
Case Report
Austin J Clin Case Rep. 2016; 3(4): 1100.
Cerebral Vasculitis in a Middle - Aged Woman with Multiple Sclerosis and Interferon Treatment
Yeh PS¹*, Wu TC², Chen TY² and Chen HA³
¹Department of Neurology, Taipei Medical University, Taiwan
²Department of Radiology, Chi-Mei Medical Center, Taiwan
³Department of Medicine, Chi-Mei Medical Center, Taiwan
*Corresponding author: Poh-Shiow Yeh, Department of Neurology, Chi-Mei Medical Center and Department of Neurology, Taipei Medical University, Taiwan
Received: July 28, 2016; Accepted: September 14, 2016; Published: September 16, 2016
Abstract
Cerebral vasculitis coexisting with multiple sclerosis is very rare. A middle aged woman was diagnosed to have multiple sclerosis from the typical clinical presentations, white matter lesions disclosed on brain magnetic resonance images and cerebrospinal fluid findings. She was treated with interferon to prevent the relapse of multiple sclerosis. The patient began to have various transient neurological deficits with ischemic-like cortical lesions shown on brain images 12 months after receiving the disease-modifying drug. Finally, the case developed bilateral and disseminated cortical ischemic lesions. Extensive cerebral vasculitis was confirmed by cerebral angiography, and there was no clinical evidence of infection and other systemic autoimmune disease. The clinical outcome was poor. Immunomodulating therapy can probably exacerbate or provoke other coexisting autoimmune diseases including vasculitis. The cerebral vasculitis should be considered among multiple sclerosis patients undergoing immunomodulating drug treatment when developing stroke or transient ischemic stroke like events.
Keywords: Cerebral vasculitis, Disease modifying therapy, Immunomodulating drug, Interferon, Multiple sclerosis
Introduction
Multiple Sclerosis (MS) is an inflammatory demyelinating disease involving the central nervous system and characterized by repeated autoimmune attacks of various neurological deficits. Several Immunomodulating Drugs (IMD) including Interferon-β (IFN-β) are commonly used as disease modifying treatment for MS to prevent autoimmune attack and neurological damage. The major problems of IMD therapy in MS are the patient’s poor response to reduce relapse rate and immunotherapy related side-effects such as fever and flulike symptoms. It is well known that different autoimmune disorders can exist in one patient. The most common autoimmune disorders among patients with MS are Hashimoto’s thyroiditis, psoriasis, and inflammatory bowel disease [1]. Cerebral vasculitis coincident with MS is very rare. The coexistence of various autoimmune disorders in the same patient suggests that the patient may carry a general susceptibility to autoimmune dysfunction [1]. In recent years, it has become well-known that IMD can cause immune dysregulation and provoke other autoimmune disease [2-4]. Here we illustrate an unusual clinical experience in which cerebral vasculitis was noted in a MS patient after receiving interferon as the disease modifying therapy.
Case Presentation
A middle aged woman who had been healthy before suffered from acute sensorimotor deficits of the left limbs and recovered soon after conservative treatment at a community hospital although white matter lesions were shown on her Magnetic Resonance Imaging (MRI). Two months later, she developed another episode of acute decline in mental-motor responses and was referred to our hospital. The initial MRI showed multiple white matter lesions in bilateral deep white matter and periventricular regions (Figure 1). The cerebrospinal fluid analysis revealed elevated protein and positive oligoclonal bands. Immunological and infection surveys were normal. Under the clinical diagnosis of Multiple Sclerosis (MS), she received intravenous methylprednisolone pulse therapy and then immunomodulation therapy with subcutaneous INF- β1a. She responded well to these therapies and the follow-up images showed regression of the white matter lesions.
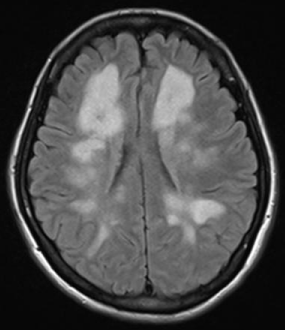
Figure 1: Axial T2 FLAIR images showed multiple white matter lesions at
periventricular white matter, deep white matter and juxtacortical region.
At the 13th month after the MS diagnosis there was an abrupt onset of ataxia, which subsided within one hour. The brain MR Diffusion- Weighted Image (DWI) and Apparent Diffusion Coefficient (ADC) maps showed multiple ischemic lesions with diffusion restriction at bilateral cerebellar vermis and left high frontoparietal cortical regions (Figure 2A); the MR angioraphy showed no narrowing of intracranial vessels (Figure 2B). The T2 Fluid Attenuation Inversion Recovery (FLAIR) image also showed regression of initial white matter lesions at bilateral periventricular regions and corona radiates (Figure 2C). Hematological, infection and cardiac studies were normal. Autoimmune surveys including anti-double strand DNA, antinuclear antibody, rheumatoid factor, anti-RO/SSA, anti- La/SSB, antiphospholipid antibody, and complements titers were unremarkable. Two months later, the patient had another episode of acute limb ataxia involving her right limbs. The MR imaging showed a new ischemic lesion at the right cerebellum (Figure 2D). Aspirin was added under the impression of recurrent ischemic strokes related to uncertain cause.
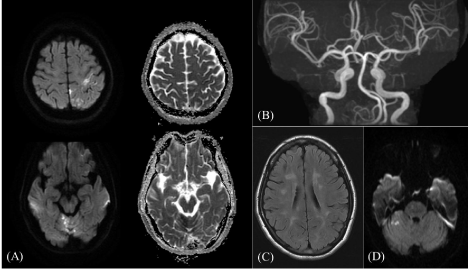
Figure 2: (A) Axial DWI and ADC maps on 13th-month imaging follow-up showed multiple lesions with diffusion restriction at bilateral superior cerebellar vermis
and left high frontoparietal cortical. (B) 3D TOF-MRA showed no significant segmental narrowing of proximal major intracranial vessels. (C) Axial T2 FLAIR image
showed regression of initial MS white matter lesions at bilateral periventricular regions and corona radiata. (D) Axial DWI image on 15th-month imaging follow-up
showed a new hyperintense lesion at right cerebellar hemisphere.
In the following two months, the patient experienced several attacks of reversible neurological deficits manifesting as right limb weakness or drop attack. Finally, she had acute deteriorating bilateral hemiparesis. This time, the MR imaging showed new acute infarcts involving right temporal cortical and subcortical, left occipital cortical and bilateral frontoparietal cortical (Figure 3); the MRA revealed multiple segmental narrowing at bilateral Middle Cerebral Arteries (MCA) branches; the arterial spin labeled perfusion study disclosed hypoperfusion at right temporoparitetal and left occipitoparietal regions. The cerebral angiography disclosed beading alternating with focal segmental narrowing and dilatation of distal branches of bilateral MCA and Posterior Cerebral Arteries (PCAs) which were compatible with cerebral vasculitis (Figure 4). Brain biopsy was refused by the family. Cerebral vasculitis was diagnosed from the typical angiographic findings and the exclusion of other infection and systemic autoimmune diseases. INF- β1a therapy was stopped instantly. Although methylprednisolone and intravenous pulses treatment of cyclophosphamide were administered in order to induce remission of cerebral vasculitis, there was limited clinical response. Unfortunately, the patient expired 3 weeks later.
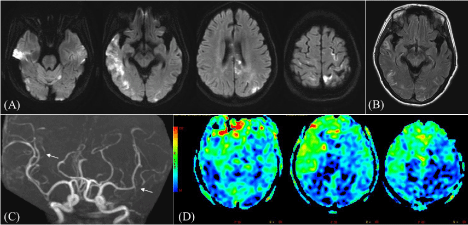
Figure 3: (A) Axial DWI on 21th-month imaging follow-up showed multiple acute infarcts at left superior cerebellar hemisphere, right temporal cortical/ subcortical,
left occipital cortical and bilateral frontoparietal cortical. (B) Postcontrast T2 FLAIR image showed leptomeningeal T2 hyperintensities at bilateral temporooccpital
regions. (C) 3D TOF-MRA showed multiple segmental narrowing (arrows) at bilateral MCA branches. (D) Arterial Spin Labeling (ASL) perfusion study showed
hypoperfusion with diminished Cerebral Blood Flow (CBF) at right temporoparitetal and left occipitoparietal, well correlated with infarction distribution.
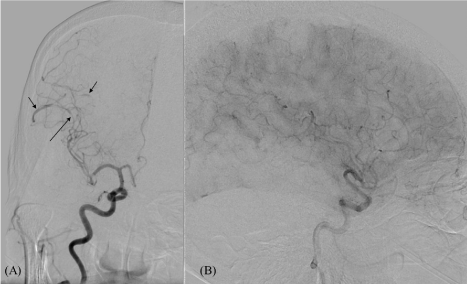
Figure 4: (A) Right ICA injection of conventional angiography showed focal segmental narrowing (long arrows) and dilatation (short arrows) of right MCA branches,
that was characteristic of vasculitis change. (B) Lateral view of right ICA injecion, capillary phase showed perfusion defect at right temporal and right parietal.
Discussion
This middle-aged woman developed clinically manifested cerebral vasculitis after starting interferon therapy. The diagnosis of MS in this case was based on recurrent clinical events, typical CSF findings, disseminated white matter lesions and normal cerebral vessels shown on the brain MR images [5]. The developing cerebral vasculitis was strongly supported by the typical cerebral angiographic findings and the absence of concurrent CNS infection, systemic autoimmune disease and systemic vasculitis, although without histopathological evidence. The cerebral vasculitis progressed while the patient continued the immunomodulating therapy.
It is not rare that two or more autoimmune diseases can coexist in one patient. To the best of our knowledge, the concurrence of cerebral vascullitis and multiple sclerosis is extremely rare. Reviewing the patient’s clinical course, cerebral vasculitis was not likely when patient first came for medical help. The findings on the serial MR images changed remarkably from the initial disseminated white matter patches to the final widespread cortical ischemic lesions. Besides, the major cerebral branches shown on MRA were normal when extensive bilateral white matter patches were present, but there were typical segmental narrowing changes over bilateral MCA branches when disseminated cortical ischemic lesions appeared. Therefore, we speculated the cerebral vasculitis was either provoked or exacerbated by the continuing IMD treatment.
In recent years, the concept of opportunistic autoimmune disease caused by various immunotherapies has been widely discussed [2]. The immunotherapy can exert a paradoxical effect [4]. The use of IMD to reduce autoimmune attacks can also lead into immune dysregulation, and cause other unexpected autoimmune disorders [2,4,6]. The IFN-β is one of the first-line treatments of MS. IFN-β can induce thyroid autoimmunity in up to 19% of the treated MS patients [3,7], and the cerebral vasculitis has rarely been reported. One case of left frontal oligoastrocytoma developing post-operative cerebral vasculitis after intravenous IFN-β injections was reported [8]. There was another case report describing deteriorating Takayasu arteritis in a MS patient with IFN-β therapy [9]. The worsening vascular inflammation was clearly demonstrated by powerful Doppler ultrasound. Bosche et al. described a MS-mimic case without typical CSF findings suffering from worsening cerebral vasculitis after IFN-ß treatment. In this case the authors considered the IFN-β treatment exacerbated the pre-existing and under diagnosed cerebral vasculitis [10].
Cerebral vasculitis is a rare and serious disease and the prognosis depends upon an early diagnosis and treatment. However, the diagnosis of cerebral vasculitis is difficult if there is no tissue proof from cerebral or meningeal biopsies or typical cerebral angiographic findings [11]. The differentiation between multiple sclerosis and cerebral vasculitis is a great challenge because both have similar clinical manifestations as encephalopathy or myelopathy and excellent response to steroid treatment. Reviewing our presenting case, cerebral vasculitis was suspected until the segmental narrowing changes over bilateral MCA branches were shown on the follow-up MRA images. Cerebral vasculitis should be taken into consideration earlier when the patient had repeated vertigo, ataxia or limb weakness and the brain MR-DWI and ADC maps disclosed corresponding ischemic lesions.
Conclusion
Immunomodulating drugs can provoke or exacerbate other autoimmune diseases. Cerebral vasculitis could be coexisted with MS, especially among the cases receiving immunomodulating treatments such as IFN-β.
References
- Barcellos LF, Kamdar BB, Ramsay PP, DeLos C, Lincoln RR, Caillier S, et al. Clustering of autoimmune diseases in families with a high-risk for multiple sclerosis: a descriptive study. Lancet neurology. 2006; 5: 924-931.
- Kong YC, Wei WZ, Tomer Y. Opportunistic autoimmune disorders: from immunotherapy to immune dysregulation. Annals of the New York Academy of Sciences. 2010; 1183: 222-236.
- Theofilopoulos AN, Baccala R, Beutler B, Kono DH. Type I interferons (alpha/beta) in immunity and autoimmunity. Annual review of immunology. 2005; 23: 307-336.
- Axtell RC, Raman C. Janus-like effects of type I interferon in autoimmune diseases. Immunological reviews. 2012; 248: 23-35.
- Polman CH, Reingold SC, Banwell B, Clanet M, Cohen JA, Filippi M, et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Annals of neurology. 2011; 69: 292-302.
- Gota C, Calabrese L. Induction of Clinical Autoimmune Disease by Therapeutic Interferon-a. Autoimmunity. 2003; 36: 511-518.
- Baccala R, Kono DH, Theofilopoulos AN. Interferons as pathogenic effectors in autoimmunity. Immunological reviews. 2005; 204: 9-26.
- Abe T, Sugita K, Morishige M, Ohnishi K, Ishii K, Kamida T, et al. Possible involvement of interferon beta in post-operative vasculitis restricted to the tumour bed: a case report. Neurosurgical review. 2008; 31: 447-450.
- Diamantopoulos AP, Hetland H, Hansen AE, Myklebust G. Severe deterioration of newly diagnosed Takayasu arteritis in a patient re-treated with interferon beta-1alpha for concomitant longstanding multiple sclerosis. Modern rheumatology/the Japan Rheumatism Association. 2012; 22: 474-478.
- Bosche B, Dohmen C, Impekoven P, Zaro-Weber O, Reske D, Rubbert A, et al. Stroke after initiation of interferon-beta treatment for relapsing-remitting disseminated white matter disease. Acta neurologica Scandinavica 2006; 113: 433-436.
- Salvarani C, Brown RD, Hunder GG. Adult primary central nervous system vasculitis. The Lancet. 2012; 380: 767-777.