
Case Report
Austin J Clin Case Rep. 2022; 9(2): 1243.
Sinonasal Angiomyolipoma at Vidian’s Nerve: Case Report with Literature Review
Zahra Aly F¹ and Lobo BC²*
¹Department of Pathology, Immunology and Laboratory Medicine, College of Medicine, University of Florida, Gainesville, FL 32610, USA
²Department of Otolaryngology, College of Medicine, University of Florida, Gainesville, FL 32610, USA
*Corresponding author: Brian C Lobo, Department of Otolaryngology, College of Medicine, University of Florida, Gainesville, FL 32610, USA
Received: February 17, 2022; Accepted: March 11, 2022; Published: March 18, 2022
Keywords
Angiomyolipoma; Vidian Artery; Vidian Nerve; Sinonasal Mass
Introduction
Angiomyolipomas (AMLs) are benign mesenchymal tumors composed of adipose tissue, smooth muscle cells, and blood vessels, in varying proportions. AMLs of the sinonasal cavity and nasopharynx are rare with only 12 cases reported in the English literature (Table 1).
First Author*
Year
Sex
Age
Size (cm)
Location
TSC association
HMB-45
Epistaxis
1
Vinita Pandey
2020
M
54
2.0×1.7× 1.0
Posterior part of the left nasal cavity
NO
Positive
Yes
2
Saiful-Azhar
2019
M
28
4.0×3.0× 2.5
Right lateral wall of the post-nasal space
NO
Negative
Yes
3
Mohammed Abdul Aleem
2017
M
60
0.8 × 0.8
Roof of the left nasal cavity
NO
Negative
4
F
50
2.0× 1.5 × 1.0
Left-sided nasal
NO
Negative
5
Ayaka J. Iwata
2013
M
60s
1.6 × 0.5 × 1.3
Right nasal cavity
NO
NA
Yes
6
Suna Erkiliç
2005
M
52
NA
NA
NO
NA
7
J C Tardío
2002
M
54
1.5 x 1.3 x 1.2
Right nasal cavity
NO
Negative
8
Sankar Banerjee
2001
F
34
2
Left nasal cavity
NO
Positive
9
Kazuo Watanabe
1999
M
66
2
Right nasal cavity
NO
Negative
10
F
88
2
Left nasal vestibule to inferior concha
NO
Negative
11
Zoran Gatalica
1994
NA
NA
NA
NA
NA
NA
12
E E Dawlatly
1988
NA
NA
NA
NA
NA
NA
NA:Information not available from the report; TSC: Tuberous sclerosis.
*References in Appendix to Table 1.
Table 1: Literature review of angiomyolipoma of sinonasal cavity and nasopharynx.
We report on a hereto-undescribed AML emanating from the vidian artery/nerve complex. Specific considerations for surgical approach and the pathologic features of sinonasal angiomyolipoma compared with the classic renal angiomyolipoma are discussed.
Case Presentation
A 76-year-old male presented to the Otolaryngology clinic for an incidental right nasopharyngeal mass. The patient has a history of type 2 diabetes, obstructive sleep apnea treated with continuous positive airway pressure, and polio. Past otolaryngologic surgical history included tonsillectomy. Four months prior, the patient was evaluated for scalp lesions that were diagnosed as sebaceous carcinomas. Resultant screening magnetic resonance imaging (MRI) of the brain revealed an enhancing 2.1 x 1.7 cm mass extending from the anterior choana into the nasopharynx (Figure 1).
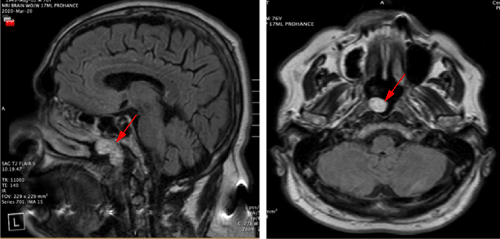
Figure 1: Magnetic resonance imaging of brain showing an enhancing mass
(arrows) extending from the anterior choana into the nasopharynx.
The patient reported recurrent nosebleeds, nasal congestion, and balance disturbance. Endoscopy confirmed a prominent vascular mass occupying the sphenoethmoidal recess and revealed an important finding: two visible vascular stalks extending to the sphenopalatine and posterior septal arteries. The next appropriate step in management was image-guided endoscopic resection under general anesthesia. Intraoperative visualization of the mass showed it to involve the face of the sphenoid, the nasopharynx, the posterior nasal septum, and the right posterior nasal cavity in the area of the vidian artery and nerve complex (Figure 2).
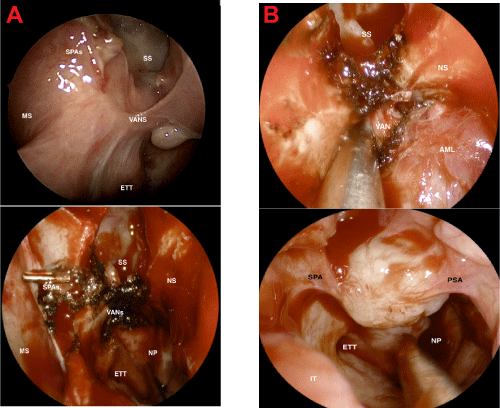
Figure 2: Intraoperative visualization of the mass showed it to involve the
face of the sphenoid, the nasopharynx, the posterior nasal septum, and the
right posterior nasal cavity in the area of the vidian artery and nerve complex
(VANs).
Ligation of the sphenopalatine artery and posterior septal artery were performed, followed by circumferential cuts with a margin along the involved mucosa. Bimanual dissection with a third-hand camera assist technique was carried posteriorly from the anterior vascularized attachments, revealing that the mass was centered on the vidian canal and attached to the vidian artery. Significant high- volume bleeding was encountered while taking the pedicle. This was controlled with endoscopic artery ligation, cautery and hemostatic packing. The patient was admitted overnight due to the intraoperative blood loss and was subsequently discharged on post-operative day 1. The patient’s post-operative course was unremarkable apart from excessive nasal crusting requiring temporarily mupirocin irrigations.
The mass was 2.7cm with reddish gray outer surface and a rubbery texture. The cut surface was pale yellow consistent with adipose tissue. Histologically, the mass was composed of thick-walled vessels set in spindle cells with intervening adipose tissue. Immunohistochemical studies revealed the spindle cells around the vessels to be positive for desmin and smooth muscle actin (Figure 3). S100 highlighted the intervening adipose tissue component while HMB45 and Melan A were negative. The histologic diagnosis of angiomyolipoma was rendered.
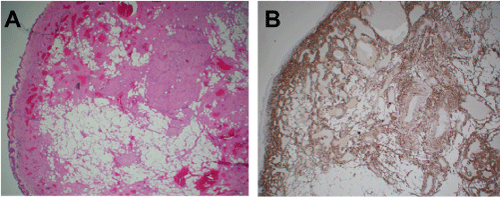
Figure 3: A) Sinonasal mucosa with underlying adipose tissue and thick
muscularized vessels (HE x20). B) Immunohistochemical stain for Desmin
highlights smooth tissue (x200). HMB45 and Melan A stains are negative
(not shown here).
Discussion
Sinonasal AMLs are extraordinarily rare, and this is the first report of an AML originating from the vidian artery/nerve complex. The incidence of all types of AML is about 0.3% [1], with the vast majority occurring in the kidney. First reported by Dawlatly, et al. [2], only eleven other sinonasal AMLs have been reported in the English language literature. AMLs often present with unilateral epistaxis and nasal obstruction and have gender and age predilections as detailed in Table 1. Complete surgical resection is thought to be curative3 unless in the setting of suspected incomplete excision.
AMLs are typically sporadic (80%), with 20% occurring in association with tuberous sclerosis. Of AMLs associated with tuberous sclerosis, 80% occur in the kidney [3]. There are also distinct histologic and clinical differences in the presentation of sinonasal AMLs compared with deep organ AMLs (Table 2). Sinonasal AMLs tend to be smaller and present in older males without associated tuberous sclerosis. In addition, they lack staining for HMB45 and Melan A [4]. Treatment is surgical, with the understanding that long surveillance is for recurrence rather than malignant transformation. There are only two reported cases of AML in the nasal cavity with the same immunophenotype feature of renal AML showing HMB-45 positivity-both, curiously, reported from the Indian subcontinent. In neither case was there an association with tuberous sclerosis.
Deep seated Angiomyolipoma
Mucocutaneous Angiomyolipoma
Sporadic and hereditary (tuberous sclerosis)
Sporadic
Female>male (3:1) (unless hereditary where M=F)
Older males
Can be up to 20cm in size
<4cm
HMB45, Melan A and desmin positive smooth muscle
HMB45 and Melan A negative; Desmin positive smooth muscle
Epithelioid variant associated with metastatic spread
Benign
Variable presence of epithelioid smooth muscle cells
Absence of epithelioid smooth muscle cells
Multiplicity or bilaterality suggests underlying tuberous sclerosis
Solitary
Table 2: Key differences between renal and extra renal angiomyolipomas compared with mucocutaneous angiomyolipoma.
Relevant to the management of nasal masses is the need for imaging and tissue diagnosis. In our case, preoperative imaging demonstrated an enhancing lesion, however its origin from the vidian complex was not evident. Although enhancement is not diagnostic of a highly vascular lesion, when combined in this setting with endoscopy demonstrating two visible arterial pedicles, it became clear that in office biopsy would not be appropriate due to the risk of high volume arterial bleeding. Moreover, this patient’s lesion required bimanual endoscopic techniques for control of high-volume arterial nasal bleeding and dissection-techniques that are typically outside of the armamentarium of an otolaryngologist without specific training. Both of these points merit some discussion.
Both instrument selection and technical preparation were critical to the success of this procedure. While the initial description of endoscopic sphenopalatine and internal maxillary artery control go back almost 30 years, it was not until 1999 that Bolger actually described a reliable landmark, the crista ethmoidalis, for endoscopic SPA ligation [5], and another 10 years until high definition video camera equipment became available. Moreover, for those who have not directly experienced a vascular injury during training, the majority of educational resources describe techniques under optimal conditions.
To counter this, there have been efforts within the last 5 years to create and disseminate vascular injury simulators that provide lifelike bleeding and emotional duress that accompanies this sort of an event14. These simulators are now featured at premier education events (American Rhinologic Society and the North American Skull Base Surgery). Practitioners who have not had first-hand experience dealing with such injuries in residency or fellowship training should strongly consider attending these courses.
Conclusion
Sinonasal angiomyolipomas are an incredibly rare entity, and we present the first reported case arising from the vidian artery/nerve complex. While nasal masses are commonly encountered by the otolaryngologist, appropriate pre-procedural evaluation is essential to prevent clinical complications, and thorough self-assessment of the techniques and equipment required to adequately control high volume endoscopic bleeding should be honest and forthright in order to avoid perioperative complications. Referral to a tertiary or quaternary referral center is not only appropriate, but often desirable.
References
- Lemaitre L, Claudon M, Dubrulle F, Mazeman E. Imaging of angiomyolipomas. Semin Ultrasound CT MR. 1997; 18: 100-114.
- Dawlatly EE, Anim JT, el-Hassan AY. Angiomyolipoma of the nasal cavity. J Laryngol Otol. 1988; 102: 1156-1158.
- Aleem MA, Fatima A, Kumudachalam P, Priyadarshini R. Nasal angiomyolipoma: Report of two cases of an extremely rare entity. Indian J Pathol Microbiol. 2017; 60: 393-395.
- Erkiliç S, Koçer NE, Mumbuç S, Kanlikama M. Nasal angiomyolipoma. Acta Otolaryngol. 2005; 125: 446-448.
- Bolger WE, Borgie RC, Melder P. The role of the crista ethmoidalis in endoscopic sphenopalatine artery ligation. Am J Rhinol. 1999; 13: 81-86.