
Case Report
Austin J Clin Immunol. 2015; 2(1):1026.
Cerebral Amyloid Angiopathy with Asymmetric Vasogenic Edema
Franco-Macías Emilio¹*, Murillo-Espejo Elva¹, Suárez-González Aida², Roldán-Lora Florinda³, Morales-Camacho Rosario María4 and Casado- Chocán José Luis¹
¹Department of Neurology, Hospital Virgen del Rocío, Spain
²Department of Neurodegeneration, University College London, United Kingdom
³Department of Radiology, Hospital Virgen del Rocío, Spain
4Department of Haematology, Hospital Virgen del Rocío, Spain
*Corresponding author: Emilio Franco-Macías, Department of Neurology. Hospital Virgen del Rocío, Avda. Manuel Siurot s/n Sevilla 41003, Spain
Received: November 19, 2015; Accepted: December 01, 2015; Published: December 05, 2015
Abstract
A 76-year-old right handed woman was admitted to hospital because of progressive headache and confusion over the previous week. Two weeks before the admission the patient had suffered a traumatic brain injury after an accidental fall in the street. At that time a brain CT-scan had showed two small hemorrhagic contusions, one in the right frontal lobe and another in the left temporal lobe. After being observed during 24 hours, the patient was discharged without neurological deficit. However, one week later, she started to complain of progressive headache. Upon the second admission the patient showed somnolence and confusion. There were no focal or lateralizing neurological signs. A control CT-scan exhibited an extensive edema involving the right frontal lobe and both temporal and parietal lobes. A brain MRI confirmed asymmetric vasogenic edema and showed multiple subcortical microbleeds on SWI sequence. Blood test ruled out a cause of bleeding. The patient was homozygous for the Apolipoprotein E (APOE) e4 genotype. Cerebral Amyloid Angiopathy-related inflammation (CAA-ri) was diagnosed and the patient was started on intravenous methylprednisolone followed by oral prednisone. She improved rapidly and was asymptomatic at discharge. A control MRI revealed disappearance of the vasogenic edema. We discuss if the joined presence of asymmetric vasogenic cerebral edema, multiple subcortical microbleeds and homozygosis for e4 is enough to diagnose CAA-ri.
Keywords: Cerebral amyloid angiopathy-related inflammation; Microbleeds; Vasogenic edema cerebral; Posterior reversible encephalopathy syndrome
Introduction
Cerebral Amyloid Angiopathy (CAA) is a common pathology in the elderly characterized by the deposition of amyloid proteins within the leptomeningeal and cortical arteries [1]. Lobar hemorrhage is the most common presentation of the disease [1]. A subset of patients who present with seizures, subacute cognitive decline or headaches and show asymmetric vasogenic edema and microbleeds on MRI, and vasculitis or perivasculitis on neuropathologic examination are diagnosed as CAA-related inflammation (CAA-ri) [2].
CAA-ri is interesting because of sharing pathophysiology with amyloid-related imaging abnormalities induced by Aß immunotherapies [3]. The definite diagnosis is reached by cerebral biopsy. However, on clinical grounds the diagnosis could be supported by the characteristic neuroimaging and the presence of allele e4 on APOE genotype without need for biopsy confirmation [4].
We herein report a case of CAA complicated with an extensive cerebral vasogenic edema probably triggered by a traumatic brain injury and diagnosed and treated as CAA-ri with a favorable outcome.
Case Report
A 76-year-old right handed woman was admitted to hospital because of progressive headache and confusion over the previous week. The patient had a medical history of hypertension and hyperlipidemia under treatment. Two weeks before she had suffered a head trauma after an accidental fall in the street. At that time a brain CT-scan had showed two small hemorrhagic contusions, one in the right frontal lobe and another in the left temporal lobe (Figure 1). After being observed during 24 hours without clinical deterioration, the patient was discharged without neurological deficit.
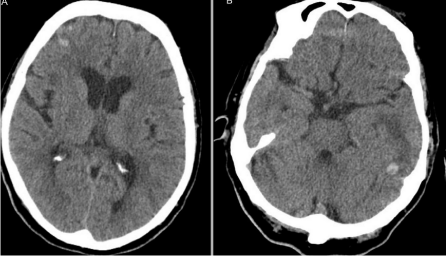
Figure 1: Brain CT-scan at the first admission showing a hemorrhagic
contusion in the right frontal lobe (Figure 1A). There is another hemorrhagic
contusion accompanied by edema in the left temporal lobe (Figure 1B).
A week later, she started to complain of progressive headache. The patient was admitted again. The neurological examination showed somnolence and confusion. Neither meningeal signs nor lateralizing neurological signs was detected. Blood pressure was within normal limits. A control CT-scan showed an extensive edema involving the right frontal lobe and both temporal and parietal lobes (Figure 2). A brain MRI confirmed asymmetric vasogenic edema and showed multiple subcortical microbleeds on SWI sequence (Figure 3). Blood test ruled out a cause of bleeding or cerebral edema. The patient was homozygous for the APOE e4 genotype.
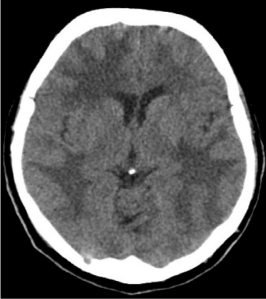
Figure 2: Control brain CT-scan at the second admission two weeks later.
There is an extensive bilateral cerebral edema. In the frontal lobes the edema
predominates in the right one. There is no acute bleeding.
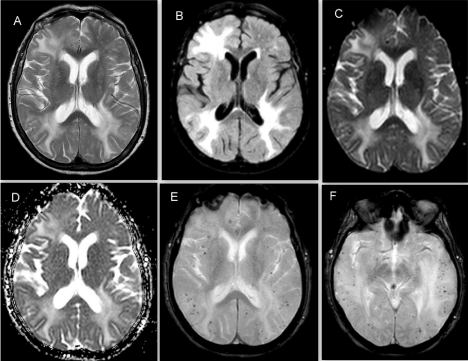
Figure 3: Brain MRI at the second admission. There is an extensive
asymmetric edema in both temporo-parietal areas and in the right frontal lobe.
It is visible as areas of hyperintensity on T2-weighted (Figure 3A), FLAIR
(Figure 3B) and DWI (Figure 3C). The edema is also hyperintense on ADC
map (Figure 3D) confirming it is vasogenic. There are multiple subcortical
microbleeds on SWI (Figure 3E & 3F).
CAA-ri was diagnosed and the patient was started on intravenous methylprednisolone (1 g/daily for 5 consecutive days) followed by oral prednisone which was tapered slowly. At discharge the patient was asymptomatic. A control MRI five weeks later showed disappearance of the vasogenic edema, persistence of microbleeds and signs of mild ischemic leukoencephalopathy (Figure 4). During two years of follow-up there have been no relapses and the patient has returned to his normal lifestyle.

Figure 4: Brain MRI performed five weeks later. The extensive vasogenic
edema has disappeared on T2 (Figure 4A), FLAIR (Figure 4B) and DWI
(Figure 4C) sequences. There is a mild periventricular hyperintensity
consistent with ischemic Leukoencephalopathy mainly visible on FLAIR
sequence (Figure 4B).
Discussion
This elderly woman with multiple subcortical microbleeds and signs of ischemic leukoencephalopathy on brain MRI and homozygosis for the APOE e4 genotype was diagnosed as CAA. The extensive and asymmetric vasogenic cerebral edema, probably triggered by the traumatic brain injury, allowed us to make the diagnosis of CAA-ri. Under steroids treatment the final outcome was favorable. So everything seems to fit. However, some aspects of this case raise questions.
First of all, the head trauma with hemorrhagic contusions seems to have played a role triggering the subsequent vasogenic cerebral edema. CAA-related complications have been associated with different factors. For instance, they have been described in some Alzheimer disease patients included in trials of monoclonal anti-Aβ antibodies suggesting that Aβ immunotherapy would have induced changes in permeability and disruption of CAA-affected vessels [5-7]. On the other hand, a first spontaneous cerebral hemorrhage seems to have been the trigger for vasogenic edema in some cases of CCA-ri [8]. In the reported case, the traumatic brain injury could have caused a breakdown of cerebral autorregulation triggering the vasogenic edema.
The second question is if the sum of clinical symptoms (headache, confusion), asymmetric vasogenic edema, multiple subcortical microbleeds and e4 homozigosis for APOE is enough to make a diagnosis of CAA-ri without performing a biopsy. This decision is important when an immunosuppressive treatment for a long time is under consideration to prevent relapses. Another entity to considerer when neuroimaging shows a vasogenic edema is a Posterior Reversible Leukoencephalopathy Syndrome (PRLS) superimposed on CAA [9,10]. Well-known triggers as renal failure, arterial hypertension or immunosuppressive treatment could lead to the breakdown of cerebral autoregulation. Amyloid-laden vessels would make this breakdown easier. CAA, but not associated vasculitis, has been demonstrated on the neuropathological examination in these cases. Treatment is very different and would be focused on removing the trigger and reduce the edema [9,10].
Conclusion
Cerebral vasogenic edema may complicate cases of CAA. Although CAA-ri must be considered and has been more frequently described, PRLS must also take into account, particularly in presence of a well-known trigger. The treatment is very different. This differential diagnosis should be made before prescribing a long-term immunosuppressive treatment. Cerebral biopsy should be maintained as the gold standard for CAA-ri diagnosis in doubtful cases.
References
- Greenberg SM, Al-Shahi Salman R, Biessels GJ, van Buchem M, Cordonnier C, Lee JM, et al. Outcome markers of clinical trials in cerebral amyloid angiopathy. Lancet Neurol. 2014; 13: 419-428.
- Salvarani C, Hunder GG, Morris JM, Brown RD Jr, Christianson T, Giannini C. Aß-related angiitis: comparison with CAA without inflammation and primary CNS vasculitis. Neurology. 2013; 81: 1596-1603.
- Yamada M. Cerebral amyloid angiopathy: emerging concepts. Journal of Stroke. 2015; 17: 17-30.
- Chung KK, Anderson NE, Hutchinson D, Synek B, Barber PA. Cerebral amyloid angiopathy related inflammation: three case reports and a review. J Neurol Neurosurg Psychiatry. 2011; 82: 20-26.
- Orgogozo JM, Gilman S, Dartigues JF, Laurent B, Puel M, Kirby LC, et al. Subacute meningoencephalitis in a subset of patients with AD after Aß42 immnunization. Neurology. 2003; 61: 46-54.
- Nicoll JA, Wilkinson D, Holmes C, Steart P, Markham H, Weller RO. Neuropathology of human Alzheimer disease after immunization with amyloid-ß peptide: a case report. Nat Med 2003; 9: 448-452.
- Sperling R, Salloway S, Brooks DJ, Tampieri D, Barakos J, Fox NC, et al. Amyloid-related imaging abnormalities in patients with Alzheimer’s disease treated with bapineuzumab: a retrospective analysis. Lancet Neurol. 2012; 11: 241-249.
- Kloppenborg RP, Richard E, Sprengers M, Troost D, Eikelenboom P, Nederkoorn PJ. Steroid responsive encephalopathy in cerebral amyloid angiopathy: a case report and review of evidence for immunosupresive treatment. Journal of Neuroinflammation. 2010; 7: 18.
- Gilo F, Alegre J, Toledano R, García-Villanueva M, Martínez-San Millán J, Martínez-Castrillo JC. Posterior leukoencephalopathy síndrome associated with amyloid angiopathy. Neurología. 2010; 25: 391-397.
- Cano LM, Martínez-Yélamos S, Majós C, Albertí MA, Boluda S, Velasco R, et al. Reversible acute leukoencephalopathy as a form of presentation in cerebral amyloid angiopathy. Neurol Sci. 2010; 288: 190-193.