
Special Article - Oral Squamous Cell Carcinoma
J Dent & Oral Disord. 2016; 2(5): 1025.
An Aggressive Anterior Mandibular Adenomatoid Odontogenic Tumor: A Case Report
Gill DG¹* and Schlieve TS²
¹Department of Oral and Maxillofacial Surgery, Meharry Medical College, USA
²Department of Oral and Maxillofacial Surgery, University of Texas Southwestern Medical Center, USA
*Corresponding author: Danielle G Gill, Department of Oral and Maxillofacial Surgery, Meharry Medical College, USA
Received: June 13, 2016; Accepted: July 07, 2016; Published: July 08, 2016
Abstract
The Adenomatoid Odontogenic Tumor (AOT) is a rare benign complex odontogenic tumor, representing about 3% of all odontogenic tumors. The literature describes the AOT as both a true benign, non-aggressive, non-invasive neoplasm versus a developmental hamartomatous odontogenic growth. The tumor is characterized by its overwhelming female predilection, presence in the second decade of life, anterior position in the maxilla, association with impacted teeth, and appearance of scattered opacities within the tumor. Treatment is typically standard enucleation. Here, we report a case of extrafollicular-type adenomatoid odontogenic tumor in a 14-year-old African American female in the anterior mandible crossing the midline as well as the clinical, radiological, histological features of the tumor.
Keywords: Adenomatoid odontogenic tumor; Extrafollicular; Mandible; Perforation
Abbreviations
AOT: Adenomatoid Odontogenic Tumor; BMI: Body Mass Index; CBCT: Cone Beam Computed Tomography; WHO: World Health Organization
Introduction
The AOT is a benign complex tumor representing 3% [1,2] of odontogenic tumors, and ranked fourth most diagnosed [2-4]. The reported frequency of the AOT differs amongst various publications with most reporting 2.2-7.1% [1,2], but others have documented ranges from 0.6-38.5% [2,5]. The AOT was first documented in 1903 as a cystis chenepithelialen Geschwülste by Dr. Nakayama, a Japanese general surgeon [6]. Since then, authors all over the globe have described characteristics of what is now called the AOT, which was officially adopted in 1971 by the World Health Organization.
Descriptions of what they the actual AOT entity may be are controversial, with some defining the lesion as a true benign, nonaggressive, non-invasive neoplasm and others adapting the lesion as a developmental hamartomatous odontogenic growth [2,4,7]. Marx and Stern argue that the AOT is a true cyst, a not a neoplasm at all, but a true cyst with a lumen, lining and connective tissue [8,9].
Due to its classic and consistent clinical and radiographic characteristics, the AOT has earned the nickname “tumor of twothirds” [8-10]. Two-thirds of diagnosed AOTs occur in females of the second decade in the anterior maxilla. Two-thirds will also be superimposed on dentigerous cysts, associated with an impacted tooth, usually a canine and have scattered opacities throughout the lesion [10].
Age of diagnosis varies from three to 82 years, but most occur in the second decade of life [1]. The AOT has a female predilection, with a ratio of 2:1 compared to males, [1,10] is more likely to present on the left side of jaws, 50.7% versus 38.8% on the right, [11] and has a maxilla-to-mandible ratio of 1.7:1 [7]. Average reported size of the AOT is 1-3 cm, [12] ranging from 0.4-12 cm [7].
There are three known subtypes of the AOT, the follicular, extrafollicular, and peripheral types [3]. The follicular or pericoronal type makes up the majority of AOTs at 70.8%. The extrafollicular or extracoronal type and the rare peripheral or extraosseous/ gingival type make up 26.9% and 2.3%, respectively.
There are numerous radiological features that are associated with the AOT. It is mostly described as a unilocular radiolucency with welldefined borders [5,7]. They are usually associated with an impacted teeth, most often a permanent canine [7,11,13]. The presence of calcifications is a common finding appearing in 78% of AOTs [13]. Periapical radiographs are the gold standard when confirming appearance of calcified deposits [7,14]. These calcifications can present as a single radiopacityor numerous radiopaque foci [12].
Surgical management of the AOT is most often enucleation and curettage with predictable results [15]. The associated impacted tooth may or may not be extracted during enucleation. Marsupialization or decompressions are also treatment options with favorable outcomes [6]. Partial resection has been indicated with larger tumors or patients who are at risk for pathologic fracture [10]. The AOT has a recurrence rate of 0.2% and is not locally invasive [10].
In this case report, we present an extrafollicular type AOT of substantial size causing discernable facial asymmetry in the lower facial one-third. Uncommon clinical, radiographic, and histologic features are discussed and compared to the classic presentation of the AOT.
Case Report
A 14-year-old African-American female presented to the University of Illinois at Chicago Oral and Maxillofacial Surgery Clinic with swelling of the anterior mandible. The swelling started two years prior and had been slowly increasing in size, with intermittent of sharp pain. No history of trauma was reported. She was a febrile and denied paresthesia, dysguesia, dysphagia, dyspnea, chills, nausea and vomiting. Medical history included asthma, seasonal allergies, and morbid obesity (BMI34). The patient was prescribed a decongestant for seasonal allergies, albuterol as needed for asthma, and over the counter pain relievers for pain relief. Previous surgical history, social history or family history was non-contributory.
Head and neck examination revealed moderate to severe asymmetry of the lower one-third of the face. The area was firm and tender when manipulated. There was no appreciable erythema or purulence, nor was there any associated lymphadenopathy, trismus, nasal deviations, or temporomandibular joint pathology. Two-point discrepancy and brush stroke testing were normal on the tongue, lower lip and chin indicating no neurological deficits. Intraorally, anterior mandibular labial and lingual expansion was appreciated, extending from teeth #22-27. There was visible splaying of mandibular anterior teeth as well as mobility of teeth #23-27. Vitality results of teeth #22- 27 were normal during examination. Maximum incisor opening was over forty millimeters. Oropharynx was clear. Uvula was midline, and no palatal draping was noted. Floor of the mouth was soft, nontender and non-extended.
CBCT revealed a 3.3 x 2.7 cm diameter, well-defined, unilocular, radiolucency mesial to tooth #22 crossing the midline to the mesial of tooth #28 (Figure 1). Resorption of the root apices on teeth #24- 27 (Figure 1) was displayed. Centered within the radiolucency was a single 2x2 mm radiopacity (Figures 2 & 3) and both labial and lingual cortices of the anterior mandible were perforated (Figure 4).
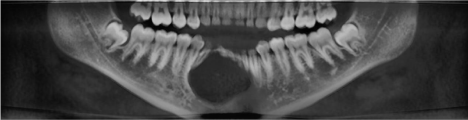
Figure 1: Panoramic View: Mixed radiolucent and radiopaque lesion of the
anterior mandible.
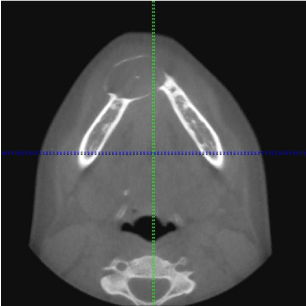
Figure 2: Axial View: Mixed radiolucent and radiopaque lesion of the anterior
mandible.
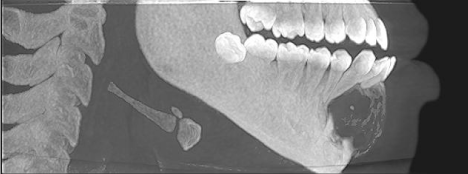
Figure 3: Sagittal View with soft tissue.
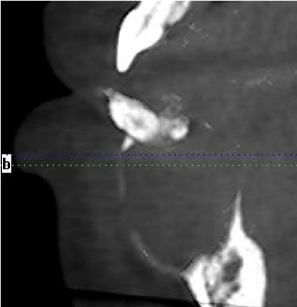
Figure 4: Sagittal view with soft tissue (40x).
The patient was consented, a timeout was performed, and an excisional biopsy was performed under local anesthesia, after a negative aspirate. The lesion was accessed via a sulcular incision, followed by a full thickness mucoperiosteal flap and completely enucleated from the bony cavity as an intact specimen. Gross examination of lesion revealed a 2.8 x 2.6 x 1.4 cm gelatinous solid-like lesion with a thick cystic lining. No teeth were extracted during the procedure. The lesion was placed in formalin and sent to pathology for histological assessment.
Histologic evaluation revealed a benign epithelial odontogenic neoplasm with dense fibrous connective tissue covered by lining epithelium (Figure 5). The lining epithelium consisted of interconnecting ribbons of cuboidal eosinophilic cells (Figure 6). Intraluminal proliferation of the epithelium occurred allowing for solid sheets and nests of polyhedral to spindle-shaped cells to form. Within the sheets of cells were somewhat circular well-defined areas that exhibited hyalinized and mineralized areas. An occasional ductlike structure was formed within the defined spheres (Figure 7).
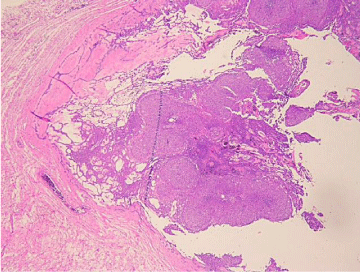
Figure 5: Low Power: Thick outer fibrous wall with fishnet lattice work of
cellular strands and cellular balls trapped within the lattice work.
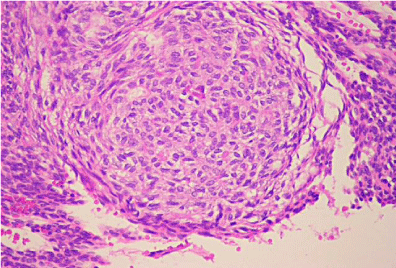
Figure 6: High Power: Cellular nests with eosinophilic droplets.
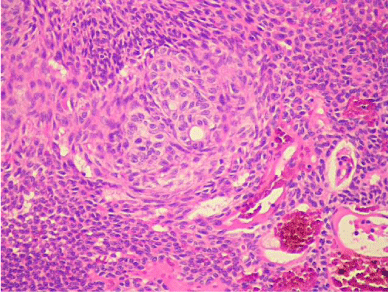
Figure 7: High Power: Cellular ball with a tiny duct-like structure. Beginning of
the ameloblasts starting to organize around the central zone of the nest. The
tied sausage will appear soon after.
A working differential diagnosis was developed based on the patient’s symptomatology, clinical and radiographic findings. The calcifying odontogenic cyst, ameloblastic fibro-odontoma, adenomatoid odontogenic tumor, unicysticameloblastoma, and keratocystic odontogenic tumor were included amongst possible diagnoses. The final diagnosis was confirmed as an extrafollicular type adenomatoid odontogenic tumor. Healing was uneventful at two-week follow-up appointment. Patient was eventually lost to follow-up.
Discussion
The tumor described was present in a female of the second decade, with radiographic presentation of calcifications without the other classic descriptive findings of the AOT. Uncommon and aggressive features will be described below.
Often labeled as a painless, slow-growing lesion, [1,4,13] pain has been described upon presentation as was the case with this lesion. Swasdison et al. showed that at least 10% of their AOT population and Becker et al. showed at least 13% of their AOT populations experienced pain upon diagnosis. This may be due to pressure from tooth movement due to increased size of the lesion, mobility of the teeth causing pain under function, or a combination.
There are three known types of the AOT. The extrafollicular variant has a mandibular predilection [7] and is most likely to cause displacement of teeth, both of which are characteristics present in reported tumor [12]. The extrafollicular in our reported tumor type is further separated into four additional subtypes based on radiographic appearance [3]. The E1 subtype has no relation to tooth structures, E2 subtype presents interradicularly, E3 subtype is located in the periapical region, and the E4 subtype is usually superimposed on other anatomical structures [3]. There have been rare reports of a multivariate subtype that has been discussed in the literature [2,4,16]. The classification of the reported tumor is an E2 type due to its position between the teeth unlike its usual association with impacted teeth.
The radiographic presentation of this type of lesion is typical of a benign odontogenic tumor. Specific to the AOT, there is a direct correlation between the presence of calcifications and the expansion of bony cortices [7]. Expansion of the bony cortices with or without perforation is rare but has been reported in the literature [7,13] and is more likely to be present in patients over the age of 16 [7]. The size of the lesion correlates directly with the expansion and perforation of the cortices, root resorption, and ill-defined borders [7]. Root resorption is more likely to be associated with patients over the age of 30 [7]. The lesion described presented with a single radiopaque focus which correlates with the presence of expansion of the bony cortices, a characteristic which this lesion possesses. The size of the lesion would also correlate with other characteristics such as perforation of the cortex as well as splayed teeth, and root resorption. The aggressive nature of this particular lesion is evident due to the age of the patient at diagnosis and the characteristics observed during evaluation.
In 2005, the histologic typing of WHO defined the AOT as a tumor composed of odontogenic epithelium, presenting a variety of histoarchitectural patterns, embedded in mature connective tissue stroma, and characterized by slow and progressive growth [15]. The tumor is usually composed of spindle-shaped or polygonal cells that form sheets and whorled masses within a connective tissue stroma [4]. There is amorphous eosinophilic material seen between the epithelial cells and in the center of the rosette-like structure [1,12]. The characterizing duct-like structures are lined by a row of columnar epithelial cells [1,12]. Although the duct-like structures are a distinguishing characteristic, diagnosis of the AOT can be made without them. Various amounts and different shapes and sizes of dystrophic calcifications can be encountered within the lumina of the duct-like structures [12]. Osteodentin and perivascular hyalinization are sometimes appreciated [5,14]. In rare instances, foci of epithelial mitotic activity have been reported. Neither epithelial nor stromal cytologicatypia have ever been reported [1,4] and there is no known malignant counterpart for the AOT [2,6].
Conclusion
Over the last forty years, the literature has provided detailed information on the description of the AOT as well as treatment options. The lesion is usually predictable in terms of its position within the jaw as well as clinical findings. However, abnormal location and a lack of association with impacted teeth, in addition to pain can make the AOT a more difficult choice when forming a differential diagnosis. It is important that the clinician be well versed in the various clinical findings of the AOT was well as its subtypes.
This article is an unusual representation of a classic lesion, both from its aggressive clinical presentation and the rarity of the subtype. It is important to continue to document atypical characteristics along with rare subtypes.
References
- Philipsen HP, Reichart PA. Adenomatoid odontogenic tumour: facts and figures. Oral Oncol. 1999; 35: 125-131.
- Philipsen HP, Reichart PA, Siar CH, Ng KH, Lau SH, Zhang X, et al. An updated clinical and epidemiological profile of the adenomatoid odontogenic tumour: a collaborative retrospective study. J Oral Pathol Med. 2007; 36: 383-393.
- Ide F, Mishima K, Kikuchi K, Horie N, Yamachika S, Ssatomura K, et al. Development and growth of adenomatoid odontogenic tumor related to formation and eruption of teeth. Head Neck Pathol. 2011; 5: 123-132.
- Rick G. Adenomatoid odontogenic tumor. Oral Maxillofac Surg Clin North Am. 2004; 16: 333-354.
- de Matos FR, Nonaka CFW, Pinto LP, Souza LB, de Almeida Freitas R. Adenomatoid odontogenic tumor: retrospective study of 15 cases with emphasis on histopathologic features. Head Neck Pathol. 2012; 6: 430-437.
- Ide F, Muramatsu T, Ito Y, Kikuchi K, Miyazaki Y, Saito I, et al. An expanded and revised early history of the adenomatoid odontogenic tumor. Oral Surg Oral Med Oral Pathol Oral Radiol. 2013; 115: 646-651.
- Becker T, Buchner A, Kaffe I. Critical evaluation of the radiological and clinical features of adenomatoid odontogenic tumour. Dentomaxillofac Radiol. 2012; 41: 533-540.
- Kurra S, Gunupati S, Prasad PR, Y SR, Reddy BVR. An Adenomatoid Odontogenic Cyst (AOC) with an Assorted Histoarchitecture: A Unique Entity. J Clin Diagn Res. 2013; 7: 1232-1235.
- Marx R, Stern D. Oral and Maxillofacial Pathology: A Rationale for Diagnosis and Treatment. 2 edn. Chicago: Quintessence. 2012.
- More C, Das S, Gupta S, Bhavsar K. Mandibular adenomatoid odontogenic tumor: Radiographic and pathologic correlation. J Nat Sci Biol Med. 2013; 4: 457-462.
- Swasdison S, Dhanuthai K, Jainkittivong A, Philipsen HP. Adenomatoid odontogenic tumors: an analysis of 67 cases in a Thai population. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008; 105: 210-205.
- Philipsen HP, Reichart PA, Zhang KH, Nikai H, Yu QX. Adenomatoid odontogenic tumor: biologic profile based on 499 cases. J Oral Pathol Med. 1991; 20: 149-158.
- Handschel JGK, Depprich, RA, Zimmermann AC, Braunstein S, Kubler N. Adenomatoid odontogenic tumor of the mandible: review of the literature and report of a rare case. Head Face Med. 2005; 1: 3.
- Philipsen HP, Reichart PA, Nikai H. The adenomatoid odontogenic tumour (AOT): an update. Oral Med Pathol. 1998; 2: 55-60.
- Baskaran P, Misra S, Kumar MS, Mithra R. Adenomatoid odontogenic tumor: A report of two cases with histopathology correlation. J Clin Imaging Sci. 2011; 1: 64.
- Larsson A, Swartz K, Heikinheimo K. A case of multiple AOT-like jawbone lesions in a young patient- a new odontogenic entity? J Oral Pathol Med. 2003; 32: 55-62.