
Review Article
J Dis Markers. 2015;2(1): 1017.
The CirculatingVE-catherin as Predictor of Poor Outcomes in Patients with Ischemic Chronic Heart Failure
Alexander E Berezin1*, Alexander A Kremzer2, Yulia V Martovitskaya3, Tatayna A Samura2 and Tatyana A Berezina4
1State Medical University, Internal Medicine Department, Zaporozhye, Ukraine
2State Medical University, Clinical Pharmacology Department, Zaporozhye, Ukraine
3State Medical University, Pathology Department, Zaporozhye, Ukraine
4Private center “VITA CENTER”, Zaporozhye, Ukraine
*Corresponding author: Alexander E Berezin, Internal Medicine Department, State Medical University, Ukraine
Received: December 27, 2014; Accepted: January 27, 2015; Published: January 29, 2015
Abstract
Aim: To evaluate the predictive value of circulating VE-catherin for cumulative cardiovascular outcomes in patients with chronic heart failure (CHF).
Methods: A total of 388 patients with CHF were enrolled in the study on discharge from the hospital. Observation period was up to 3 years. Blood samples for biomarkers measurements were collected. ELISA method for measurements of circulating level of CD144 (VE-catherin) was used. Concentrations of VEcatherin for cumulative cardiovascular outcomes were tested.
Results: Median follow-up was of 2.32 years (IQR=1.8-3.0). During followup, 110 cardiovascular events (including 43 fatal cases) were determined. Thirty five patients were died due to advance of CHF, and eight cases of death were related with suddenly death, fatal myocardial infarction, and systemic thromboembolism. Therefore, 74 subjects were readmitted in the hospital due to worsening CHF and 16 subjects were readmitted due to other cardiovascular reasons. Medians of circulating levels of VE-catherin in both patient cohorts (without and with experienced clinical outcomes) were 0.63 ng/mL (iterquartile range [IQR] = 0.55-0.64 ng/mL) and 1.03 ng/ml (IQR = 0.97-1.07 ng/mL) (P<0.001). Receive Operation Curve analysis has shown that the best balanced the cut-off of VE-catherin concentration for combined cardiovascular events was 0.755ng/ml. A significantly divergence of Kaplan-Meier curves in high VE-catherin patients (>0.755ng/ml) and low VE-catherin (<0.755 ng/ml) was reported. Multivariate age-adjusted Cox regression shown that NYHA class, VEcatherin, NT-pro-BNP and LVEF remained independent predictors for combined cardiovascular events, whereasT2DM and hs-CRP did not.
Conclusion: We found that increased circulating VE-catherin associated with increased three-year combined cardiovascular events among patients with CHF. Adding VE-catherin level to NT-pro BNP and NYHA class sufficient improve the prediction of standard model of CHF.
Keywords: VE-catherin; Chronic heart failure; Cardiovascular outcomes; Prognosis
Introduction
Chronic heart failure (CHF) is considered a leading cause of morbidity and mortality worldwide [1]. Nature evolution of CHF associates with endothelial dysfunction that is result in shear stress disorders on endothelium. However, several factors, such as angiotensin-aldosterone system activation, oxidative stress, inflammatory response, exaggerated extracellular matrix remodelling in vasculature, degradation of vasodilators, etc. are referred as causality modulator of endothelial dysfunction [2]. There are evidences regarding pivotal role of endothelial dysfunction in cardiovascular mortality in patients with CHF especially ischemic origin [3]. Therefore endothelial dysfunction predicts cardiovascular outcomes independent of conventional cardiovascular risk factors [3]. Mechanical interaction of endothelial cells (ECs) mediated by blood flow may lead to junction of vascular remodelling [4]. Particularly this phenomenon is realized by involvement of junctional protein vascular endothelial-cadherin (VE-cadherin) that is essential for supporting endothelial function [5]. Currently VE-cadherin is considered a component of endothelial cell-to-cell adherence junctions and it has a key role in the maintenance of vascular integrity [6]. The mechanisms of action of VE-cadherin are complex and include reshaping and organization of the ECs cytoskeleton and modulation of gene transcription [6]. Therefore, VE-cadherin mediates cell adhesion and monolayer integrity, regulates angiogenesis and actin-driven to be support a mechanical capacity of endothelium after activation by soluble vascular cell adhesion molecules [7]. There is currently evidence affected VE-cadherin as a marker of endothelial dysfunction in several settings including CHF [8]. Therefore, several factors may modulate CHF evolution, such as ischemia. We tested hypothesis that VE-cadherin added to traditional biomarkers may improve a risk stratification of the patients with ischemic-induced CHF, although VE-cadherin alone probably has not a predictive value. However, the prognostic value of circulating VE-catherin in CHF has not defined. The objective of this study was to evaluate the prognostic value of circulating VE-catherin for combined cardiovascular events in patients with chronic heart failure.
Methods
The study population consisted of 388 consecutive patients with CHF who were reposted myocardial infarction or underwent quantitative coronary artery angiography or PCI between April 2010 to June 2014 (Figure 1). All these patients were selected after reviewing 1427 discharge reports obtained from persons who were treated in Zaporozhye Regional Hospital, City Hospital #6, City Hospital #10, Zaporozhye Regional Center of Cardiovascular Diseases and Private center “VITA CENTER” with primary diagnosis ischemic heart disease. One hundred fifthly five subjects were excluded due incompliance of the study protocol because of no documented ischemic heart disease was presented, which was determined when exiting myocardial infarction and/ or stenosis of coronary arteries were found. Among 1272 discharge reports were enrolled data regarding 388 patients with ischemic-induced CHF diagnosed according to current clinical guidelines [8]. Patients with severe kidney and liver diseases; malignancy; creatinine plasma level above 440μmol/L; estimated GFR index< 35 ml/min/м2; brain injury within 3 months before the enrollment; pulmonary edema; tachyarrhythmia; valvular heart disease; thyrotoxicosis; ischemic stroke; intracranial hemorrhage; acute infections; surgery; trauma; all the ischemic events within 3 previous months; inflammations within a previous month; pregnancy; implanted pacemaker, any disorder that may discontinue patient’s participation in the study according to investigators were excluded from the study.
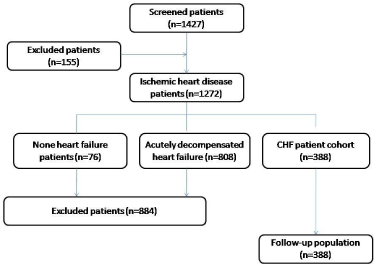
Figure 1: Figure shows flow chart for procedures of subject recruitment.
The study protocol was approved by the Zaporozhye State medical University Ethnics committee review board. The study complied with the Declaration of Helsinki and voluntary informed written consent was obtained from all patients included in this study.
Prognosis was assessed by the composite endpoint related to combined all-cause death, CHF-related death or CHF hospitalization, censored at 3 years. We used contact by phone with patients, their relatives, review of medical records and local hospital registers, analysis of electrocardiogram, ultrasound echocardiogram and cardiac enzyme data for identification of end-points.
Methods for visualization of coronary arteries
Multispiral computed tomography angiography was performed for patients with high probability of ischemic heart disease prior to their inclusion in the study as screening procedure on SomatomVolum Zoom scanner (Siemens, Erlangen, Germany) and Optima CT660 (GE Healthcare, USA) [10]. Coronary angiography was performed for patients with exiting ischemic heart disease per conventional protocol prior to study entry. Stenosis > 50% of one and more coronary arteries was defined as criteria of clinically significant ischemic heart disease.
Echocardiography
Transthoracic B-mode echocardiography and Tissue Doppler Imaging were performed according to a conventional procedure on ACUSON scanner (SIEMENS, Germany) using phased probe of 2.5- 5 МHz. Left ventricular ejection fraction (LVEF) were measured by modified Simpson’s method [11]. Inter- and intra-observer variability coefficients for LVEF were 3.2% and 1.1% respectively. Tissue Doppler Echocardiography was performed in 4-, 3- and 2-chamber views in each of 16 segments of the left ventricle and in 4 spots of the mitral annulus: at the base of posterior septal, lateral, inferior, and anterior left ventricular walls [12].
Calculation of glomerular filtration rate
Glomerular filtration rate (GFR) was calculated by CKD-EPI formula [13].
Measurement of VE-catherin, NT-pro-BNP, total cholesterol and its fractions
To determine VE-catherin, N-terminal pro-brain natriuretic peptide (NT-pro-BNP), total cholesterol and cholesterol fractions, blood samples were drawn in the morning (at 7-8a.m.) into cooled silicone test tubes. Samples wereprocessed according to the recommendations ofthe manufacturer of the analytical technique used. They were centrifuged upon permanent cooling at 6,000 rpm for 3 minutes. Then, plasma was refrigerated immediately to be stored at a temperature not higher than -35°С.
Circulating CD144 (VE-catherin) level was determined by ELISA method (Bender Med Systems GmbH, Vienna, Austria). NT-pro- BNP content was measured by immune electro chemo luminescent assay using sets by R&D Systems (USA). Concentrations of total cholesterol (TC) and cholesterol of high-density lipoproteins (HDLP) were measured by fermentation method. Concentration of cholesterol of low-density lipoproteins (LDL-C) was calculated according to the Friedewald formula (1972).
Statistical analysis
Statistical analysis of the results obtained was performed in SPSS system for Windows, Version 22 (SPSS Inc, Chicago, IL, USA). The data were presented as mean (М) and error of mean (±EM) or 95% confidence interval (CI); median (Ме) and interquartile range (IQR). The hypothesis of normal distribution of the parameters analyzed was checked by means of Shapiro–Wilk test and Kolmogorov-Smirnov test. To compare the main parameters of patients’ groups (subject to the type of distribution of the parameters analyzed), one-tailed Student t-test or Shapiro–Wilk U-test were used. To compare categorical variables between groups, Chi2 test (χ2) and Fisher F exact test were used. The circulating CD144 (VE-catherin) and NT-pro-BNP level in the blood failed to have a normal distribution, while distribution of the total cholesterol and cholesterol fractions had a normal character (estimated by means of Kolmogorov-Smirnovtest) and was not subjected to any mathematical transformation. The factors, which could be associated potentially with circulating VE-catherin, were determined by logistic regression analysis. Receive Operation Characteristic (ROC) Curve analysis was performed to identify the well-balanced cutoff points regarding VE-catherin concentration with optimal predicted value. Kaplan-Meier curves were constructed for patients depended on cut-off point of VE-catherin level. Cox regression was used for calculation of Hazard Ratio (OR) and 95% CI for all predictors of combined clinical outcomes. A calculated difference of P<0.05 was considered significant.
Results
General characteristics of study patient population
At baseline mean age of patients in entire cohort was 58.34 years (Table 1). The prevalence of II (37.9%) and III (21.4%) NYHA class was determined. At least 55.5% of CHF subjects enrolled in the study were hypertensive. Other cardiovascular risk factors, such as dyslipidemia, type two diabetes mellitus and obesity, were reported 66.0%; 37.6%; and 44.3% respectively. Mean value of left ventricular ejection fraction was decreased slightly.
Entire patient cohort (n=388)
Subjects who did not experienced the composite endpoint (n=278)
P value
Age, years
58.34±9.60
57.32±6.15
58.73±7.22
0.86
Male, n (%)
207 (53.3%)
64 (58.2%)
143 (51.4%)
0.88
I NYHA class, n (%)
77 (19.8%)
-
77 (27.7%)
0.001
II NYHA class, n (%)
147 (37.9%)
26 (23.6%)
121 (43.5%)
0.001
III NYHA class, n (%)
83 (21.4%)
52 (47.3%)
31 (11.2%)
0.001
IV NYHA class, n (%)
81 (20.9%)
32 (29.1%)
49 (17.6%)
0.001
Hypertension, n (%)
214 (55.5%)
62 (56.4%)
152 (54.7%)
0.96
Dyslipidemia, n (%)
256 (66.0%)
48 (43.6%)
208 (74.8%)
0.024
Type two diabetes mellitus, n (%)
146 (37.6%)
42 (38.2%)
104 (37.4%)
0.94
Obesity, n (%)
172 (44.3%)
54 (49.1%)
118 (42.4%)
0.82
Adherence to smoke, n (%)
76 (19.6%)
25 (22.7%)
51 (18.3%)
0.77
BMI, kg/m2
24.1 (95% CI = 21.6 – 28.7)
23.9 (95% CI=20.7–25.9)
23.3 (95% CI=21.5–24.8)
0.68
Systolic BP, mm Hg
131±8
130±5
133±5
0.84
Diastolic BP, mm Hg
78±5
77±4
78±4
0.92
Heart rate, beat per min.
70.52±3.34
74.60±4.6
69.10±6.2
0.48
LVEF, %
42.80±5.76
42.20±3.11
43.20±6.18
0.76
GFR, 1,73 ml/ min/ m2
82.3 (95% CI = 68.7 – 102.6)
81.5 (95% CI=71.3–94.7)
83.9 (95% CI=77.1–102.6)
0.055
Creatinine, µmol/L
72.3 (95% CI = 58.7 – 92.6)
73.1 (95% CI=60.9–80.5)
70.7 (95% CI = 59.1 – 88.1)
0.048
Fasting glucose, mmol/L
5.20 (95% CI =3.3-9.7)
5.27(95% CI=3.5-9.4)
4.98 (95% CI=3.8-8.1)
0.28
HbA1c, %
6.8 (95% CI =4.1-9.5)
6.9 (95% CI=4.3-9.2)
6.6 (95% CI=4.6-8.3)
0.36
Hemoglobin, g/L
135.4 (95% CI = 128.5 – 140.1)
134.1 (95% CI = 126.2 – 136.4)
136.1 (95% CI = 125.1 – 144.8)
0.06
Total cholesterol, mmol/L
5.1 (95% CI = 3.9 – 6.1)
5.3 (95% CI=4.6-6.0)
5.0 (95% CI = 3.5 – 5.9)
0.047
Cholesterol HDL, mmol/L
0.91 (95% CI = 0.89 – 1.12)
0.96 (95% CI = 0.93–1.05)
0.88 (95% CI = 0.84 – 1.01)
0.044
Cholesterol LDL, mmol/L
3.23 (95% CI = 3.11 – 4.40)
3.71 (95% CI =3.50–4.20)
3.53 (95% CI =3.11–3.97)
0.06
NT-pro-BNP, pg/mL
1977.2 (95% CI 984.7 – 2993.2)
2616.5 (95% CI 1085.3 – 3683.5)
1530.6 (95% CI = 644.5 – 2560.6)
0.042
hs-CRP, mg/L
7.34 (95% CI =6.77-7.95)
8.04 (95% CI =6.81-9.52)
6.96 (95% CI =5.03-8.13)
0.036
Notes: P value was calculated between variables for subjects who experienced the composite endpoint and did not; data were presented as median and 95 confidence interval (CI); NYHA – New York Heart Association; GFR – glomerular filtration rate; BMP – brain natriuretic peptide; BP – blood pressure; LVEF – left ventricular ejection fraction; BMI – body mass index, HbA1c – glycated hemoglobin, HDL - high-density lipoprotein; LDL - Low-density lipoprotein.
Table 1: General characteristic of patients enrolled in the study.
Subjects who experienced the composite endpoint presented frequently III-IV NYHA classes, lower glomerular filtration ratio (GFR), as well as creatinine level, NT-pro-BNP, hs-CRP, and lipid abnormalities. Other parameters were similar for both patient cohorts.
CHF patients were treated with ACE inhibitors or ARBs, betaadrenoblockers, I/f blocker ivabradine, mineralocorticoid receptor antagonists, and antiplatelets (Table 2). Loop diuretics were added when fluid retention was determined. Dihydropyridine calcium channel blockers were added when elevated was uncontrolled by previous treatment scheme. Metformin and / or sitagliptin were used in type two diabetes patients as a component of contemporary treatment of CHF. Loop diuretics and aspirin were prescribed frequently in subjects who experienced the composite endpoint. In opposite, beta-adrenoblockers, statins and sitagliptin were given frequently among patients who did not.
Entire patient cohort (n=388)
Subjects who experienced the composite endpoint (n=110)
Subjects who did not experienced the composite endpoint (n=278)
P value
ACE inhibitors or ARBs, n (%)
388 (100%)
110 (100%)
278 (100%)
1.0
Aspirin, n (%)
305 (78.6%)
96 (87.3%)
209 (75.2%)
0.022
Other antiplatelet drugs, n (%)
83 (21.4%)
14 (12.7%)
69 (24.8%)
0.026
Beta-adrenoblockers, n (%)
324 (83.5%)
73 (66.4%)
251 (90.3%)
0.001
Dihydropyridine calcium channel blockers, n (%)
63 (16.2%)
17 (15.5%)
46 (16.5%)
0.88
Ivabradine, n (%)
137 (35.3%)
43 (39.0%)
94 (33.8%)
0.78
Mineralocorticoid receptor antagonists, n (%)
152 (39.2%)
45 (40.9%)
107 (38.5%)
0.66
Loop diuretics, n (%)
311 (80.1%)
110 (100%)
201 (72.3%)
0.043
Statins, n (%)
294 (75.7%)
48 (43.6%)
246 (88.5%)
0.012
Metformin, n (%)
146 (37.6%)
42 (38.2%)
104 (37.4%)
0.86
Sitagliptin, n (%)
48 (12.4%)
9 (8.2%)
40 (14.4%)
0.001
Notes: P value was calculated between variables for subjects who experienced the composite endpoint and did not; data were presented as numerous and frequency; ACE – angiotensin-converting enzyme; ARBs – angiotensin-2 receptor blockers.
Table 2: Treatment strategy in CHF patients enrolled in the study.
Clinical event determination
Median follow-up was of 2.32 years (IQR=1.8-3.0). During follow-up, 110 cardiovascular events (including 43 fatal cases) were determined. Thirty five patients were died due to advance of CHF and eight cases of death were related with suddenly death, fatal myocardial infarction and systemic thromboembolism. No other causes of death were defined. Therefore, 74 subjects were readmitted in the hospital due to worsening CHF and 16 subjects were readmitted due to other cardiovascular reasons.
Circulating VE-catherin level in both patient cohorts
Median values of circulating VE-catherin levels in both patient cohorts (without and with experienced clinical outcomes) were 0.63 ng/mL (IQR = 0.55-0.64 ng/mL) and 1.03 ng/mL (IQR = 0.97-1.07 ng/mL) (P<0.001). The data suggested that VE-catherin plasma levels were directly related to NYHA functional class of CHF (r = 0.474, P = 0.006), NT-pro-BNP (r = 0.344, P = 0.001), T2DM (r = 0.42, P = 0.006), gender (r = 0.366, P < 0.001 for male), TC (r = 0.32, P = 0.001), and inversely to LVEF (r = -0.426, P = 0.001) and GFR (r = -0.416, P = 0.002). No significant association between the levels of circulating VE-catherin with fasting plasma glucose, HbA1c, mean systolic and diastolic BP, and medications for both cohorts of the patients was found.
The predictive value of VE-catherin concentrations in study patient population
Receive Operation Curve (ROC) analysis has shown that the best balanced the cut-off of VE-catherin concentration for combined cardiovascular events was 0.755 ng/ml. Area under cure was 0.937 (Std. error = 0.20; 95% CI = 0.895-0.980), sensitivity and specificity were 96.0% and 70.6% respectively.
The model was robust and it has provided a significant results using well balanced cut-off point of VE-catherin level (over 0.755ng/ ml versus under 0.755ng/ml). Kaplan-Meier survival curves were constructed for CHF patients depended on VE-catherin level. A significantly divergence of Kaplan-Meier curves in high VE-catherin patients (>0.755ng/ml) and low VE-catherin (<0.755 ng/ml) was reported (Figure 2).
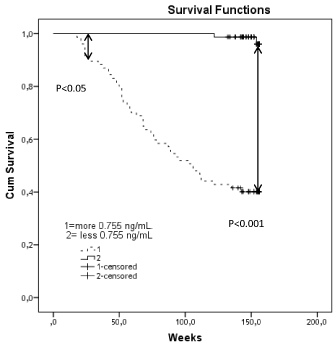
Figure 2: Results of Kaplan-Meier survival analysis: The cumulative cardiovascular outcomes in both groups of patients with low (<0.755ng/ml) and high (>0.755ng/ml) circulating VE-catherin.
In the univariate Cox regression age-adjusted analysis, NYHA class, VE-catherin, NT-pro-BNP, LVEF, and T2DM were found the main factors independently related to combined cardiovascular events. Multivariate age-adjusted Cox regression shown that NYHA class, VE-catherin, NT-pro-BNP and LVEF remained independent predictors for combined cardiovascular events, whereasT2DM and hs-CRP did not (Table 3).
Variables
Combined cardiovascular events
Univariate Cox regression
Multiivariate Cox regression
HR
95% CI
P value
HR
95% CI
P value
NYHA class (II-IV vs I)
1.18
1.12–1.23
0.001
1.10
1.03–1.16
0.003
VE-catherin (over 0.755 ng/ml vs under 0.755 ng/ml)
1.12
1.02–1.19
0.001
1.10
1.05–1.14
0.001
NT-pro-BNP ( >2560 pg/mL vs <2560 pg/mL)
1.15
1.11–1.21
0.002
1.09
1.02–1.16
0.002
LVEF, (< 45% vs >45%)
1.10
1.06–1.19
0.003
1.06
1.03–1.10
0.003
T2DM (present vs absent)
1.07
1.04–1.12
0.001
1.05
1.00–1.08
0.058
hs-CRP (>6.5 mg/L vs <6.5 mg/L)
1.05
1.01–1.09
0.002
1.02
1.00–1.04
0.062
Note: HR – hazard ratio, CI – confidence interval; LVEF – left ventricular ejection fraction; BNP – brain natriuretic peptide; T2DM – type two diabetes mellitus, CRP – C reactive protein.
Table 3: Predictors related to three-year combined cardiovascular events included all-cause mortality, CHF-related death, and CHF-related rehospitalisation. Results of Cox Regression age-adjusted Analysis.
Using C-statistics for Models with NYHA class, LVEF and circulating biomarkers (VE-catherin, NT-pro-BNP) as Continuous Variables we found that adding of combination of biomarkers (VEcatherin, NT-pro-BNP) to the based model (NYHA class) improved the relative IDI by 10.1% for combined cardiovascular events (Table 4).
Models
Dependent variable: combined cardiovascular events
AUC (95% CI)
ΔAUC
IDI (±SE)
Relative IDI (%)
Model 1 (based model: NYHA class)
0.626
-
-
-
Model 1 + LVEF
0.633
-
-
-
Model 1 + LVEF vs Model 1
-
0.055; P=0.42
0.02±0.007
2.1%
Model 1 (based model: NYHA class)
0.626
-
-
-
Model 1 + NT-proBNP
0.668
-
-
-
Model 1 + NT-proBNP vs Model 1
-
0.042; P=0.026
0.03±0.009
5.5%
Model 1 (based model: NYHA class)
0.626
-
-
-
Model 1 + NT-proBNP + LVEF
0.663
-
-
-
Model 1 + NT-proBNP + LVEF vs Model 1
-
0.037; P=0.045
0.03±0.011
5.7%
Model 1 (based model: NYHA class)
0.626
-
-
-
Model 1 + VE-catherin
0.665
-
-
-
Model 1 + VE-catherin vs Model 1
-
0.039; P=0.033
0.02±0.010
5.6%
Model 1 (based model: NYHA class)
0.626
-
-
-
Model 1 + VE-catherin + NT-proBNP
0.690
-
-
-
Model 1 + VE-catherin + NT-proBNP vs Model 1
-
0.064; P<0.001
0.03±0.015
10.1%
Model 1 (based model: NYHA class)
0.626
-
-
-
Model 1 + VE-catherin + NT-proBNP + LVEF
0.672
-
-
-
Model 1 + VE-catherin + NT-proBNP + LVEF vs Model 1
-
0.046; P<0.05
0.02±0.008
8.9%
Note: Relative IDI – calculated as the ratio of IDI over the discrimination slope of the model without NYHA class. Abbreviations: AUC – area under curve, SE – standard error, LVEF – left ventricular ejection fraction; BNP – brain natriuretic peptide, hs-CRP – high sensitive C-reactive protein.
Table 4: C-statistics for Models with NYHA class, LVEF and circulating biomarkers (VE-catherin, NT-pro-BNP) as Continuous Variables.
When we used other model constructed on entering variables IDI appears to be improved up to 5% for combined cardiovascular events (available for VE-catherin and NT-pro-BNP) (Table 5). Two biomarkers (VE-catherin and NT-pro-BNP) improve significantly predictive model based on NYHA class for combined cardiovascular events. In patient study population for category-free NRI, 5% of events (p=0.001) and 11% of non-events (p=0.001) were correctly reclassified by the addition of circulating inflammatory biomarkers (hs-CRP, OPG and adiponectin) to the base model (NYHA class) for combined cardiovascular events. Thus, we suggest that combination of VE-catherin and NT-pro-BNP remains statistically significant predictor for three-year combined cardiovascular events.
Model 2 vs Model 1
Categorical NRI
0.12 (95% CI=0.10-0.15)
Percentage of events correctly reclassified
5 (p=0.012)
Percentage of non-events correctly reclassified
8 (p=0.001)
Categorical free NRI
0.23 (95% CI=0.20-0.28)
Percentage of events correctly reclassified
5% (p=0.001)
Percentage of non-events correctly reclassified
11% (p=0.001)
Note: Model 1- NYHA class; Model 2 – NYHA class + VE-catherin and NT-pro-BNP
Abbreviations: NRI - net reclassification improvement.
Table 5: Prediction Performance Analyses for Models with NYHA class and circulating biomarkers (VE-catherin, NT-pro-BNP) as Continuous Variables for combined cardiovascular events.
Discussion
It has defined that beyond clinical guidance; echocardiography and natriuretic peptides have suboptimal test characteristics for population-wide CHF risk stratification [14]. While B-type natriuretic peptides re a strong prognostic indicator in CHF and possibly superior when compared with conventional prognostic biomarkers, the levels of both NT-pro-BNP and BNP must be interpreted according to the clinical picture [15]. In fact, the endothelium is considered an important targeting for traditional risk factors because the endothelial dysfunction remains to be independently associated with cardiovascular mortality and nature evolution of CHF. Thus, biomarkers of endothelial dysfunction and endothelial cell injury are determined as perspective tool for risk stratification in CHF patients. Looking for novel biomarkers are required to be improving reclassification of the risk in CHF over a clinical prediction model. We found that raised level of VE-catherin especially added to NTpro BNP associated with a greater risk of combined cardiac events in CHF patients. Because, VE-catherin plays a pivotal role in functional activity ECs, but did not effect on their proliferation and migration [5, 6], we suggested that VE-catherin might be considered a marker of endothelial dysfunction. Currently, lack of clinical investigations reflected a prognostic role of VE-catherin in CHF patients and predictive value for combination constructed two biomarkers (VEcatherin and NT-pro-BNP). Results of our study suggested that combination of VE-catherin with traditional prognostic biomarker NT-pro-BNP appears superior to standard model and probably has a complementary prognostic value in a patient with CHF. Hens, adding VE-catherin to NT-pro-BNP might be proposed as a suitable tool to improve medical decision making for an identification of CHF patients at high risk. It necessary notes that no any association circulating VE-catherin with demographics parameters, GFR, fasting glucose, lipids were found. Therefore, NYHA class and other cardiovascular risk factors, i.e. T2DM, produced a strong relation with VE-catherin level that might be reflected a severity of endothelial dysfunction as picture of CHF evolution. It is no clear whether elevated VE-catherin in CHF subjects is a result of ECs damage or opposite an endothelial dysfunction is exerted due to an impact of VE-catherin resulting in other reasons. Data regarding the causality role of VE-catherin in nature evolution of CHF are very limited and evidences affected of CD144 are presented for non-ischemic cardiac failure [16-18]. Our results showed that predictive value of model based on NT-pro BNP may be improved by adding of VE-catherin. Further research is necessary to understand the prognostic value of VE-catherin and to identify potential combinations of biological markers that determine the development and prognosis of ischemic CHF.
Conclusion
We found that increased circulating VE-catherin associated with increased three-year combined cardiovascular events among patients with ischemic CHF. Adding VE-catherin level to NT-pro BNP and NYHA class sufficient improve the prediction of standard model of CHF.
ethical Principles
The study was approved by the local ethics committee of State Medical University, Zaporozhye, Ukraine. The study was carried out in conformity with the Declaration of Helsinki.
Study Restrictions
This study has some restrictions. The authors believe that a greater cohort of patients with more incidences detected is desirable to improve the power of the study. The authors suppose that these restrictions might have no significant impact on the study data interpretation.
Acknowledgement
We thank all patients for their participation in the investigation, staff of the Regional Zaporozhye Hospital (Ukraine) and the doctors, nurses, and administrative staff in City hospital # 6 (Zaporozhye, Ukraine), general practices, and site-managed organizations that assisted with the study.
Authors’ Contributions
Alexander E Berezin initiated the hypothesis and designed the study protocol, contributed to analyze and interpret the data, and wrote the manuscript. Alexander A. Kremzer contributed to enroll the patients, collected and analyzed the data, checked clinical events, reviewed the source documents, and performed statistical analysis; Yulia V. Martovitskaya contributed circulating biomarker determination and interpreted the obtained results. Tatyana A. Samura preformed visualization procedures and analyzed the results of examinations. Tatyana A. Berezina contributed to enroll the patients in the study and collect the data.
References
- Roger VL. The heart failure epidemic. Int J Environ Res Public Health. 2010; 7: 1807-1830.
- Matsuzawa Y, Sugiyama S, Sumida H, Sugamura K, Nozaki T, Ohba K, et al. Peripheral endothelial function and cardiovascular events in high-risk patients. J Am Heart Assoc. 2013; 2: e000426.
- Veerasamy M, Bagnall A, Neely D, Allen J, Sinclair H, Kunadian V. Endothelial Dysfunction and Coronary Artery Disease: A State of the Art Review. Cardiol Rev. 2014; .
- Young JA, Ting KK, Li J, Moller T, Dunn L, Lu Y, et al. Regulation of vascular leak and recovery from ischemic injury by general and VE-cadherin-restricted miRNA antagonists of miR-27. Blood. 2013; 122: 2911-2919.
- Giannotta M, Trani M, Dejana E. VE-cadherin and endothelial adherens junctions: active guardians of vascular integrity. Dev Cell. 2013; 26: 441-454.
- Abu Taha A, Taha M, Seebach J, Schnittler HJ. ARP2/3-mediated junction-associated lamellipodia control VE-cadherin-based cell junction dynamics and maintain monolayer integrity. Mol Biol Cell. 2014; 25: 245-256.
- Ley K. Leukocytes talking to VE-cadherin. Blood. 2013; 122: 2300-2301.
- Suematsu M, Suzuki H, Delano FA, Schmid-Schönbein GW. The inflammatory aspect of the microcirculation in hypertension: oxidative stress, leukocytes/endothelial interaction, apoptosis. Microcirculation. 2002; 9: 259-276.
- McMurray JJV., Adamopoulos S, Anker SD. Auricchio A, Böhm M, Dickstein K. et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012. Eur Heart J. 2012; 33: 1787–1847.
- Bluemke DA, Achenbach S, Budoff M, Gerber TC, Gersh B, Hillis LD, et al. Noninvasive coronary artery imaging: magnetic resonance angiography and multidetector computed tomography angiography: a scientific statement from the American Heart Association Committee on Cardiovascular Imaging and Intervention of the Council on Cardiovascular Radiology and Intervention, and the Councils on Clinical Cardiology and Cardiovascular Disease in the Young. Circulation. 2008; 118: 586–606.
- Schiller NB, Shah PM, Crawford M, DeMaria A, Devereux R, Feigenbaum H, et al. Recommendations for quantitation of the left ventricle by two-dimensional echocardiography. American Society of Echocardiography Committee on Standards, Subcommittee on Quantitation of Two-Dimensional Echocardiograms. J Am Soc Echocardiogr. 1989; 2: 358-367.
- Pellerin D., Sharma R., Elliott P., Veyrat C. Tissue Doppler, strain, and strain rate echocardiography for the assessment of left and right systolic ventricular function. Heart. 2003; 89 (90003): iii9-17.
- Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009; 150: 604-612.
- Kalogeropoulos AP, Georgiopoulou VV, deFilippi CR, Gottdiener JS, Butler J; Cardiovascular Health Study. Echocardiography, natriuretic peptides, and risk for incident heart failure in older adults: the Cardiovascular Health Study. JACC Cardiovasc Imaging. 2012; 5(2): 131-140.
- Korenstein D, Wisnivesky JP, Wyer P, Adler R, Ponieman D, McGinn T. The utility of B-type natriuretic peptide in the diagnosis of heart failure in the emergency department: a systematic review. BMC Emerg Med. 2007; 7: 6.
- Kudo-Sakamoto Y, Akazawa H, Ito K, Takano J, Yano M, Yabumoto C, et al. Calpain-dependent cleavage of N-cadherin is involved in the progression of post-myocardial infarction remodeling. J Biol Chem. 2014; 289: 19408-19419.
- Vite A, Gandjbakhch E, Prost C, Fressart V, Fouret P, Neyroud N, et al. Desmosomal cadherins are decreased in explanted arrhythmogenic right ventricular dysplasia/cardiomyopathy patient hearts. PLoS One. 2013; 8(9): e75082.
- Glukhov AV, Fedorov VV, Kalish PW, Ravikumar VK, Lou Q, Janks D, et al. Conduction remodeling in human end-stage nonischemic left ventricular cardiomyopathy. Circulation. 2012; 125: 1835-1847.