
Case Report
Austin J Clin Case Rep. 2019; 6(4): 1154.
Meningitis by Listeria monocytogenes in a Milkweed
Sahin R*
Microbiology Laboratory, Mersin City Hospital, Mersin, Turkey
*Corresponding author: Rasim Sahin, Microbiology Laboratory, Mersin City Hospital, Mersin, Turkey
Received: August 23, 2019; Accepted: October 04, 2019; Published: October 11, 2019
Abstract
Listeria monocytogenes may rarely cause meningitis in healthy children. A seven-monthly girl who was previously healthy was admitted to child emergency with complaints of coughing, fever, vomiting and diarrhea. The patient was hospitalized with a preliminary diagnosis of etiology of fever with the purpose of patient examination and treatment. Support treatment was provided. Ceftriaxone treatment was given to the patient with very high fever (39°C). On the third day of hospitalization, fever did not decrease and Lumbal Ponksiyon (LP) was performed because of meningitis suspicious. There was production in cerebrospinal fluid culture. It was defined as Listeria monocytogenes/innocua (98%) with automated bacterial identification system (Phoenix, Becton Dickinson, Sparks, Maryland, USA). The patient’s treatment was revised as Vancomycin, Meropenem. Our case was discharged 3 weeks after hospitalization, having completely recovered.
Keywords: Listeria; Meningitis; Milkweed
Introduction
Listeria monocytogenes are non-spore-forming, aerop and facultative analgesic gram-positive bacilli. Because of its mobility, it is distinguished from the genus Corynebacterium, which causes confusion in diagnosis [1,2]. L. monocytogenes is a bacterium that spreads mainly in macrophages and epithelial cells. It grows in the best neutral or slightly alkaline pH and in a wide temperature range (1-45oC) [3,4]. Listeria is the only gram-positive bacterium with endotoxin. Although there are seven different species of Listeria, it is L. monocytogenes found in humans and leading to disease [4,5]. In general transmission routes; fresh water, salt water, sewage, dust, soil, animal feed, fertilizer and dreaming plants, animals’ feet, animal uncooked foods, fresh and frozen poultry, seafood, red meat and meat products, fish, raw and pasteurized milk, cheese, frozen, cooked sausage, sausage and chicken It contains a large area such as uncooked vegetables and fruits [2-4]. After ingestion of the listeria-contaminated food, the bacteria and the intestine barrier are presumably via M-cells [4]. Lymphatic nodes, the spleen and the liver. L. monocytogenes and L. ivanovii are facultative intracellular pathogens, which can be used for the treatment of macrophages [4,5]. After entry into the cell, Listeria escapes from the host cell in the host cell. The bacteria are usually reported to cause meningitis, meningoencephalitis, bacterium, endocarditis and brain abscess. In addition to virulence of the bacterium, infections are also closely related to the cellular response level of the host [4,6,7]. Therefore, Listeria infection is rare in healthy people in the general population, but in pregnant women, and in the elderly it is quite common [8-11].
Case Presentation
A seven-monthly girl who was previously healthy was admitted to child emergency with complaints of coughing, fever, vomiting and diarrhea. Cough and diarrhea for 3-4 days the patient had a fever for 2 days. The patient was hospitalized with a preliminary diagnosis of etiology of fever with the purpose of patient examination and treatment. Support treatment was provided. Blood values were found as C reactive protein (CRP): 9.99-12.20 mg/dl, white blood cell (WBC): 33.28x103/μl, neutrophil: 24x103/μl, monocytes: 1.82x103/ μl, other hemogram values were normal. Ceftriaxone treatment was given to the patient with very high fever (39°C). Ceftriaxone was given to the patient who was very high fever. On the third day of hospitalization, fever did not decrease and Lumbal Ponksiyon (LP) was performed because of meningitis suspicious. 4-5 leukocytes, 1-2 erythrocytes were seen in all fields in unstained direct microscopy of cerebrospinal fluid (CSF). Cell count was determinate in CSF (Table 1).
RBC, CSF 0 x10^6/Ul
WBC, CSF 0,18 x10^3/Ul
NEU%, CSF 17,8 %
LYM%, CSF 69,4 %
MONO%, CSF 9,2 %
BASO%, CSF 1,2 %
EOS%, CSF 0%
Table 1: Cell count was determinate in CSF.
There was production in cerebrospinal fluid cultures. It was seen beta-hemolytic on sheep blood agar (Figure 1).
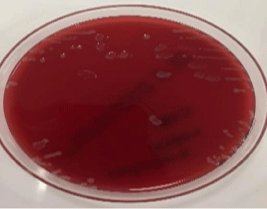
Figure 1: Listeria monocytogenes is beta-hemolytic on sheep blood agar, L.
innocua not.
Gram-positive bacilli was seen in grame stain (Figure 2).
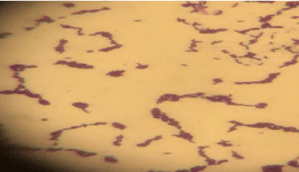
Figure 2: Grame stain.
It was defined as Listeria monocytogenes/innocua (98%) with automated bacterial identification system (Phoenix, Becton Dickinson, Sparks, Maryland, USA). The patient’s treatment was revised as Vancomycin, Meropenem and Clarithromycin.
In CSF biochemistry was determined; glucose 7mg/dl (Simultaneous blood glucose 121mg/dl) as protein: 401gr/dl. Computerize Brain Tomography (CBT) was observed as minimal expansion in ventricles and no subdural effusion. The patient was asked for Brain surgery. The evaluation of the eye from the point of view was recommended. Patient’s eye was dilated. However, no evaluation was made. The patient was consulted with the Pediatric Infectious Diseases Clinic of University Hospital. In addition, shipped by 112 emergency. Our case was discharged 3 weeks after hospitalization, having completely recovered. Early diagnosis and treatment for central nervous system involvement with Listeria are important factors for the prognosis of illness.
Discussion
L. monocytogenes infection is rare in healthy children, difficult to distinguish from clinical and laboratory findings and other infections, and definitive diagnosis is made by isolating the agent from blood or CSF samples. L. monocytogenes should be considered in the differential diagnosis when clinically suspected, when CSF or diphteroid, streptococcus or enterococci are reported in blood culture [3,4].
Although the most common clinical picture is diarrhea, bacteremia, meningitis, brain abscess, endocarditis, osteomyelitis or pneumonia may occur [5-7]. Listeriose was detected as bacteremia most frequently (52%) in immunocompromised patient, while menengitis was seen in healthy patients (64%) diagnosed and reported between the years (1987-2001) in Turkey [6]. L. monocytogenes was most frequently reported as meningitis, meningoencephalitis, spinal cord abscess and brain abscess. Although it ranks 5th among the purulent meningitis in the general population (11-18%), it has the highest mortality rate (22%) [8,9]. It may be acute or subacute as in initial patient. Clinical findings include high fever, neck stiffness, ataxia, and movement disorders such as tremor and seizures. Seizures were more common in L. monocytogenes-related meningitis than in others [6]. Our patient presented with high fever, cough, vomiting and diarrhea. Serological tests are not helpful in diagnosing patients with suspected infection with L. monocytogenes. In 60-75% of the patients with CNS involvement, blood cultures were positive in the early period, while CSF cultures were approximately. The protein level in CSF is generally high, whereas glucose low. In CSF protein and glucose were detected as 401mg/dl and 7mg/dl in our patient.
While 60% had monocytic cell predominance in the direct examination of CSF, the third generation cephalosporin was started considering the bacterial meningitis because of PNL predominance with monocytic cell and nuchal rigidity. Although there was no growth in blood culture in our hospital, the growth of L. monocytogenes was shown in the second CSF culture. CSF Gram staining showed off the presence of gram-positive rods seen in the 2nd CSF sample in our patient.
When compared with the studies abroad, it is reported that there is a high rate of contamination in meat and meat products than in dairy products [13,14].
It is reported that 20-30% L. monocytogenes contamination in meat products used in burgers [15]. Listeria isolations were found to be 5-8% in the researches on cheeses [16]. We think that most of these studies are regional and not sufficient to comment on Turkey’s conditions.
As in our patient, contamination can occur at the production stage (soil, feces, water, workers, tools, materials, etc.), especially in cheese made with raw milk. It is seen that the use of milk and dairy products may pose a significant potential risk to public health if consumed without raw or heat treatment.
It is recommended to pay attention to hygiene in the consumption of contaminated cheese and dairy products, raw chicken, fish, meat and vegetables in patients at risk.
Mortality due to listeria infection is highly variable. Early diagnosis and treatment is very important. The mortality rate is 33-100% in newborns and 22% in Listeria meningitis, whereas it is generally good in patients without pre-dispensing factor [6,12].
As a result, it is important to ensure safe and standardized production with modern hygienic methods in the use of foodstuffs in order to minimize the danger from L. monocytogenes for public health.
We want to emphasize that, although rare, the causative agent of bacterial meningitis that does not respond to appropriate treatment may be L. monocytogenes, and that the history should be questioned well in the diagnosis, and that early diagnosis and causative treatment may prevent complications.
References
- Lorber B. Listeria monocytogenes. In: Mandell Gl, Bennet JE, Dlin r eds. Principles and Practice of Infectious Disease. 5th ed. Phila- delphia: churchill livingstone. 2000: 2208-2215.
- Evirgen Ö. [Clinical, diagnosis and treatment features of infection due to Listeria monocytogenes]. Van Tip Dergisi. 2005; 12: 32-35.
- Heredia N, García S. Animals as sources of food-borne pathogens. Animal Nutrition. 2018; 4: 250-255.
- José A. Vázquez-Boland, Kuhn M, Trinad Chakraborty PB, Domínguez- Bernal G, Goebel W, González-Zorn B, et al. Listeria Pathogenesis and Molecular Virulence Determinants. Clin Microbiol Rev. 2001; 14: 584–640.
- Kessler SL, Dajani AS. Listeria meningitis in infants and children. Pediatr Infect Dis. 1990; 9: 61-63.
- Doganay M. Listeriosis: clinical presentation. FEMs Immunology and Medical Microbiology. 2003; 35: 173-175.
- Sahu RN, Kumar R and Mahapatra AK. Central nervous system infection in the pediatric population. J Pediatr Neurosci. 2009; 4: 20–24.
- DeJesus-Alveloa I and Merendab A. A Case Report of Listeria monocytogenes Abscesses Presenting as Cortically Predominant Ring-Enhancing Lesions. Case Rep Neurol. 2015; 7: 105–109.
- Hernandez-Milian A and Payeras-Cifre A. What Is New in Listeriosis? BioMed Research International. 2014.
- Charpentier E, Gerbaud G, Jacquet c, roco-urt J, courvalin P. Incidence of antibiotic resistance in listeria species. J Infect Dis. 1995; 172: 277-281.
- Julián A, Jiménez A , de Górgolas M, Fer-nández r, Fernández Ml. [Listeria monocytogenes infections in the adult. clinical and microbiological issues of a changing disease]. Enferm Infecc Microbiol clin. 2001; 19: 297-303.
- Tim MW, Jackson MA, shannon k, cohen B, Mccracken GH Jr. Non-neonatal infection due to Listeria monocytogenes. Pediatr Infect Dis. 1984; 3: 213-217.
- Fleming DW, Cochi sl, MacDonald kl, Bron-dum J, Hayes Ps, Plikaytis BD, et al. Paste-urized milk as a vehicle of infection in an outbreak of listeriosis. n Engl J Med. 1985; 312: 404-407.
- Berktas M, Bozkurt E, Bozkurt H, Alisarli M, Güdücüoğlu H. [Isolation of listeria species in meat and meat products]. Van Tip Dergisi. 2006; 13: 36-41.
- Sireli uT, Erol I, Sahin S, Terzi G, Gürbüz oA. [Prevalence and contamination levels of listeria spp. in poultry minced, poultry meatballs and poultry burgers]. Turk J Vet Anim sci. 2002; 26: 1271-1276.
- Kara AA, Algur ÖF, kaya M. [Investigation on the isolation and identification of the Listeria species in the white and civil cheeses purchased from Erzurum region]. Tr J Biology. 1999; 23: 331-337.
- Castellazzi ML, Marchisio P and Bosis S. Listeria monocytogenes meningitis in immunocompetent and healthy children: a case report and a review of the literature. Italian Journal of Pediatrics. 2018; 44: 152.