could be moved from the blood to the peritoneal fluid.
In patients who died, for example, the patients in the non-survival group, the quantity of inflammatory cytokines removed might not be enough or might continue to increase even with Peritoneal dialysis therapy.
Introduction
Acute Renal Failure (ARF) is a frequently reported complication after open heart surgery with Cardiopulmonary Bypass (CPB) in infants. The reported incidence of ARF ranged from 2.4% to 8%. Previous studies have reported a high mortality rate ranging from 30% to 79% [1-3]. According to previous reports, the application of CPB relates directly to the occurrence of ARF. Longer CPB time and longer aortic clamp time corresponded with a higher incidence of ARF and higher mortality [4-6]. A probable mechanism of ARF after CPB is that CPB induced an acute systemic inflammatory response characterized by complement activation, adhesion molecule upregulation, neutrophil activation, and release of the proinflammatory cytokines, which are associated with pulmonary and myocardial dysfunction, neurological injury, and renal failure [3,7].
Peritoneal Dialysis (PD) is frequently used for ARF following open-heart surgery in pediatric patients. Compared to hemofiltration, which has some disadvantages including requirements for systemic anticoagulation and vascular access, PD represents a simple, safe system for fluid removal, especially in newborns and infants. According to some reports, PD resulted in satisfactory outcomes in the therapy of ARF after open heart surgery in pediatric patients [8-12]. Some previous reports also indicated that PD could remove proinflammatory cytokines effectively, including interleukin (IL)-6, IL-8, and IL-10, which could lead to the occurrence and development of ARF [13,14]. However, deaths of ARF after open heart surgery were still high with a range from 32% to 57%, even with PD. Whether removing proinflammatory cytokines by PD could decrease the number of deaths is still under discussion. Our goal was to investigate the impact of PD on cytokine homeostasis and mortality at our institution.
Patients and Methods
Study Population
From July 2018 to May 2022, a total of 29 pediatric patients (body weight <10 kg) who experienced ARF after open heart surgery with CPB in the Xijing Hospital underwent PD. All children were diagnosed by echocardiography, computed tomography scan or angiography, and cardiac catheterization. These examinations assessed cardiac function and associated anomalies. Clinical records were reviewed to document clinical features, operative procedures, and perioperative courses. A total of 15 patients (51.7%) died after PD, and 14 patients (48.3%) were weaned successfully from PD and survived after PD. These patients were divided into 2 groups, survivors and nonsurvivors. This study was approved by the ethics committee of Xijing Hospital and was performed in accordance with the Declaration of Helsinki and all relevant Chinese laws. All participants provided written informed consent from the children’s guardians before inclusion in the study.
Acute Renal Failure and Indications for Peritoneal Dialysis
ARF was defined as a serum creatinine level of more than 1.2 mg/dL or oliguria (< 0.5 mL/kg/hour) for more than 4 hours despite aggressive diuretic therapy and optimization of the inotropic support or a combination of both. The indications for PD were (1) anuria or oligoanuria (<0.5 mL/kg/hour for more than 4 hours) resistant to controlled volume expansion, inotropes, vasodilators, and diuretics; (2) fluid overload; (3) hyperkalemia (> 5.5 mmol/L); (4) metabolic acidosis (serum pH < 7.3, HCO3 < 18 mmol/L); and (5) a serum creatinine level greater than 1.2 mg/dL. Fluid overload was defined as edema, increase of body weight in serial measurements, and difference between fluid uptake and urine output.
Peritoneal Dialysis Procedures
All these patients underwent PD in the intensive care unit postoperatively. In brief, a standard Dacron-cuffed silicone rubber peritoneal catheter (Tenckhoff, Sherwood Medical GmbH, Schwalbach, Germany) was inserted surgically through an infraumbilical midline or a paraumbilical left-sided approach in the intensive care unit when postoperative hemodynamic and renal complications were anticipated according to previous indications for PD.
The PD catheter was connected to a closed system for peritoneal drainage. Standard dialysate solutions were prepared under sterile conditions at our pharmacy. The dextrose concentration varied from 1.5 to 4.5%. A 2.5% solution was used initially. A low-volume dialysis technique was used that included 10 minutes of indwelling time and 20 to 50 minutes of drainage time in one cycle with 10 mL/kg body weight irrigation. The recovery of urine output was defined as urine output > 1 mL/kg/hour, and the recovery of serum creatinine was defined as a decline in serum creatinine to preoperative levels. Indications for stopping PD included a return to a sufficient urine output, maintaining a negative fluid balance, and normalization of the serum electrolyte and acid-base status.
Clinical Data
The data related to age, weight, sex, and diagnosis were recorded for all patients. The Risk Adjustment for Congenital Heart Surgery-1 was used to categorize the surgical intervention. Total duration of CPB, aorta clamp time, and the lowest temperature during CPB were recorded. The perioperative inotropic score for each patient was calculated in accordance with the previously published equation: inotropic score = 1 × dopamine (mg/kg/minute) + 1× dobutamine (mg/kg/minute) + 10 × milrinone (mg/kg/minute) + 100 × epinephrine (mg/kg/minute). The duration of the mechanical ventilation, duration of PD, time to onset of ARF, time interval between the beginning of PD and ARF, incidence of complications during PD, all kinds of hemodynamic data, and other perioperative data were collected. Blood urea nitrogen and serum creatinine levels were measured in the pre- and postoperative periods. Arterial blood gas analysis and electrolyte analysis were performed routinely preoperatively. These clinical data were obtained retrospectively from medical records.
Proinflammatory Cytokines Detections
For all patients who needed PD with ARF after open heart surgery, the venous blood samples (1 mL) were taken under sterile conditions from the patients before the beginning of PD, the first day after PD, and then every 3 days. Added preoperative and postoperative blood samples were centrifugated at 1500 g ×15 minutes at room temperature and at 14000 g ×15 minutes at 4°C. After centrifugation, all serum samples were quickly frozen at -70°C and stored until processed.
Serum proinflammatory cytokine levels, including TNF-a, IL-6, IL-8, and IL-10, were measured by the Enzyme-Linked Immunosorbent Assay (ELISA) technique (Enzyme-Amplified Sensitivity Immunoassay kits, BioSource Europe SA, 8 B-1400, Nivelles, Belgium) according to the manufacturer’s directions. These assays detected only human cytokines, and the minimum detectable concentrations were 5 pg/ml for TNF-a, less than 3 pg/ml for IL-10, less than 2 pg/ml for IL-8, and less than 1 pg/ml for IL-6.
Statistical Analyses
All data were analyzed on the computer using the IBM SPSS Statistics 13.0 for Windows program (IBM SPSS Statistics, Armonk, NY, USA). All data were given as mean ± Standard Deviation (SD). The Χ² test was used to compare differences between the frequencies. Serum cytokine levels were analyzed using the normality test. The Mann- Whitney U test and the Student t-test were used to compare mean values between groups. The Spearman rank correlation test was used for the assessment of correlation. The statistical significance was accepted as P value < 0.05.
Results
All patients were similar in weight and age (Table 1). All patients had congenital heart disease and needed surgical correction. Most patients’ congenital cardiac malformations were more severe than tetra logy of Fallot (Table 2). CPB time was significantly longer in the nonsurvival group than in the survival group. The total number of blood transfusions was significantly higher in the nonsurvival group than in the survival group. All other demographic and operative data are summarized in Table 1; all postoperative data are summarized in (Table 3).
Variables
Survival group
Nonsurvival group
P value
(mean±SD) (n=14)
(mean±SD) (n=15)
Age (months)
8.53±5.79
6.21±3.29
0.2003
Gender (male: female)
7:08
7:07
Body weight (kg)
6.44±2.15
5.95±1.70
0.5032
RACHS-1
2.36±0.63
2.80±0.77
0.1048
Cyanotic
7
7
Operative profiles
CPB time (minutes)
107.07±38.75
140.80±45.05
0.0403
X-aorta time (minutes)
60.36±26.56
78.73±30.22
0.0943
Lowest temperature (°C)
27.36±2.37
25.79±3.73
0.1884
Total blood transfusion (mL)
655.0±137.3
792.0±174.3
0.0269
Fluid output during the operation (mL)
443±47
411±37
0.4252
Total mediastinal drainage after the operation (mL)
112±13
115±21
0.3781
RACHS-1: Risk Adjustment for Congenital Heart Surgery; CPB: cardiopulmonary bypass.
Table 1: The demographic and operative data for the survival group and the nonsurvival group.
Diagnosis
Survivor
Nonsurvivor
VSD ASD PAH
2
1
VSD ASD PDA PAH
3
0
TOF
4
3
TOF PA
0
2
TAPVC
1
2
TGA/IVS
1
1
TGA VSD PAH
0
1
VSD COA PAH
1
2
VSD IAA PAH
0
2
DORV
1
1
Taussig Bing
1
0
Total
14
15
VSD: ventricular septal defect; ASD: atrial septal defect; PAH: pulmonary arterial hypertension; PDA: patent ductus arteriosus; TOF: tetralogy of Fallot; PA: pulmonary atresia; TAPVC: total anomalous pulmonary venous connection; TGA: transposition of the great arteries; TGA/ IVS: transposition of the great arteries with intact ventricular septum; COA: coarctation of the aorta; IAA: interrupted aortic arch; DORV: double outlet right ventricle.
Table 2: Diagnosis of patients in the survival and nonsurvival groups.
Survival group
Nonsurvival group
P
Variables
(n=14)
(n=15)
value
(Mean±SD)
(Mean±SD)
Duration of mechanical ventilation, days
4.43±2.10
8.87±3.31
0.0002
Postoperative inotropic score
5.07±1.98
7. 13±2.33
0.0163
Ventilative time before PD, days
2.00±1.11
3.27±1.62
0.0218
Postoperation to ARF, days
2.43±1.55
3.47±2.03
0.1359
Postoperation to PD, days
3.07±1.73
4.73±2.37
0.0415
Duration of PD, days
11.29±4.25
6.40±3.70
0.0027
Postoperative MAP, mm Hg
70.14±10.08
69.27±11.98
0.8335
Postoperative CVP, cm H2O
7.57±1.79
8.20±1.70
0.3399
MAP before PD, mm Hg
64.07±6.02
59.27±8.58
0.0944
CVP before PD, cm H2O
11.64±1.34
12.67±1.29
0.0454
PD: peritoneal dialysis; ARF: acute renal failure; MAP: mean arterial pressure; CVP: central venous pressure.
Table 3: Postoperative data in the survival and nonsurvival groups.
There was no significant difference in the blood urea nitrogen and serum creatinine levels between the 2 groups until the beginning of PD (p > 0.05). However, the peak values of blood urea nitrogen and serum creatinine were significantly higher in the nonsurvival group than in the survival group (p < 0.05) (Table 4).
Variables
Survival group
Nonsurvival group
P value
(mean±SD) (n=14)
(mean±SD) (n=15)
Preoperative
Blood urea nitrogen (mg/dL)
14.29 ± 6.66
13.33± 3.79
0.6369
Creatinine (mg/dL)
0.86±0.15
0.88 ± 0.16
0.7883
Postoperative
Blood urea nitrogen (mg/dL)
26.07±12.38
27.93±13.19
0.6987
Creatinine (mg/dL)
1.09±0.34
1.04±0.21
0.6666
Before PD
Blood urea nitrogen (mg/dL)
100.07±32.62
108.93±25.97
0.4238
Creatinine (mg/dL)
1.83±0.61
2.33±0.75
0.059
Peak value
Blood urea nitrogen (mg/dL)
117.21±20.22
173.80±65.03
0.0043
Creatinine (mg/dL)
4.35±1.64
7.85±2.04
< 0.0001
PD: peritoneal dialysis.
Table 4: Perioperative blood urea nitrogen and serum creatinine in survival and nonsurvival groups.
Most patients had metabolic acidosis preoperatively because their cardiac malformation led to a critical hemodynamic condition. The pH values were 7.28±0.09 in the survival group and 7.30±0.05 in the nonsurvival group. However, there were no significant differences in blood gas results between the survival and the nonsurvival groups during the perioperative period until the beginning of PD. The metabolic acidosis was corrected in the survival group during PD. In the nonsurvival group, it could not be corrected and got even worse. Bicarbonate, BE, and lactate had similar pH values. At the same time, potassium levels were higher than 5.5 mmol/L in both groups. However, the potassium level in the survival group decreased gradually to the normal range during PD. In contrast, it could not be corrected in the nonsurvival group. The potassium levels were significant higher in the nonsurvival group than in the survival group on PD day 4 and PD day 7. Glucose levels were higher in both groups postoperatively. Then, they were lower because of metabolic acidosis. However, glucose levels were significant lower in the nonsurvival group than in the survival group in the late stage of PD (Figure 1).
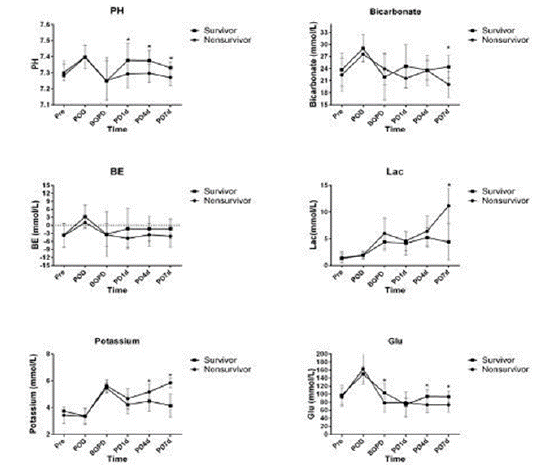
Figure 1: The blood gas results including pH value, bicarbonate, BE, lactate, potassium, and glucose levels in the survival and nonsurvival groups. BE: base
excess; Lac: lactate; Glu: glucose; Pre: preoperative; POD: postoperative day; PD: peritoneal dialysis; BOPD: before PD; PD1d: PD day 1; PD4d: PD day 4; PD7d:
PD day 7.
Serum concentrations of IL-6, IL-8, IL-10, and TNF-a were elevated in all patients preoperatively, postoperatively, before the beginning of PD, on PD day 1, and then every third day. There were no significant differences in preoperative and postoperative serum concentrations of IL-6, IL-8, IL-10, and TNF-a between the survival and the nonsurvival groups. The inflammatory cytokines, in all patients, increased significantly after CPB and in the postoperative period (Figure 2).
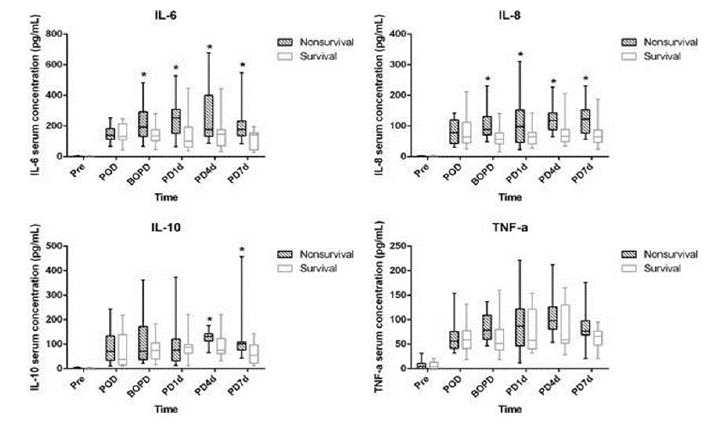
Figure 2: The development of serum concentrations of IL-6, IL-8, IL-10, and TNF-a in survival and nonsurvival groups. IL: interleukin; Pre: preoperative; POD:
postoperative day; PD: peritoneal dialysis; BOPD: before PD; PD1d: PD day 1; PD4d: PD day 4; PD7d: PD day 7; TNF: tumor necrosis factor.
There was no significant difference in the preoperative serum concentration of IL-6 between the survival and the nonsurvival groups. In fact, it was almost undetectable in both groups. Then, they apparently increased postoperatively in both groups. From the beginning of PD, the serum concentrations of IL-6 were always stable at a high level in the nonsurvival group. In contrast, they were decreased in the survival group. Furthermore, they were significantly higher in the nonsurvival group than in survival group before PD and on PD day 1, PD day 4, and PD day 7, respectively (223.80±120.54 and 145.36±66.26 pg/mL, 252.33±130.91 and 150.86±129.73 pg/ mL, 282.33±195.98 and 144.00±102.35 pg/mL, 203.47±112.79 and 114.64±59.20 pg/mL, respectively; p < 0.05)
The serum concentration of IL-8 was similar to that of IL-6. IL-8 was almost undetectable in both groups preoperatively. Then, they apparently increased postoperatively, but there was no significant difference between them. They were always stable at a high level in both groups from the beginning of PD, whereas they were higher in the nonsurvival group than in the survival group before PD and on PD day 1, PD day 4, and PD day 7, respectively (103.13±46.56 and 62.78±33.60 pg/mL, 117.07±85.65 and 67.07±30.18 pg/mL, 122.80±47.84 and 77.42±43.18 pg/mL, 125.93±53.67 and 77.64±52.10 pg/mL, respectively; p < 0.05).
The serum concentrations of IL-10 were also barely detected preoperatively. Then they apparently increased postoperatively in both groups. Then they decreased in the survival group, but they were stable at a high level in the nonsurvival group until PD 7 day (PD4: 128.93±26.22 and 93.21±53.44 pg/mL; PD7: 122.40±97.71 and 61.93±41.89 pg/mL, respectively; p < 0.05)
The preoperative serum concentrations of TNF-a were at a low level in both groups. Then, they increased and remained stable at a high level until PD day 7 in both groups. However, there were no significant differences between the 2 groups during the perioperative period and during PD (p > 0.05).
Discussion
Most operations to correct congenital heart diseases in infants require CPB. Despite improvements in surgical procedures and perioperative care, mortality remains high due to postoperative complications and multiple organ dysfunctions including ARF. The complexity of the underlying heart malformation and of the surgical procedure, the duration of CPB, cardiac arrest, postoperative low cardiac output syndrome, the use of positive inotropic drugs, low body weight, and young age have been shown to be associated with postoperative ARF and death [1,3,8,15].
When treating ARF after CPB, renal replacement therapy is helpful, although the choice remains controversial. Especially for neonates and infants, compared with hemodialysis, PD has been shown to be useful because of the ease of application, the effectiveness of fluid removal, the avoidance of the need for anticoagulation, and the establishment of additional vascular access.
In this study, 51.7% of the patients treated with PD because of ARF after CPB died, which was similar to previous reports of 57.5% by Giuffre et al. (16), of 68.4% by Baskin et al. (4), of 43% by Chien et al. (5), and 40.3% by Boigner et al. (6). Almost half of the patients died of ARF even with PD. What are the reasons for the high number of deaths? According to the report of Giuffre et al. (16), no preoperative, operative, or postoperative variables could be identified to be risk factors between survivors and nonsurvivors after PD. In our study, however, the CPB times were significantly longer in the nonsurvival group than in the survival group, and the patients received more blood transfusions perioperatively in the nonsurvival group. Therefore, the deaths of patients with PD after open heart surgery could be related to the duration of CPB and the total number of blood transfusions administered perioperatively. We also noted longer mechanical ventilation times and longer mechanical ventilation times before PD in the nonsurvival group than in the survival group. The reason could be that the conditions of the patients in the nonsurvival group who could not be separated from the mechanical ventilation were more unstable. On the contrary, most patients in the survival group could be extubated during the early stages of the postoperative period. The higher postoperative inotropic scores also indicated that the conditions of the patients in the nonsurvival group were more serious than those in the survival group. These results were consistent with those in the report by Alkan et al. [9].
Meanwhile, the optimal timing for intervention with PD is still controversial. Many authors have suggested early intervention with PD as soon as ARF occurs. Dittrich et al. [13] used a prophylactic, early start of PD when the risk factors for ARF were present; they reported better control of fluid balance and a more favorable patient outcome, with a mortality rate of 27%. Some authors even suggested that a peritoneal catheter should be placed while the patient is in the operating room if CPB time exceeds 90 minutes and circulatory arrest time exceeds 60 minutes [14]. In this study, there were no significant differences in the duration from the operation to the diagnosis of ARF between the 2 groups. However, the time from the operation to the beginning of PD in the nonsurvival group was longer than that in the survival group. Therefore, dialysis should probably be performed as soon as possible after ARF occurs.
In addition, there were no significant differences in hemodynamic conditions between the 2 groups according to the postoperative mean arterial pressure and central venous pressure. However, the central venous pressure was higher in the nonsurvival group than in the survival group, although there was no significant difference in mean arterial pressure between the 2 groups, which meant that the extent of renal failure could be more serious in the nonsurvival group. Furthermore, there were no significant differences between the 2 groups in urea nitrogen and serum creatinine levels in the early postoperative period. Therefore, urea nitrogen and serum creatinine levels could hardly be used to predict the mortality earlier in patients with PD after open heart surgery. However, the higher peak values in the nonsurvival group meant serious and irreversible renal failure in that group. Continued irreversible metabolic acidosis, hyperkalemia, and hypoglycemia also appeared in the nonsurvival group.
As we know, CPB can induce an acute systemic inflammatory response characterized by complement activation, up-regulation of adhesion molecules, neutrophil activation, and release of the proinflammatory cytokines. This post-CPB inflammatory response is called capillary leak syndrome. With hypothermic CPB, blood flow to organs and tissues declines significantly. Diminished perfusion activates neutrophils and endothelial cells, which decrease the blood flow further in the microcirculation and cause capillary leakage. Then an inflammatory cascade is triggered that can lead to further organ dysfunction. In this process, the increase in the level of proinflammatory cytokines was representative [13]. According to our study, the inflammatory cytokines TNF-a, IL-6, IL-8, and IL-10 in all patients were barely detectable before surgery, although all of the patients developed ARF and had PD postoperatively. However, the levels of both proinflammatory and anti-inflammatory cytokines increased rapidly after open heart surgery with CPB. Also, the serum concentrations of proinflammatory cytokines did not decrease within 24 to 48 hours after surgery, as was reported in other papers [14].
A normally functioning kidney metabolizes filtered proinflammatory cytokines efficiently. Cytokine clearance by the kidney has been hypothesized to be the key to controlling the required balance of proinflammatory and anti-inflammatory responses after CPB. Ordinarily, small proinflammatory cytokines like IL-6 and IL-8 are presented to the proximal renal tubules and absorbed by the proximal tubular cells. In a patient with serious renal dysfunction, proinflammatory cytokines cannot filter normally but reveal a pattern of nonselective mixed glomerular and tubular injury. Further, more serious multiorgan dysfunction occurred. According to the results reported in previous studies [13,14], intraoperative hemodialysis could not only adjust the negative body fluid balance but could also remove proinflammatory cytokines effectively. This technique has been used routinely in our clinic. However, it is rarely performed postoperatively. Postoperatively, dialysis could be an alternative for proinflammatory cytokines clearance except for adjustment of body fluid balance. Bokesch et al. [14] proved that inflammatory cytokines can be removed with peritoneal catheters by measuring the concentrations of serum and peritoneal fluid cytokines.
Removing inflammatory cytokines after CPB may decrease the inflammatory response, which could cause organ dysfunction after open heart surgery with CPB. However, we wondered whether treatment with PD could decrease the number of deaths by removing the inflammatory cytokines? In this study, the serum concentrations of all the proinflammatory cytokines (TNF-a, IL-6, IL-8) and the antiinflammatory cytokines (IL-10) apparently increased postoperatively in both groups. IL-6 and IL-10 were always stable at a high level from the beginning of PD in the nonsurvival group, but they were decreased in the survival group. Also, IL-6, IL-8, and IL-10 were significantly higher in the nonsurvival group than in the survival group on PD day 4 and PD day 7, respectively. Therefore, we deduced the following: (1) Cytokines could be moved from the blood to the peritoneal fluid. (2) In patients who died, for example, the patients in the nonsurvival group, the quantity of inflammatory cytokines removed might not be enough or might continue to increase even with PD therapy. (3) In the group with mild ARF, for example, the patients in the survival group, adequate amounts of the inflammatory cytokines could be removed.
Limitations
Firstly, this study has a retrospective design, and it was not possible to provide missing values in the same way as in a prospective design. Secondly, the number of patients is limited. Thirdly, the patients were seriously ill, and multiple therapies were used. Finally, there were no follow-up results except for the perioperative outcomes. Long-term renal injuries were not clear in the survival group. We recommend that future studies should involve more patients and have a prospective design to further clarify the relationship between PD and inflammatory cytokine clearance and mortality in pediatric patients with ARF after open heart surgery.
Conclusion
During PD, infants developed ARF after open heart surgery. The levels of proinflammatory and anti-inflammatory cytokines in the serum remained stable in both mild and fatal cases of ARF. They decreased in patients with mild ARF in the early stage of PD. They remained stable at a high level instead of deceasing in patients whose ARF proved fatal. Therefore, we deduced that inflammatory cytokines could be removed effectively from patients with mild ARF.
Author Contribution
Yang Liu (Conceptualisation; Writing – original draft preparation); Yu Mao (Writing – original draft preparation); Yanyan Ma (Data curation); Mengen Zhai (Data curation); Lanlan Li (Data curation); Ping Jin (Data curation); Jian Yang (Funding acquisition, Writing – review and editing).
Clinical Trial Registration
ClinicalTrials.gov Protocol Registration System (NCT02917980).
Funding
This work was supported by the National Key R&D Program of China (No. 2020YFC2008100), the Shaanxi Province Innovation Capability Support Plan – Innovative Talent Promotion Plan (No. 2020TD-034).
Conflict of Interest
The authors have no conflicts of interest to declare.
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Ethics Approval
The studies involving human participants were reviewed and approved by Clinicaltrials Organization: Xijing Hospital, Air Force Medical University.
Consent to Participate
The patients/participants provided their written informed consent to participate in this study.
Consent for Publication
Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
References
- Karkouti K, Wijeysundera DN, Yau TM, Callum JL, Cheng DC, et al. Acute kidney injury after cardiac surgery: focus on modifiable risk factors. Circulation. 2009; 119: 495-502.
- Maarslet L, Møller MB, Dall R, Hjortholm K, Ravn H. Lactate levels predict mortality and need for peritoneal dialysis in children undergoing congenital heart surgery. Acta Anaesthesiol Scand. 2012; 56: 459-464.
- Sirvinskas E, Andrejaitiene J, Raliene L, Nasvytis L, Karbonskiene A, et al. Cardiopulmonary bypass management and acute renal failure: risk factors and prognosis. Perfusion. 2008; 23: 323-327.
- Baskin E, Gulleroglu KS, Saygili A, Aslamaci S, Varan B, Tokel K. Peritoneal dialysis requirements following open-heart surgery in children with congenital heart disease. Ren Fail. 2010; 32: 784-787.
- Chien JC, Hwang BT, Weng ZC, Meng LC, Lee PC. Peritoneal dialysis in infants and children after open heart surgery. Pediatr Neonatol. 2009; 50: 275-279.
- Boigner H, Brannath W, Hermon M, Stoll E, Burda G, et al. Predictors of mortality at initiation of peritoneal dialysis in children after cardiac surgery. Ann Thorac Surg. 2004; 77: 61-65.
- Chan KL, Ip P, Chiu CS, Cheung YF. Peritoneal dialysis after surgery for congenital heart disease in infants and young children. Ann Thorac Surg. 2003; 76: 1443-1449.
- Pedersen KR, Hjortdal VE, Christensen S, Pederson J, Hjortholm K, et al. Clinical outcome in children with acute renal failure treated with peritoneal dialysis after surgery for congenital heart disease. Kidney Int Suppl. 2008; S81-S86.
- Alkan T, Akçevin A, Türkoglu H, Paker T, Sasmazel A, et al. Postoperative prophylactic peritoneal dialysis in neonates and infants after complex congenital cardiac surgery. ASAIO J. 2006; 52: 693-697.
- McNiece KL, Ellis EE, Drummond-Webb JJ, Fontenot EE, O’Grady CM, Blaszak RT. Adequacy of peritoneal dialysis in children following cardiopulmonary bypass surgery. Pediatr Nephrol. 2005; 20: 972-976.
- Kist-van Holthe tot Echten JE, Goedvolk CA, Doornaar MB, Vorst MM van, Bosman-Vermeeren JM, et al. Acute renal insufficiency and renal replacement therapy after pediatric cardiopulmonary bypass surgery. Pediatr Cardiol. 2001; 22: 321-326.
- Fleming F, Bohn D, Edwards H, et al. Renal replacement therapy after repair of congenital heart disease in children. A comparison of hemofiltration and peritoneal dialysis. J Thorac Cardiovasc Surg. 1995; 109: 322-331.
- Dittrich S, Aktuerk D, Seitz S, Mehwald P, Schulte-Monting J, et al. Effects of ultrafiltration and peritoneal dialysis on proinflammatory cytokines during cardiopulmonary bypass surgery in newborns and infants. Eur J Cardiothorac Surg 2004; 25: 935-940.
- Bokesch PM, Kapural MB, Mossad EB, Cavaglia M, Appachi E, et al. Do peritoneal catheters remove pro-inflammatory cytokines after cardiopulmonary bypass in neonates? Ann Thorac Surg 2000; 70: 639-643.
- Madenci AL, Thiagarajan RR, Stoffan AP, Emani SM, Rajagopal SK, Weldon CB. Characterizing peritoneal dialysis catheter use in pediatric patients after cardiac surgery. J Thorac Cardiovasc Surg. 2012; 146: 334-338.
- Giuffre RM, Tam KH, Williams WW, Freedom RM. Acute renal failure complicating pediatric cardiac surgery: a comparison of survivors and nonsurvivors following acute peritoneal dialysis. Pediatr Cardiol. 1992; 13: 208-213.