
Research Article
Austin J Clin Med. 2018; 5(1): 1031.
Effects of Various Commercial Whole-Grain Breads on Postprandial Blood Glucose Response and Glycemic Index in Healthy Subjects
Lanzerstorfer P¹, Rechenmacher E², Lugmayr O¹, Stadlbauer V¹, Höglinger O¹, Vollmar A² and Weghuber J¹*
¹University of Applied Sciences Upper Austria, Stelzhamerstrasse, Austria
²Backaldrin GmbH, Kornspitzstrabe, Austria
*Corresponding author: Weghuber J, University of Applied Sciences Upper Austria, Stelzhamerstrasse 23, A-4600 Wels, Austria
Received: January 16, 2018; Accepted: February 08, 2018; Published: February 15, 2018
Abstract
Postprandial hyperglycemia is a serious risk factor for cardiovascular disease and Type 2 Diabetes Mellitus (T2DM). As diet is a cornerstone of both T2DM prevention and therapy, we investigated the effects of various commercially available breads on postprandial glucose response and glycemic index. Furthermore, the impact of oilseed- and protein-enriched breads was evaluated, as well as the influence of different wheat flour coarseness settings. Three experiments were conducted to study the effects of various breads on the postprandial glucose levels of randomly selected subjects (42 in total). First, putative differences of seven types of commercially available whole-grain breads were studied. Second, the influence of oilseed- and protein-enriched breads was investigated and third, the influence of different wheat flour coarseness settings was characterized. The total Area Under Curve (AUC), postprandial glucose response, peak glucose level and glycemic index were calculated for each condition. Consumption of various types of whole-grain breads resulted in large variations in postprandial glucose response and significant differences in calculated glycemic indices. Furthermore, postprandial glucose levels and glycemic indices appeared significantly lower for protein-rich bread than compared to oil-seed-rich bread. Interestingly, no significant differences in postprandial glucose levels or glycemic indices were detected for fine, medium or coarse wheat flour breads. Based on the correlation study, breads with low total carbohydrates and high amounts of protein are advisable for the prevention of high glucose loads and should be considered by people suffering from T2DM. Trial Registration: O2413 (Ethics Commissions of Upper Austria).
Keywords: Glycemic index; Postprandial glucose response; Coarseness settings; Bread; Diabetes; Blood glucose levels
Abbreviations
ANOVA: Analysis Of Variance; AUC: Area under the Curve; GI: Glycemic Index; T2DM: Type 2 Diabetes Mellitus
Introduction
Bread is one of the most important staple foods worldwide [1]. Therefore, it represents a major source of carbohydrates and, thus, energy in human nutrition, especially in Western civilization. Carbohydrates are key modulators and the main determinants of postprandial glucose response, as the ingestion of carbohydrates is accompanied by an increase in blood glucose levels [2,3]. Carbohydrate-rich foods are classified into three Glycemic Index (GI) categories based on their effect on postprandial glucose response (low <55, medium 55-69 and high <70) [4]. The ingestion of high-GI foods causes excessive postprandial glucose levels and can trigger a disproportionately high insulin response, which is associated with insulin resistance, hyperinsulinemia and, ultimately, the development of Type 2 Diabetes Mellitus (T2DM) [5]. On the contrary, low-GI foods are thought to cause moderate increases in blood glucose concentrations [5]. To prevent increased blood glucose loads, carbohydrates should be predominantly composed of starches, with reduced amounts of mono- and disaccharides. With respect to T2DM, foods and diets should be energy controlled and low GI. In this context, lower GI breads are of particular interest, especially in regions that consume large amounts of bread (e.g., US, Europe) [3]. In recent years, several strategies and approaches to attenuating the GI of bread have appeared, including the inclusion of ingredients with healthful properties. This includes the addition of viscous and non-viscous fibers, legume flours, fruit-based ingredients, specific functional micronutrients and enrichment with intact structures not accessible to human amylase [6-8].
Unfortunately, nutritional characterization of various commercially available breads, including white, whole-meal and seeded breads, has indicated high glycemic properties [9]. However, breads rich in intact grain kernels, oat bran and natural fibers (e.g., beta glucans) have been reported to improve glycemic control and insulin sensitivity [9-11]. Furthermore, they promote satiety via slowed gastric emptying. The ingestion of whole-grain foods in particular has been associated with lower risks of T2DM, cardiovascular disease and weight gain [12,13].
The aims of this study were to i) examine the postprandial glycemic response and GI of seven commercially available whole-grain breads (with white bread as the reference), ii) investigate the influence of fat and protein via oilseed- and protein-enriched breads on postprandial glucose response and GI, and iii) evaluate the role of flour coarseness (fine, medium and coarse) on the selected nutritional parameters.
Materials and Methods
5.1. Study design
The study protocols were approved by the Ethics Commission of Upper Austria (trial number O2413). In total, 42 men (14) and women (28) were included in the study. Study subjects were healthy people of normal weight (mean BMI of 22.5 ± 3.0), with a mean age of 28.1 ± 7.2 years. All study participants provided written informed consent. The first experiment consisted of 15 female and 9 male subjects, the second of 7 female and 1 male subjects and the third of 7 female and 3 male participants. Study subjects were urged to observe the following conditions: Abstention from i) alcohol, ii) fiber-rich foods, iii) medicines, iv) dietary supplements, v) excessive physical exercise and vi) smoking for at least 24 hours prior to each study day. Furthermore, the participants were encouraged not to eat or drink anything except water or unsweetened tea for ten hours before measuring fasting plasma blood glucose levels. Fasting blood samples were collected prior the consumption of the respective test breads (empty stomach). Each test meal was designed to contain 50g available carbohydrates from the test bread ingested over a period of 15min. White bread was used as the reference food. Each subject was served 250mL of drinking water. Blood samples were collected 15, 30, 45, 60, 90 and 120 minutes after bread consumption. The time interval between testing different breads was at least three days.
5.2. Test breads
All test breads were provided by backaldrin GmbH (Asten, Austria). In total, 9 different commercial whole-grain breads were studied in addition to the white bread reference: 1) whole-grain rolls, 2) multi-seed whole-grain bread, 3) whole-grain spelt bread, 4) whole-grain rye bread, 5) whole-grain mixed rye bread, 6) whole-grain oat bread, 7) whole-grain wheat bread, 8) oilseed bread and 9) protein bread. Detailed information about the main ingredients is given in Table 1. Furthermore, the nutrient composition results, as well as the sieve analysis of different coarseness settings, of the tested breads are shown in Tables 2, 3.
Type of bread
Main ingredients
White bread
Wheat flour, water, yeast, wheat sourdough, salt
Whole-grain roll
Wheat flour, water, coarse-grain wheat, coarse-grain rye, yeast, wheat bran, coarse-grain soy, flax seeds, coarse-grain wheat malt, rye sourdough, toasted flour (rye, wheat), rye flour, salt, soy flour, sugar, malted wheat flour, spices, wheat gluten
Multi-seed whole-grain bread
Water, coarse-grains (rye, wheat, oat, barley, buckwheat, sorghum), whole-grain wheat flour, rye sourdough, sunflower seeds, rice, flax seeds, malted rye flour, salt, sugar, oat flakes, yeast
Whole-grain spelt bread
Spelt (whole-grain flour, flakes, sourdough), water, sunflower seeds, rye flour, sugar, potato flakes, salt, yeast, buttermilk powder
Whole-grain rye bread
Coarse-grain rye flour, water, coarse-grain rye sourdough, salt, yeast, sugar
Whole-grain mixed rye bread
Water, whole-grain wheat flour, whole-grain rye flour, sunflower seeds, flax seeds, coarse-grain soy, wheat gluten, sprout mixture (spelt, maize, wheat, sorghum, red clover, alfalfa, flax, lentil, pea), rye sourdough, salt, yeast, sugar, wheat germ, amaranth flour, psyllium seed husks, potato flakes
Whole-grain oat bread
Water, oat flakes, sunflower seeds, spelt flakes, flax seeds, vegetable fibers (psyllium seeds, citric, oat, apple, wheat), pumpkin seeds, sesame, rye, potato starch, salt, wheat germs, rye sourdough, wheat gluten, barley malt extract, acidifiers (lactic acid, acetic acid)
Whole-grain wheat bread
Coarse-grain wheat, water, rye sourdough, salt, wheat gluten, yeast
Oilseed bread
Coarse-grain wheat, water, flax seeds, sunflower seeds, rye sourdough, salt, wheat gluten, yeast
Protein bread
Water, wheat flour, wheat gluten, soy (soy protein, soy flour), potato starch, yeast, salt, rape oil, emulsifier (E481, E472e), dextrose, acidifiers (malic acid)
Table 1: Main ingredients of breads under study.
Energy content
Total fat
Saturated fat
Total carbohydrate
Sugars
Dietary fiber
Protein
Salt (Na x 2.5)
(kJ/100g)
(g/100g)
(g/100g)
(g/100g)
(g/100g)
(g/100g)
(g/100g)
(g/100g)
White bread
990
1.4
0.2
46.6
0.9
3
7.3
1.3
Whole-grain roll
1033
2.4
0.5
42.8
1.4
7.2
9.1
2.3
Multi-seed whole-grain bread
849
3.8
0.5
31.4
2.9
7
7
1.1
Whole-grain spelt bread
1009
5.3
0.7
36.6
4.3
6
8.4
1.4
Whole-grain rye bread
833
1
0.2
37
2.2
7.5
6.3
1.5
Whole-grain mixed rye bread
1036
9.3
1.1
23.6
3
8.9
12.6
1.4
Whole-grain oat bread
995
11.6
1.6
18.6
2.4
11
9.3
1.4
Whole-grain wheat bread
763
1.1
0.2
31.1
0.7
7.7
7.4
1.2
Oilseed bread
990
8.5
0.9
24.7
1.7
9.9
10.1
1.1
Protein bread
889
1.9
0.2
25.6
0.6
3.1
21.1
1.5
Table 2: Nutrient composition of breads under study.
Particle fractions (%)
> 1400 μm
< 1000 μm
< 400 μm
Fine
~ 3
~ 48
~ 43
Medium
~ 75
~ 15
~ 2
Coarse
~ 94
max. 6
---
Table 3: Sieve analysis of milled whole-grain wheat flour.
5.3. Blood glucose determination
Finger-prick capillary samples were collected after fasting and 15, 30, 45, 60, 90 and 120 minutes after bread consumption to measure postprandial glucose levels. Blood glucose concentrations were measured via a blood glucose meter (Accu-Check Performa, Roche Diabetes Care GmbH, Mannheim, Germany).
5.4. Calculations and statistics
The Area under the Curve (AUC) of glucose was calculated for each bread test as the area beneath the curve and above the fasting level from 0 to 120 min using GraphPad Prism 6.0 for Windows (GraphPad Software, Inc., San Diego, CA, USA). The glycemic index was then calculated as follows:
Significance testing was performed using Graph Pad Prism 6.0 for Windows. Differences were considered significant with p < 0.05 for t-tests and two-way ANOVA. All values are given as the means ± SE if not otherwise stated.
Results
Impact of commercially available whole-grain breads on postprandial glucose response and glycemic index
Before consumption of the different test breads, the basal fasting glucose levels of subjects were examined, which resulted in a mean glucose level of 5.1 ± 0mmol/L, and were comparable for each test day. Significant differences between the different test breads and the white bread reference were found in postprandial glucose response at various points (Figure 1A). The largest mean AUC was detected for the whole-grain roll (178.2 ± 28.8mmol/L), which even exceeded the mean AUC for the white bread reference (161.5 ± 14.4mmol/L). The smallest mean AUC was calculated for whole-grain oat bread (82.2 ± 9.6mmol/L). Plasma glucose levels reached peak values (Figure 1B) between 30 and 45 minutes after the ingestion of test breads and decreased slowly thereafter. Significantly decreased peak blood glucoses levels were found for whole-grain rye bread (7.2 ± 0.2mmol/L), whole-grain mixed rye bread (7.2 ± 0.2mmol/L) and whole-grain oat bread (6.5 ± 0.2mmol/L) compared to white bread (7.9 ± 0.2mmol/L). Based on mean AUC values, GI was calculated for each test bread (Figure 1C). Again, significantly lower GI values were obtained for whole-grain mixed rye bread (67.2 ± 11.2%) and whole-grain oat bread (55.3 ± 10.1%) compared to white bread (100%). Interestingly, whole-grain rye bread, which exhibited a lower peak glucose level, did not show a significant difference (90.2 ± 8.6%). However, multi-seed whole-grain bread, which showed a higher peak glucose level (7.4 ± 0.3mmol/L), was found to have a significantly lower GI (70.7 ± 7.0%). Based on these data, we conclude that peak blood glucose level is appropriate for estimating the GI of different breads, although some exceptions were detected in our study.
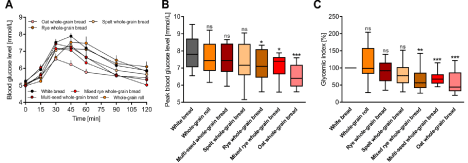
Figure 1: Comparison of (A) mean fasting and postprandial glucose responses over 120min, (B) mean peak blood glucose levels and (C) calculated glycemic
indices after ingestion of indicated breads. Values are presented as the means ± SE; n = 24. ns, not significant, * p < 0.05, ** p < 0.01 and *** p < 0.001, significantly
different from the positive control (white bread).
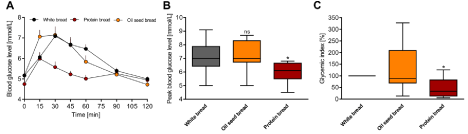
Figure 2: Influence of ingested oilseed- and protein-enriched breads on (A) mean fasting and postprandial glucose responses over 120min, (B) mean peak blood
glucose levels and (C) calculated glycemic indices. Values are presented as the means ± SE; n = 10. ns, not significant and * p < 0.05, significantly different from
the positive control (white bread).
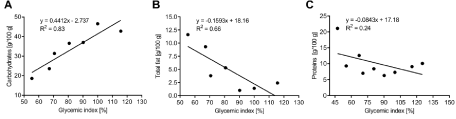
Figure 3: Linear correlation regression analysis. Correlations between (A) total carbohydrate and GI, (B) total fat and GI and (C) protein and GI.
6.1. Investigation of the influence of oilseeds and proteins
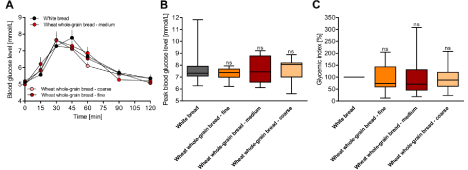
Figure 4: Influence of coarseness (fine, medium and coarse) of whole-grain wheat flour on (A) mean fasting and postprandial glucose responses over 120min, (B)
mean peak blood glucose levels and (C) calculated glycemic indices. Values are presented as the means ± SE; n = 8. ns, not significantly different from the positive
control (white bread) and within indicated the coarseness setting.
Whole-grain oat bread exhibited the lowest peak blood glucose level and GI. As they are especially rich in total fat (11.6g/100g), protein (9.3g/100g) and dietary fiber (11.0g/100g) compared to other breads under study, we investigated the influence of oilseed- and protein-enriched breads on the measured parameters. The oilseed bread was especially rich in fat (8.5g/100g) and dietary fiber (9.9g/100g), whereas the protein bread had lower fat content (~4-fold) but was significantly higher in protein (21.1g/100g, 2-fold higher than the oilseed bread). These breads had comparable levels of total carbohydrates. The detailed nutrient composition of the oilseed and protein breads can be found in Table 1. Significant differences in postprandial glucose response, peak blood glucose levels and GI were found for the protein bread but not for the oilseed bread (Figure 2A-C).
6.2. Influence of macronutrients on GI
To determine the most influential nutritional parameters on the GI, linear regression and correlation analyses were carried out for all breads under study (Figure 3A-C). We found a clear positive correlation between total carbohydrate and GI (R value 0.83; p < 0.01), as well as an inverse correlation between total fat and GI (R value 0.66; p < 0.05). No significant correlation was found between protein content and GI (R value 0.24; p = 0.17). Therefore, high protein and fat content in combination with a reduced carbohydrate level seems to favor a lower GI.
6.3. Determination of the effect of different coarseness
There is evidence that the coarseness of flour has an impact on postprandial glucose response and directly influences the glycemic index of baked goods [14,15]. In the last part of this study, we investigated the effect of fine, medium and coarse wheat flour in whole-grain bread. The results of the sieve analysis and the respective particle fractions are shown in Table 3. Fine whole-wheat flour consisted of at least ~48% grain smaller than 1mm and ~43% smaller than 0.4mm. Medium whole-wheat flour consisted of ~75% wheat grains larger than 1.4mm and ~15% smaller than 1mm, and coarse whole-wheat flour had approximately 94% wheat grain particles larger than 1.4mm. To our surprise, we did not detect significant differences in postprandial glucose response, peak blood glucose levels or GI across the different coarseness settings (Figure 4A-C).
Discussion
As bread is consumed all over the world, variations in processing techniques and ingredients are manifold. Consequently, bread’s nutritional composition, energy content and GI values vary considerably [16]. In the present study, we evaluated the effects of different commercially available whole-grain breads (whole-grain roll, multi-seed whole-grain bread, whole-grain spelt bread, whole-grain rye bread, whole-grain mixed rye bread, whole-grain oat bread, whole-grain wheat bread, oilseed bread and protein bread) in comparison to white bread on postprandial glucose response, peak blood glucose levels and GI after standardized carbohydrate consumption.
As expected, the main ingredients and, thus, the nutritional composition of whole-grain breads under study varied considerably (Tables 1, 2) and led to significant differences in the measured parameters. The highest postprandial glucose response, which was even higher than that of white bread, was induced by the whole-grain roll. Except for whole-grain oat bread, all tested breads promoted postprandial hyperglycemia with peak blood glucose levels above 7mmol/L (Figure 1B). In addition to the whole-grain oat bread, whole-grain rye bread and whole-grain mixed rye bread showed significantly lower peak blood glucose levels compared to the reference bread. Surprisingly, the calculated GI values for whole-grain rye bread were not significantly different from those of white bread (100%), whereas the GI values of whole-grain mixed rye bread, whole-grain oat bread and multi-seed whole-grain bread were significantly lower.
In general, products based on whole-grain rye have been reported to modulate postprandial insulin secretion and blood glucose profile [17-20] and to induce higher subjective satiety [21-23]. So far, the underlying regulatory mechanisms have not been elucidated, but several studies suggest that the effects are related to the high fiber content of rye, which might influence the glucose absorption rate and hormonal response through decreased nutrient diffusion rates [24]. In addition, structural features of rye products might inhibit starch hydrolysis through the formation of an amylose layer around starch granules, thereby inhibiting glucose uptake [17]. The whole-grain oat and multi-seed whole-grain breads are especially rich in dietary fibers that are reported to have positive effects on glucose metabolism by reducing the pH of the intestinal tract, consequently inhibiting starch digestion and delaying gastric emptying. Furthermore, intact fiber structures are not accessible to human amylases, and conversion into monosaccharides might be reduced [7]. In addition, the whole-grain oat bread was the most protein- and fat-enriched bread with the lowest energy content of all the commercially available breads included in the first study. To identify the macro-nutritional parameters that reduce GI in more detail, protein and oilseed breads were investigated in the second stage of this study. Compared to the white bread control, only the protein bread exhibited significantly reduced peak blood glucose level and glycemic index. The oilseed bread had comparable amounts of total fat, carbohydrates, dietary fiber and protein with respect to the identified low GI whole-grain oat bread. As the energy content of these two breads is comparable, this finding is quite surprising. One explanation could be that whole-grain oat bread is rich in non-cereal fibers (e.g., psyllium seeds, citric and apple) and cereals (oat, wheat) (Table 2). Soluble dietary fiber, which mainly occurs in legumes but is also found in oats, may have direct or indirect effects on reduced glucose reabsorption and thus on the glycemic index of the respective bread [7]. On the contrary, the protein content of the protein-enriched bread is twice as high as that of the oilseed bread, whereas the oilseed bread possesses a fat content four times higher and fiber content three times higher than the protein-based bread. The results suggest that oilseeds do not have any beneficial effects on glycemic index. On the contrary, protein is a parameter that can be increased to successfully reduce the GI of bread. Moreover, it is known that the natural gluten content of some cereals can limit starch bioavailability and enzyme accessibility [3]. However, as revealed by linear regression and correlation analyses, carbohydrate content had the most prominent effect on GI, followed by total fat and protein content. These results indicate that postprandial glucose response and, therefore, GI are determined by a multitude of nutritional parameters rather than by a single contributing factor. Moreover, the individual responses of subjects have to be taken into account, as large variations were observed for all analyzed parameters (as indicated by the large ranges in the respective figures). Finally, the effects of flour coarseness on postprandial glucose response and GI were investigated. Epidemiological data consistently suggest an inverse relation between the consumption of whole-grain foods and the risk of coronary heart disease and, possibly, of T2DM [25-28]. As whole-grain breads are mostly made with milled flour of small particle size, we examined breads baked with fine, medium and coarse whole-grain wheat flour with comparable nutritional composition. Studies showed that milled cereals as well as rice exhibit a higher glycemic index as in its original shape, as starch hydrolysis is facilitated [14,15,29]. We therefore expected differences in postprandial glucose response and peak blood glucose levels. To our surprise, we did not find a significant effect of the coarseness of the tested whole-grain wheat flours on our selected parameters (Figure 4). Again, the study found that some individuals are more sensitive to the milling degree than others. A possible explanation for these results is that the particles in the coarse flour were still too small and contained starch that could be digested, as in bread baked with finely milled whole-grain wheat flour.
Conclusion
Whole-grain products are reported to have several positive effects on human health [30], including reducing coronary heart disease [26,31,32], T2DM [25,27,33] and metabolic syndrome [7,34]. This study reports large variations in the impacts of various whole-grain-based breads on relevant T2DM-related parameters and indicates complex nutritional relationships and individual response characteristics. Therefore, it is difficult to provide recommendations for effective food-based dietary guidance for patients. In general, bread with reduced carbohydrate content in combination with high fiber, protein and fat content seems to be preferable, but dietary optimization for each individual is necessary.
Declarations
9.1. Ethics approval and consent to participate
This study was approved by the Ethics Commission of Upper Austria (Austria, O2413). Prior to the study, each subject received a full written and oral explanation of the purpose and procedures of the study, and written informed consent was obtained from each subject.
Competing Interests
The authors declare that they have no competing interests. backaldrin GmbH provided test breads and detailed nutritional information but did play any role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Authors’ Contributions
Conceived and designed the study: AV, OH, PL, JW. Performed the study: ER. Analyzed the data: OL, PL, VS. Wrote the paper: PL, JW. Supervised all aspects of this work: JW. All authors drafted and approved the final manuscript.
References
- Wirfalt E, McTaggart A, Pala V, Gullberg B, Frasca G, Panico S, et al. Food sources of carbohydrates in a European cohort of adults. Public health nutrition. 2002; 5: 1197-1215.
- Lafiandra D, Riccardi G, Shewry PR. Improving cereal grain carbohydrates for diet and health. Journal of cereal science. 2014; 59: 312-326.
- Stamataki NS, Yanni AE, Karathanos VT. Bread making technology influences postprandial glucose response: a review of the clinical evidence. The British journal of nutrition. 2017; 117: 1001-1012.
- Augustin LS, Kendall CW, Jenkins DJ, Willett WC, Astrup A, Barclay AW, et al. Glycemic index, glycemic load and glycemic response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutrition, metabolism, and cardiovascular diseases. NMCD. 2015; 25: 795-815.
- Jenkins DJ, Kendall CW, Augustin LS, Franceschi S, Hamidi M, Marchie A, et al. Glycemic index: overview of implications in health and disease. The American journal of clinical nutrition. 2002; 76: 266S-273S.
- Grajek W, Olejnik A, Sip A. Probiotics, prebiotics and antioxidants as functional foods. Acta biochimica Polonica. 2005; 52: 665-671.
- Scazzina F, Siebenhandl-Ehn S, Pellegrini N. The effect of dietary fibre on reducing the glycaemic index of bread. The British journal of nutrition. 2013; 109: 1163-1174.
- Stamataki NS, Yanni AE, Karathanos VT. Non-cereal ingredients for the attenuation of glycaemic response to bread: a review of the clinical evidence. Food & function. 2016; 7: 2926-2936.
- Atkinson FS, Foster-Powell K, Brand-Miller JC. International tables of glycemic index and glycemic load values: 2008. Diabetes care. 2008; 31: 2281-2283.
- Jenkins DJ, Wolever TM, Jenkins AL, Giordano C, Giudici S, Thompson LU, et al. Low glycemic response to traditionally processed wheat and rye products: bulgur and pumpernickel bread. The American journal of clinical nutrition. 1986; 43: 516-520.
- Pick ME, Hawrysh ZJ, Gee MI, Toth E, Garg ML, Hardin RT. Oat bran concentrate bread products improve long-term control of diabetes: a pilot study. Journal of the American Dietetic Association. 1996; 96: 1254-1261.
- Pereira MA, Jacobs DR, Pins JJ, Raatz SK, Gross MD, Slavin JL, et al. Effect of whole grains on insulin sensitivity in overweight hyperinsulinemic adults. The American journal of clinical nutrition. 2002; 75: 848-855.
- Ye EQ, Chacko SA, Chou EL, Kugizaki M, Liu S. Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease, and weight gain. The Journal of nutrition. 2012; 142: 1304-1313.
- Jenkins DJ, Wesson V, Wolever TM, Jenkins AL, Kalmusky J, Guidici S, et al. Wholemeal versus wholegrain breads: proportion of whole or cracked grain and the glycaemic response. BMJ. 1988; 297: 958-960.
- Liljeberg H, Granfeldt Y, Bjorck I. Metabolic responses to starch in bread containing intact kernels versus milled flour. European journal of clinical nutrition. 1992; 46: 561-575.
- Fardet A, Leenhardt F, Lioger D, Scalbert A, Remesy C. Parameters controlling the glycaemic response to breads. Nutrition research reviews. 2006; 19: 18-25.
- Juntunen KS, Laaksonen DE, Autio K, Niskanen LK, Holst JJ, Savolainen KE, et al. Structural differences between rye and wheat breads but not total fiber content may explain the lower postprandial insulin response to rye bread. The American journal of clinical nutrition. 2003; 78: 957-964.
- Leinonen K, Liukkonen K, Poutanen K, Uusitupa M, Mykkanen H. Rye bread decreases postprandial insulin response but does not alter glucose response in healthy Finnish subjects. European journal of clinical nutrition. 1999; 53: 262-267.
- Rosen LA, Ostman EM, Bjorck IM. Postprandial glycemia, insulinemia, and satiety responses in healthy subjects after whole grain rye bread made from different rye varieties. 2. Journal of agricultural and food chemistry. 2011; 59: 12149-12154.
- Rosen LA, Silva LO, Andersson UK, Holm C, Ostman EM, Bjorck IM. Endosperm and whole grain rye breads are characterized by low post-prandial insulin response and a beneficial blood glucose profile. Nutrition journal. 2009; 8: 42.
- Isaksson H, Fredriksson H, Andersson R, Olsson J, Aman P. Effect of rye bread breakfasts on subjective hunger and satiety: a randomized controlled trial. Nutrition journal. 2009; 8: 39.
- Isaksson H, Rakha A, Andersson R, Fredriksson H, Olsson J, Aman P. Rye kernel breakfast increases satiety in the afternoon - an effect of food structure. Nutrition journal. 2011; 10: 31.
- Isaksson H, Tillander I, Andersson R, Olsson J, Fredriksson H, Webb DL, et al. Whole grain rye breakfast - sustained satiety during three weeks of regular consumption. Physiology & behavior. 2012; 105: 877-884.
- Johansson DP, Gutierrez JLV, Landberg R, Alminger M, Langton M. Impact of food processing on rye product properties and there in vitro digestion. European journal of nutrition. 2017.
- Fung TT, Hu FB, Pereira MA, Liu S, Stampfer MJ, Colditz GA, et al. Whole-grain intake and the risk of type 2 diabetes: a prospective study in men. The American journal of clinical nutrition. 2002; 76: 535-540.
- Jacobs DR, Meyer KA, Kushi LH, Folsom AR. Whole-grain intake may reduce the risk of ischemic heart disease death in postmenopausal women: the Iowa Women's Health Study. The American journal of clinical nutrition. 1998; 68: 248-257.
- Liu S, Manson JE, Stampfer MJ, Hu FB, Giovannucci E, Colditz GA, et al. A prospective study of whole-grain intake and risk of type 2 diabetes mellitus in US women. American journal of public health. 2000; 90: 1409-1415.
- Sahyoun NR, Jacques PF, Zhang XL, Juan W, McKeown NM. Whole-grain intake is inversely associated with the metabolic syndrome and mortality in older adults. The American journal of clinical nutrition. 2006; 83: 124-131.
- Panlasigui LN, Thompson LU. Blood glucose lowering effects of brown rice in normal and diabetic subjects. International journal of food sciences and nutrition. 2006; 57: 151-158.
- Richardson DP. Wholegrain health claims in Europe. The Proceedings of the Nutrition Society. 2003; 62: 161-169.
- Jensen MK, Koh-Banerjee P, Hu FB, Franz M, Sampson L, Gronbaek M, et al. Intakes of whole grains, bran, and germ and the risk of coronary heart disease in men. The American journal of clinical nutrition. 2004; 80: 1492-1499.
- Liu S, Stampfer MJ, Hu FB, Giovannucci E, Rimm E, Manson JE, et al. Whole-grain consumption and risk of coronary heart disease: results from the Nurses' Health Study. The American journal of clinical nutrition. 1999; 70: 412-419.
- Montonen J, Knekt P, Jarvinen R, Aromaa A, Reunanen A. Whole-grain and fiber intake and the incidence of type 2 diabetes. The American journal of clinical nutrition. 2003; 77: 622-629.
- Esmaillzadeh A, Mirmiran P, Azizi F. Whole-grain consumption and the metabolic syndrome: a favorable association in Tehranian adults. European journal of clinical nutrition. 2005; 59: 353-362.