
Research Article
J Dis Markers. 2015; 2(4): 1032.
An Association of Insulin Resistance with Numerous of Circulating Microparticles Originated from Endothelial Cells in Cardiac Failure Individuals without History of Diabetes Mellitus
Berezin A¹*, Kremzer A², Berezina T³ and Martovitskaya Y4
¹Internal Medicine Department, State Medical University, Zaporozhye, Ukraine
²Clinical Pharmacology Department, State Medical University, Zaporozhye, Ukraine
³Private Medical Center “Vita-Center”, Zaporozhye, Ukraine
4Clinical Laboratory “Dia-Service”, Immunology and Pathology Unit, Zaporozhye, Ukraine
*Corresponding author: Alexander Berezin, Internal Medicine Department, State Medical University, Ukraine
Received: October 19, 2015; Accepted: November 12, 2015; Published: November 16, 2015
Abstract
Background: The causality role of insulin resistance (IR) in chronic heart failure (CHF) subjects has not determined. The study was conducted to examine a relationship between IR and numerous of circulating endothelial cell-derived microparticles (EMPs) in patients with CHF
Methods: Three hundreds ischemic-induced CHF patients aged 48 to 62 years who underwent multispiral computed tomography angiography or coronary angiography was involved in the study. Ischemic-induced CHF has documented when atherosclerotic stenos is > 50% of at least one coronary artery was presented or previously defined myocardial infarction was reported. Biomarkers were measured at baseline of the study. Circulating EMPs were isolated from peripheral blood and measured using flow cytometry technique.
Results: These were not significant differences between patients with and without IR in EMPs labeled as CD144+/CD31+, CD144+/annexin V+, and CD62E+ microparticles. Higher concentrations of CD144+/CD31+/annexin V+ EMPs and CD31+/annexin V+ EMPs were found in IR subjects when compared with none IR patients. Multivariate linear regression analyses has shown HOMAIR (OR = 1.14, 95% CI=1.08-1.21, P = 0.001), NT-proBNP (OR = 1.07, 95% CI=1.04-1.10, P = 0.001), hs-CRP (OR = 1.04, 95% CI=1.02-1.07, P = 0.001), and NYHA class (OR = 1.03, 95% CI=1.01-1.05, P = 0.001) were predictors for increased CD31+/annexin V+ EMPs. Additionally, HOMA-IR (OR = 1.10, 95% CI=1.05-1.17, P = 0.001), NT-proBNP (OR = 1.08, 95% CI=1.04-1.12, P = 0.001), and NYHA class (OR = 1.05, 95% CI=1.02-1.09, P = 0.001) significantly predicted elevation of CD144+/CD31+/annexin V+ EMPs. C-statistics for Models with HOMA-IR, NYHA class, and CHF biomarkers (hs-CRP, NT-proBNP) as continuous variables reported that adding of combination of these biomarkers to the based model constructed with HOMA-IR did not improve the relative IDI for increased CD144+/CD31+/annexin V+ and CD31+/annexin V+ microparticles.
Conclusion: we found that IR was statistically significant predictor for increased apoptotic EMPs labelled as CD144+/CD31+/annexin V+ and CD31+/ annexin V+ microparticles in CHF patients without history of T2DM. We suggest that these findings might reflect an impaired phenotype of circulating EMPs in this patient population.
Keywords: Chronic heart failure; Insulin resistance; Endothelial cell-derived Microparticles; Immune phenotype
Abbreviations
ACEI: Angiotensin-Converting Enzyme Inhibitors; ARBs: Angiotensin Receptor Blockers; AUC: Area Under Curve; BMI: Body Mass Index; BNP: Brain Natriuretic Peptide; CHF: Chronic Heart Failure; CV: Cardiovascular; EMPs: Endothelial-Derived Microparticles; GFR: Glomerular Filtration Rate; hs-CRP: High Sensitive C-Reactive Protein; HbA1c: Glycated Hemoglobin; HDL-C: High-Density Lipoprotein Cholesterol; HFpEF: CHF with Precerved LVEF; HFrEF: CHF with Reduced LVEF; IR: Insulin Resistance; LDL-C: Low-Density Lipoprotein Cholesterol; LVEF: Left Ventricular Ejection Fraction; MetS: Metabolic Syndrome; T2DM: Type 2 Diabetes Mellitus.
Introduction
Chronic heart failure (CHF) remains a major public health problem worldwide leading to growth of cardiovascular (CV) morbidity and mortality [1]. During the past decades prevalence and incidence of CHF has increased [2]. Despite contemporary understanding of the underlying disease mechanisms of CHF there is knowledge gap with respect to nature evolution CHF under influence of co-existing CV risk factors [3]. Indeed, the results of few population-based and epidemiological investigations have shown that multiple actual CV risk factors and various metabolic comorbidities presented in CHF patients may affect cardiac failure development [4,5]. There is still debate in the scientific community about whether identification of numerous of CV risk factors / the metabolic co-morbidities improves ability to predict CHF development beyond use of single risk factor [6].
Recent clinical investigations have revealed insulin resistance (IR) is as a distinct cause of cardiac dysfunction and CHF in diabetic and non-diabetic patients [7-9]. IR mediates excessive or inadequate proliferation of the extracellular matrix accelerates apoptosis via increased oxidative stress, neurohumoral and inflammatory activation that effect cardiac remodeling and endothelial function [10-13]. Despite IR is considered a main component of metabolic syndrome (MetS) and type two diabetes mellitus (T2DM), a lot of individuals with CHF may present IR prior to other dysmetabolic conditions including MetS / T2DM [14,15]. However, IR is persisted component of CV risk factors and it role in CHF development in the patients without history of T2DM is still unclear.
Recent studies have shown the association of circulating endothelial cell-derived microparticles (EMPs) with CV risk factors and nature evolution of CHF [16-20]. Extracellular EMPs are defined as microvesicles with sizes ranging between 50 and 1000 nm that released from plasma membrane of endothelial cells due to apoptosis or cell activation by specific (cytokine stimulation, mononuclear cooperation, coagulation, etc) and non-specific (shear stress) stimuli [21]. Apoptotic endothelial cell-derived or activated endothelial cellderived EMPs are capable of transferring biological information (miRNA, DNA), as well as hormones, proteins, lipid components, regulating peptides without direct cell-to-cell contact to maintain cell homeostasis and regulate cell response [22,23]. Interestingly, circulating EMPs derived from activated endothelial cells did not contain nuclear components and they have also been shown to have pro-angiogenic and cardio-protective properties [24,25]. In contrast, apoptotic EMPs realize wide spectrum immune mediators, which generate powerful signaling by the simultaneous receptor interaction. In this context apoptotic EMPs are considered a marker of endothelial cell injury and vascular aging [26,27]. However, the role of different immune phenotypes of EMPs in IR and CHF has not determined. The aim of the study was to assess relationship between IR and immune phenotypes of circulating EMPs in patients with CHF.
Methods
The study prospectively involved 300 ischemic-induced CHF patients aged 48 to 62 years who underwent multispiral computed tomography angiography or coronary angiography between February 2011 and November 2013.
As inclusion criteria in the study we used defined CHF with reduced left ventricular (LV) dysfunction (LV ejection fraction) presented due to stable CAD or myocardial infarction. CHF was defined accordingly clinical practice guideline recommendations as asymptomatic (NYHA I class) and symptomatic (NYHA II-IV classes) associated with declined LVEF (<50%) [28]. All enrolled subjects have demonstrated elevated level of NT-proBNP (>600 pg/ mL). Singes and symptoms of CHF were determined through classes of CHF as sodium and fluid retention, increased jugular venous pressure, peripheral edema, orthopnoea, paroxysmal nocturnal dyspnoea, fatigue. The relevant medical history, certain features / comorbidities were checked and interpreted also. Ischemic-induced CHF has documented when atherosclerotic stenosis > 50% of at least one coronary artery was presented or previously defined myocardial infarction was reported.
We excluded patients with acute infections; active inflammation; pulmonary edema; tachyarrhythmia; valvular heart disease; thyrotoxicosis; ischemic stroke; intracranial hemorrhage; surgery; trauma, autoimmune disease, malignancy, and acute coronary syndrome within 3 months prior to the study entry, and diabetes mellitus (DM). We checked past medical history and the results of laboratory report received prior to the study entry.
DM was diagnosed with revised criteria provided by American Diabetes Association when source documents were reviewed [29]. When one or more of the following components were found (glycated hemoglobin [HbA1c] ≥6.5%; fasting plasma glucose ≥7 mmol/L; 2-h plasma glucose ≥11.1 mmol/L during an oral glucose tolerance test; a random plasma glucose ≥11.1 mmol/L; exposure of insulin or oral antidiabetic drugs; a previous diagnosis of T2DM) Type 2 DM was determined.
All participants gave full written informed consent.
Sample size is calculated by using single population proportion formula by considering the following assumptions; 50% prevalence assumption, 95% confidence level of significance alpha 0.05 = 1.96, and 5% margin of error, which results in the sample size of 299.
Methods for visualization of coronary arteries
Contrast-enhanced multispiral computed tomography angiography has been performed for all patients with dysmetabolic disorder prior to their inclusion in the study on Optima СТ660 scanner (GE Healthcare, USA) using non-ionic contrast “Omnipaque” (Amersham Health, Ireland) [30].
Echocardiography and doppler examination
Transthoracic echocardiography was performed according to a conventional procedure on ACUSON ultrasound system (SIEMENS, Germany) using 2.5-5 МHz phased probe. The LV ejection fraction (EF) was measured by modified Simpson’s method [31].
Insulin resistance assessment
Insulin resistance was assessed by the homeostasis model assessment for insulin resistance (HOMA-IR) [32] using the following formula:
HOMA-IR (mmol/L × μU/mL) = fasting glucose (mmol/L) × fasting insulin (μU/mL) / 22.5
Insulin resistance was defined when estimated HOMA-IR value was over 2.77 mmol/L × μU/mL.
Calculation of glomerular filtration rate
Glomerular filtration rate (GFR) was calculated with CKD-EPI formula [33].
Blood sampling
After an overnight fast blood samples were drawn in the morning (at 7-8 a.m.) into cooled silicone test tubes wherein 2 mL of 5% Trilon B solution were added; then they were centrifuged upon permanent cooling at 6,000 rpm for 3 minutes. Then, plasma was refrigerated immediately to be stored at a temperature -70оС. All laboratory tests were performed using standard methods to measure the serum HbA1c, fasting plasma glucose, fating serum insulin and lipid profiles.
Fasting insulin level was measured by a double-antibody sandwich immunoassay (Elecsys 1010 analyzer, F. Hoffmann-La Roche Diagnostics, Mannheim, Germany). The intra-assay and interassay coefficients of variation were <5%. The lower detection limit of insulin level was 1.39 pmol/L.
Direct Enzymatic HbA1c Assay was used for glycated hemoglobin A1c (HbA1c) measurements on Beckman Synchron LX20 chemistry analyzer.
N-terminal pro-brain natriuretic peptide (NT-pro-BNP) level was measured by immunoelectrochemoluminescent assay using sets by R&D Systems (USA) on Elecsys 1010 analyzer (Roche, Mannheim, Germany).
Concentrations of total cholesterol (TC), cholesterol of highdensity lipoproteins (LDL-C), and cholesterol of high-density lipoproteins (HDL-C) were measured by enzymatic colorimetric method according standardized methodology on Beckman Synchron LX20 chemistry analyzer.
Identifying immune phenotype of EMPs
Circulating MPs were isolated from 5 ml of venous citrated blood drawn from the fistula-free arm. To prevent contamination of samples platelet-free plasma (PFP) was separated from whole blood. PFP was centrifugated at 20,500 × rpm for 30 min. MP pellets were washed with DMEM (supplemented with 10 μg/mL polymyxin B, 100 UI of streptomycin, and 100 U/ml penicillin) and centrifuged again (20,500 rpm for 30 min). The obtained supernatant was extracted, and MP pellets were re-suspended into the remaining 200 μL of supernatant. PFP, MPs, and supernatant were diluted five-, 10-, and five-fold in PBS, respectively. Only 100 μL of supernatant was prepared for further analysis through incubation with different fluorochromelabeled antibodies or their respective isotypic immunoglobulins (Beckman Coulter).
MPs were labeled and characterized by flow cytometry technique per HD-FACS (High-Definition Fluorescence Activated Cell Sorter) methodology independently after supernatant diluted without freeze [34].
CD41a+ was used as a more specific marker of platelets, and CD64+ was considered a more specific marker of monocytes. CD31 antigen was determined as essential marker for endothelial cells, platelets, and leukocytes. CD144+ was used to identify a pure population of endothelial cells. CD31+/annexin V+ and CD144+/ CD31+/annexin V+ MPs were defined as apoptotic endothelial cellderived MPs, MPs labeled for CD105+ or CD62E+ were determined as MPs produced due to activation of endothelial cells [35].
We used anti–CD31 [(platelet endothelial cell adhesion molecule [PECAM]-1)]-phycoerythrin (PE; 20 μl/test), anti–CD41a-PC5 (10 μl/test), anti–CD144 [(vascular endothelial [VE]-cadherin)]- allophycocyanin [APC] (10 μl/test), anti–CD64-FITC (20 μl/test), anti–CD105-FITC (20 μl/test), and anti–CD62E [E-selectin]-FITC (20 μl/test) antibodies obtained from Beckman Coulter. MPs that expressed phosphatidylserine were labeled using fluoresceinconjugated Annexin V solution (20 μl/test; BD Biosciences, USA) in the presence of CaCl2 (5 mM) according to the recommendation of the supplier.
The samples were incubated in the dark for 15 min at room temperature according to the manufacturer’s instructions. It was performed the analysis of area, height, and width forward scatter (FSC) and side scatter (SSC) parameters as well as side scatter width (SSC-W). The gate for MPs was defined by size, using 0.5 and 1.0 μm beads (Sigma, St Louis, MO, USA). For each sample, 500 thousand events have been analyzed. Compensation tubes were used with similar reagents as were used in the sample tubes. Data were constructed as numerous of MPs depending on marker presentation (positive or negative) and determination of MP populations.
Calculation of the number of MPs per liter plasma was based upon the particle count per unit time, the flow rate of the flow cytometer, and the net dilution during sample preparation of the analyzed MP suspension. MP-exposed antigen concentrations were calculated in each sample by multiplying the total concentration of positive MPs by the mean fluorescence intensity of the antigen exposure of the total positive MP population. CD31+/annexin V+ and CD144+/CD31+/ annexin V+ MPs were defined as apoptotic EMPs, EMPs positively labeled for CD62E+ were determined as EMPs produced due to activation of endothelial cells [36].
Statistical analysis
Statistical analysis of the results obtained was carried out in SPSS system for Windows, Version 20 (IBM Corp., Armonk, NY, USA). Baseline and biochemical characteristics were summarized as the mean (M) ± standard deviation (SD) and the median (Ме) and the 25%-75% interquartile range (IQR) for continuous variables, and as absolute numbers and percentages for discrete variables.
The hypothesis of normal distribution of the parameters analyzed was checked by Shapiro–Wilk test and Kolmogorov-Smirnov test. To compare the main parameters of patients’ groups (subject to the type of distribution of the parameters analyzed), one-tailed Student t-test or Mann–Whitney U-test were used. To compare categorical variables between groups, Chi2 test (χ2) and Fisher’s exact test were used. The factors, which could be associated potentially with circulating EMPs, were determined by univariate analysis of variance. Finally, we used univariate and multivariate linear regression to calculate the odds ratio (OR) and a 95% CI for all predictors of elevated circulating EMPs. Statistical significance was accepted for bilateral p< 0.05.
Results
The study population consisted of three hundred none diabetes patients with ischemic-induced CHF (62.0% males) with mean age for 59.50±7.30 years. CHF with reduced and preserved LVEF was found in 37.7% and 62.3% respectively. At least 40.7% patients were obese, 47.7% individuals were dyslipidemic, and 61.3% subjects were hypertensive. General characteristic of the patients included in the study is reported in Table 1.
Parameters
Entire cohort patients (n=300)
Males, n (%)
186 (62.0%)
Age, years
59.50±7.30
Adherence to smoking, n (%)
66 (22.0%)
Hypertension, n (%)
184 (61.3%)
CHFrEF, n (%)
113 (37.7%)
CHFpEF, n (%)
187 (62.3%)
NYHA class I, n (%)
76 (25.3%)
NYHA class II, n (%)
74 (24.7%)
NYHA class III, n (%)
98 (32.7%)
NYHA class IV, n (%)
52 (17.3%)
Dyslipidemia, n (%)
143 (47.7%)
Obesity, n (%)
122 (40.7%)
BMI, kg/m2
24.2 (22.0–27.9)
Systolic BP, mm Hg
129±4
Systolic BP, mm Hg
77±5
Heart rate, beats per 1 min
76±6
LVEF, %
46.07±3.73
GFR, mL/min/1.73 m2
85.2 (70.3–112.5)
HbA1c, %
5.8 (4.3-6.3)
Fasting blood glucose, mmol/L
5.10 (3.4-6.1)
Insulin, μU/mL
13.12 (12.22-14.01)
Creatinine, μmol/L
74.9 (65.1–90.3)
Total cholesterol, mmol/L
5.0 (4.2-5.8)
LDL-C, mmol/L
3.02 (2.80–3.90)
HDL-C, mmol/L
0.88 (0.82–0.97)
NT-pro-BNP, pg /mL
1533.6 (644.5 – 2560.6)
hs-CRP, mg/L
7.34 (6.77-7.95)
ACEI or ARBs, n (%)
300 (100%)
MRA, n (%)
83 (27.7%)
Beta-blockers, n (%)
237 (79.0%)
Aspirin, n (%)
278 (92.7%)
Other antiplatelet drugs, n (%)
22 (7.3%)
Ivabradine, n (%)
89 (29.7%)
Loop diuretics, n (%)
251 (83.7%)
Statins, n (%)
143 (47.7%)
Note: Categorical variables are expressed as numerous (n) and percentages (%), continued variables ae presented as median and interquartile range.
Abbreviations: BP: Blood Pressure; NYHA: New York Heart Association; GFR: Glomerular Filtration Rate; CHFrEF: Chronic Heart Failure with Reduced Ejection Fraction; CHFpEF: Chronic Heart Failure with Preserved Ejection Fraction; HbA1c: Glycated Hemoglobin; HDL-C: High-Density Lipoprotein Cholesterol; LDL-C: Low-Density Lipoprotein Cholesterol; BMI: Body Mass Index; BNP: Brain Natriuretic Peptide; LVEF: Left Ventricular Ejection Fraction; IR: Insulin Resistance; ACEI: Angiotensin-Converting Enzyme Inhibitors; ARBs: Angiotensin Receptor Blockers; MRA: Mineralocorticoid Receptor Antagonists.
Table 1: General characteristic of entire patient group.
All CHF subjects had the mean HOMA-IR of 2.77 ± 2.32 mmol/L × μU/mL. For this study, we used the HOMA-IR > 2.77 mmol/L × μU/mL to quantify insulin resistance in 171 none diabetic individuals. However, patients with HOMA-IR < 2.77 mmol/L × μU/ mL were classified as none IR subjects (n=129). Figure 1 is reported a distribution of HOMA-IR in entire group and both subgroups of CHF subjects enrolled in the study.
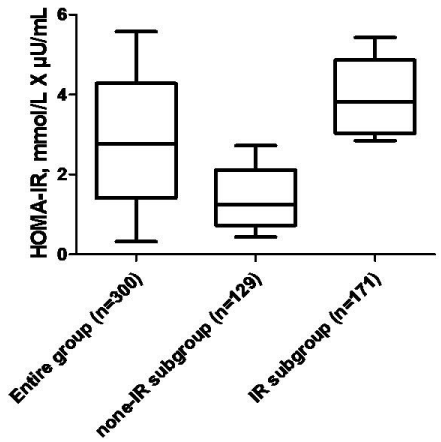
Figure 1: Distribution of HOMA-IR in entire group and both subgroups of CHF
subjects enrolled in the study.
Abbreviations: IR: Insulin Resistance; EMPs: Endothelial-Derived
Microparticles.
The values are presented as the median and 25-75% interquartile range, the
differences validity values obtained by two-tailed Mann-Whitney test.
The patients of both subgroups were age- and sex-matched (Table 2). Cardiovascular risk factors (adherence to smoking, hypertension, dyslipidemia) were found in similar proportion in both subgroups. There was no significant difference in GFR, creatinine, serum total cholesterol, high-density lipoprotein cholesterol between both patient subgroups. Obesity has determined more frequent in IR patients. Additionally, CHF patients with IR had significantly higher BMI, and serum levels of HbA1c, fasting blood glucose, insulin, hs- CRP, low-density lipoprotein cholesterol, and NT-pro-BNP. Systolic and diastolic blood pressures, heart rate in both patient subgroups were comparable. Prescription of concomitant medicine, i.e. ACE inhibitors or ARBs, mineralocorticoid receptor antagonists, diuretics, aspirin, and statins, in both patient subgroups were similar. Betablockers and i/f blocker ivabradine were prescribed statistically much more in CHF subjects with IR when compared with none IR subjects (P=0.016). Other antiplatelet drugs were used frequently in none IR subjects (9.3%) than in IR patients (5.8%; P=0.046).
Parameters
None IR subjects(n=129)
IR subjects (n=171)
P value
Males, n (%)
77 (59.7%)
109 (63.7)
0.23
Age, years
57.90±8.10
60.30±6.33
0.26
Adherence to smoking, n (%)
28 (21.7%)
38 (22.2%)
0.56
Hypertension, n (%)
82 (63.6%)
102 (59.6%)
0.44
CHFrEF, n (%)
49 (38.0%)
64 (37.4%)
0.82
CHFpEF, n (%)
80 (62.0%)
107 (62.6%)
0.86
NYHA class I, n (%)
34 (26.4%)
42 (24.5%)
0.62
NYHA class II, n (%)
32 (24.8%)
42 (24.6%)
0.63
NYHA class III, n (%)
45 (34.9%)
53 (31.0%)
0.60
NYHA class IV, n (%)
18 (13.9%)
34 (19.9%)
0.12
Dyslipidemia, n (%)
58 (45.0%)
85 (49.7%)
0.36
Obesity, n (%)
44 (34.1%)
78 (45.6%)
0.042
BMI, kg/m2
23.07 (22.3–25.7)
25.99(23.5–28.6)
0.054
Systolic BP, mm Hg
131±6
129±5
0.52
Systolic BP, mm Hg
78±4
77±6
0.48
Heart rate, beats per 1 min
75±4
77±6
0.54
LVEF, %
48.62±2.04
44.15±1.98
0.002
GFR, mL/min/1.73 m2
82.8 (71.5–103.1)
87.4 (73.5–110.1)
0.24
HbA1c, %
5.5 (4.7-6.1)
6.1 (5.4-6.5)
0.012
Fasting blood glucose, mmol/L
4.97 (4.87-5.07)
5.47 (5.14-6.0)
0.001
Insulin, μU/mL
10.41 (9.92-10.91)
15.15 (13.69-16.62)
0.016
Creatinine, μmol/L
72.6 (69.31–88.1)
78.6 (70.2–89.1)
0.52
Total cholesterol, mmol/L
4.9 (4.1-5.3)
5.2 (4.5-5.7)
0.21
LDL-C, mmol/L
3.00 (2.82–3.75)
3.11 (2.86–3.82)
0.044
HDL-C, mmol/L
0.91 (0.86–0.95)
0.86 (0.83–0.92)
0.24
NT-pro-BNP, pg /mL
1066.9 (910.3 – 1223.6)
1480.5 (1310.4 – 1650.7)
0.001
hs-CRP, mg/L
7.11 (6.38-7.84)
7.51 (6.68-8.33)
0.016
ACEI or ARBs, n (%)
129 (100%)
171 (100%)
1.0
MRA, n (%)
33 (25.6%)
50 (29.2%)
0.14
Beta-blockers, n (%)
88 (68.21%)
149 (87.1)
0.016
Aspirin, n (%)
117 (90.7%)
161 (94.2%)
0.23
Other antiplatelet drugs, n (%)
12 (9.3%)
10 (5.8%)
0.046
Ivabradine, n (%)
26 (20.2%)
63 (36.8%)
0.026
Loop diuretics, n (%)
109 (84.5%)
142 (83.0%)
0.48
Statins, n (%)
58 (45.0%)
85 (49.7%)
0.36
Note: Categorical variables are expressed as numerous (n) and percentages (%), continued variables are presented as median and interquartile range, the differences validity values obtained by two-tailed Mann-Whitney test.
Table 2: General characteristic of both patient subgroups with and without insulin resistance.
The results of immune phenotyping of EMPs in patients with CHF were presented in Figure 2. One can see these are not significant differences between both cohort patients in EMPs labeled as CD144+/ CD31+, CD144+/annexin V+, and CD62E+ microparticles (Figure 2A, 2B, and 2E). Contrary, higher concentrations of CD144+/CD31+/ annexin V+ EMPs and CD31+/annexin V+ EMPs were found in IR subjects when compared with none IR patients (Figure 2C and 2D).

Figure 2: Numerous of EMPs in entire group and both patient subgroups
depended on IR presentation.
Abbreviations: IR: Insulin Resistance; EMPs: Endothelial-Derived
Microparticles.
The values are presented as the median and 25-75% interquartile range, the
differences validity values obtained by two-tailed Mann-Whitney test.
The univariate linear regression analysis has shown that numerous of CD144+/CD31+/annexin V+ EMPs has associated with NYHA class (r2=0.30; P=0.001), HOMA-IR (r2=0.22; P=0.001), NT-pro-BNP (r2=0.18; P=0.001), LVEF (r2=0.14; P=0.001), CHF with reduced LVEF (r2=0.14; P=0.003), low-density lipoprotein cholesterol (r2=0.11; P=0.001), hs-CRP (r2=0.10; P=0.005), and TG (r2=0.09, P=0.001). The numerous of CD31+/annexin V+ EMPs has related with NYHA class (r2=0.34; P=0.001), HOMA-IR (r2=0.22, P = 0.003), BMI (r2=0.15, P = 0.001), NT-proBNP (r2=0.27, P = 0.001), CHF with reduced LVEF (r2=0.15; P=0.001), hs-CRP (r2=0.18, P = 0.001), GFR (r2=0.15, P=0.001), TG (r2=0.12, P=0.001), creatinine (r2=0.13, P=0.001), gender (r2=0.11, P < 0.001 for male), dyslipidemia (r2=0.098, P = 0.001), age (r2=0.075, P=0.001), hypertension (r2=0.06, P = 0.003), and smoking (r2=0.05, P =0.001).
Univariate linear regression has shown that HOMA-IR, NTproBNP, LVEF, and hs-CRP significantly predicted increased CD31+/annexin V+ EMPs in peripheral blood, but CHF with reduced and preserved LVEF did not demonstrate similar ability (Table 3). Elevated CD144+/CD31+/annexin V+ EMPs were predicted HOMAIR, NT-proBNP, NYHA class, and hs-CRP.
Predictors
Univariate logistic regression
Multivariate logistic regression
Increased CD31+/annexin V+ EMPs
OR (95% CI)
P value
OR (95% CI)
P value
HOMA-IR
1.15 (1.02-1.28)
0.022
1.14 (1.08-1.21)
0.001
NT-proBNP
1.12 (1.06-1.17)
0.001
1.07 (1.04-1.10)
0.001
hs-CRP
1.05 (1.01-1.12)
0.001
1.04 (1.02-1.07)
0.001
NYHA class
1.06 (1.02-1.09)
0.003
1.03 (1.01-1.05)
0.001
LVEF
1.04 (1.00-1.07)
0.044
1.02 (1.00-1.03)
0.12
HFpEF
1.02 (1.00-1.05)
0.62
Not included
HFrEF
1.04 (1.00-1.07)
0.63
Not included
Increased CD144+/CD31+/annexin V+ EMPs
LVEF
1.03 (0.98-1.05)
0.77
Not included
HFpEF
1.03 (0.97-1.06)
0.82
Not included
HFrEF
1.01 (1.00-1.03)
0.88
Not included
BMI
0.98 (0.91-1.05)
0.72
Not included
GFR
0.94 (0.88-1.06)
0.28
Not included
HOMA-IR
1.14 (1.07-1.21)
0.001
1.10 (1.05-1.17)
0.001
NT-proBNP
1.12 (1.05-1.19)
0.001
1.08 (1.04-1.12)
0.001
hs-CRP
1.03 (1.00-1.06)
0.042
1.01 (1.00-1.04)
0.63
NYHA class
1.07 (1.05-1.11)
0.001
1.05 (1.02-1.09)
0.001
Abbreviations: LVEF: Left Ventricular Ejection Fraction; HFpEF: CHF with Precerved LVEF; HFrEF: CHF with Reduced LVEF; BMI: Body Mass Index; GFR: Glomerular Filtration Rate; BNP: Brain Natriuretic Peptide.
Table 3: The independent predictors for elevated apoptotic-derived EMPs. Results of univariate and multivariate logistic regression analyses.
Using multivariate linear regression analyses, we identified independent predictors for elevation of apoptotic-derived EMPs labelled as CD144+/CD31+/annexin V+ and CD31+/annexin V+ microparticles. The results have shown that HOMA-IR, NT-proBNP, hs-CRP, and NYHA class were determined as predictors for increased CD31+/annexin V+ EMPs. Additionally, HOMA-IR, NT-proBNP, and NYHA class significantly predicted an elevation of CD144+/ CD31+/annexin V+ EMPs. No evidences were received regarding independent predictive value of CHF with reduced / preserved LVEF, concomitant medications for CD144+/CD31+/annexin V+ and CD31+/annexin V+ and CD144+/CD31+/annexin V+ EMPs.
Using C-statistics for Models with HOMA-IR, NYHA class, and circulating biomarkers (hs-CRP, NT-proBNP) we found that these biomarkers (NYHA class, hs-CRP, and NT-proBNP) added to the based model constructed with HOMA-IR did not significantly improve the relative IDI for increased CD144+/CD31+/annexin V+ and CD31+/annexin V+ microparticles (Table 4). Moreover, when we used another model constructed on entering variables, IDI avoids to be improved for increased CD144+/CD31+/annexin V+ and CD31+/annexin V+ microparticles (available for NYHA class and circulating hs-CRP and NT-proBNP as continuous variables). Thus, we found that IR remained a statistically significant predictor for increased apoptotic endothelial cell-derived microprticles with phenotypes CD144+/CD31+/annexin V+ and CD31+/annexin V+ microparticles in none-diabetic CHF individuals.
Models
Dependent variable: CD144+/CD31+/annexin V+ EMPs
Dependent variable: CD31+/annexin V+ EMPs
AUC (95% CI)
IDI (±SE)
Relative IDI (%)
AUC (95% CI)
IDI (±SE)
Relative IDI (%)
Model 1
0.669 (0.642-0.691)
-
-
0.664 (0.643-0.687)
-
-
Model 1 + NYHA class versus Model 1
0.674 (0.663-0.695)
0.008±0.005
0.3% P=0.44
0.669 (0.641-0.691)
0.006±0.01
0.2% P=0.63
Model 1 + biomarkers (hs-CRP, NT-proBNP) versus Model 1
0.681 (0.660-0.711)
0.01±0.011
1.2% P=0.42
0.685 (0.648-0.703)
0.009±0.01
1.0% P=0.74
Model 1 + NYHA class + biomarkers (hs-CRP, NT-proBNP) versus Model 1
0.686 (0.640-0.745)
0.02±0.015
1.8% P=0.36
0.689 (0.643-0.718)
0.03±0.012
2.2%
Note: Model 1: HOMA-IR > 2.77 mmol/L × μU/mL; relative IDI – calculated as the ratio of IDI over the discrimination slope of the model without IR.
Abbreviations: AUC: Area Under Curve; SE: Standard Error; IR: Insulin Resistance; BNP: Brain Natriuretic Peptide; hs-CRP: High Sensitive C-Reactive Protein.
Table 4: C-statistics for Models with HOMA-IR, NYHA class, hs-CRP, NT-proBNP as Continuous Variables.
Discussion
In this study we demonstrate that IR in population of CHF patients without history of T2DM may consider a predictor of impaired phenotype of circulating EMPs, which reflects surpass of apoptotic EMPs in circulation association with probably relatively deficiency of activated EMPs in peripheral blood. Recent studies have shown that IR may frequently associate with CHF development [37,38]. Moreover, IR may appear prior to clinical manifestation of LV dysfunction as early finding in asymptomatic CHF [39,40].
To our knowledge IR is associated with important predictors of CV disease among patients with MetS, T2DM, and visceral obesity. However, the role of IR in development of CHF beyond metabolic co-morbidities is not completely clear. In fact, we did not know whether impaired phenotype of circulating EMPs appears to be prior IR or after dysmetabolic disorders. However, IR emerges at early stage of CHF and probably it associates with other CV risk factors [41]. Indeed, elevated level of circulating EMPs was found in several CV diseases and CV risk factors (abdominal obesity, IR, T2DM, hypertension, CHF) and it was associated with severity of vascular tone disorders [42]. Contrary to expectation, in our study an independent association has not verified between the circulating EMPs and such CV factors as smoking, obesity and hypertension, as well as CHF with reduced LVEF.
In fact, EMPs are expressed on their surface CD144 (VE-cadherin) and CD62E (E-selectin) that used as flow cytometry markers to recognized MPs originated from endothelial cells. Therefore, EMPs present on the cell surface other molecules determined the biological role of the MPs and allowed them to interplay in several processes i.e. inflammation, angiogenesis, adhesion, coagulation, cell survival, tissue remodeling, tumor growth and malignancy. It has been postulated that the final result depends closely on origin of the MPs. In fact, MPs derived from activated endothelial cells may play a pivotal role in the angiogenesis and endothelial reparation [43]. In opposite, EMPs originated from apoptotic endothelial cells are considered a direct trigger of vascular injury [44]. It is still unclear whether endothelial dysfunction is result in impaired balance between activated EMPs and apoptotic EMPs or disorders of vascular tone appears to be prior altered secretion of EMPs.
However, the causality relation IR with impaired immune phenotype in CHF patients without history of T2DM is required detail explanation, while the innate exact molecular mechanisms affected abovementioned phenomenon is still under recognized. Probably, oxidative stress and inflammation may elicit or exacerbate IR in CHF subjects [45,46], although our results did not confirm independent causality role of these mechanisms in impaired phenotype of EMPs. Hypothetically, circulating level of MPs released from activated and / or apoptotic endothelial cells may link to deleterious effects on target cells from the vascular wall, adipose tissue, antigen-presenting cells, as well as secretion of cytokines, active molecules, signal proteins, pro-coagulant factors. All these factors might contribute in CHF manifestation and development sometimes beyond traditional CV risk factors and especially at early stages of LV dysfunction. The data obtained within last decade are controversial and are frequently under various speculations. These speculations affect not only different biological effect of EMPs originated from activated and apoptotic endothelial cells, but possibility to define a sufficient imbalance between both immune phenotypes of EMPs in peripheral blood prior to CHF. The deficiency of EMPs with potent angiopoetic capacity associated with elevated level of apoptotic EMPs is frequently found in CV risk factors and CV disease. Indeed, an elevated level of CD31+/annexin V+ apoptotic MPs were detected in coronary artery disease patients and it was positively correlated with endothelial dysfunction and CV disease risk factors i.e. such as smoking, age, male sex, hypertension, dyslipidemia, and T2DM [47,48]. Therefore, MPs shedding from endothelial progenitor cells labeled as CD34+/ KDR+ subset were predictors of aortic stiffness, independent of the Framingham risk [49]. CV disease risk patients with overweight / obesity, diabetes, IR demonstrated impaired inflammatory state and endothelial microvascular function, hypercoagulation, increased arterial stiffness associated with elevated endothelial-derived MPs labeled as CD31+/CD42+, CD31+/CD41-, CD31+/CD62+, and CD144+/CD146+ [50]. Whether different immune phenotypes of circulating EMPs in CV disease play a key role in IR beyond CHF development in this patient population is still not fully understood. We suggested that IR affects wide spectrum target cells including endothelial cells and suppresses their ability to release MPs with “proangiogenic” phenotypes. Probably IR did not allow endothelial cells to be sufficiently resistant to various inflammatory stimuli and direct injury. All these processes may lead to increased apoptotic-derived EMPs that are able to mediate endothelial inflammation and decrease ability to endothelial repair [51].
Finally, beyond traditional CV risk factors impaired phenotype of circulating EMPs in CHF patients reflects limited capacity of endothelial cells to maintain cardiac function in the face of comorbidities such as IR. Probably, imbalance between EMPs with potent “pro-angiogenic” and apoptotic capacities might consider a surrogate biomarker of CHF development. It seems the impaired phenotype of circulating EMPs could significantly have an important prognostic role, while more studies are needed to confirm the assumption among CHF subjects.
Conclusion
We found that IR remained a statistically significant predictor for increased apoptotic-derived EMPs labelled as CD144+/CD31+/ annexin V+ and CD31+/annexin V+ EMPs in none-diabetic patients with CHF.
Limitations of the Study
This study has some restrictions that need to be taken into consideration. First our study is limited by ischemic aetiology of CHF. All enrolled patients have known coronary artery disease defined by angiographically and by received evidence with respect to previous myocardial infarction. Additionally, there were several technical-related difficulties in the measurement of EMPs. In fact, lack of standard protocol for isolating and detecting circulating EMPs obtained from the plasma. The optimal pool of MPs cells identified using combinations of antigenic markers that reflects imbalance in vascular tone enhances and severity of endothelial dysfunction is not determined. According opinion of the majority experts, centrifugation is became the main factor mediated reliability of the EMP determination in samples and contributed in biological variability of EMP count. Although HD-FACS methodology is widely used, theoretically overlap between two or more fluorochromes might reflect some obstacles for further interpretation of obtained results. Another limitation of the present study is that a specific role of EMPs is also possible and has not been characterized in depth in none T2DM patients with CHF. Additionally, relative small sample size may limit the significance of the present study. However, the authors suppose that these restrictions might have no significant impact on the study data interpretation.
Acknowledgement
We thank all patients for their participation in the investigation, staff of the Regional Zaporozhye Hospital (Ukraine) and the doctors, nurses, and administrative staff in City hospital # 6 (Zaporozhye, Ukraine), general practices, and site-managed organizations that assisted with the study.
Ethical Principles
All the patients have given their written informed consent for participation in the study. The investigators followed strictly all the requirements to clinical trials in conformity with the World Medical Association Declaration of Helsinki, 1964, Good Clinical Practice proveided by International Conference on Harmonization, Council of Europe Conventionfor theProtectionof Human Rights and Dignityofthe Human Being in view of using achievements in biology and medicine, Convention on Human Rights and Biomedicine, including Additional Protocol to the Conventionon Human Rights and Biomedicine,concerningBiomedical Research, and legislation of Ukraine.
Authors’ Contributions
AB initiated the hypothesis and designed the study protocol, contributed to collect, analyze and interpret the data, including results of contrast-enhanced multispiral computed tomography angiography, performed statistical analysis, wrote the manuscript and approved final version of the paper. AK enrolled the patients; interpreted data of contrast-enhanced multispiral computed tomography angiography, collected and analyzed the data reviewed the source documents. TB contributed to enroll the patients in the study and collected the data. YM performed of cytofluometry and interpreted of the obtained results. All authors read the manuscript before submitting and agree with final version of the paper.
References
- Maggioni AP. Epidemiology of Heart Failure in Europe. Heart Fail Clin. 2015; 11: 625-635.
- Verdejo HE, Ferreccio C, Castro PF. Heart Failure in Rural Communities. Heart Fail Clin. 2015; 11: 515-522.
- von Lueder TG, Krum H. New medical therapies for heart failure. Nat Rev Cardiol. 2015.
- Goldberg RJ, Spencer FA, Farmer C, Meyer TE, Pezzella S. Incidence and hospital death rates associated with heart failure: a community-wide perspective. Am J Med. 2005; 118: 728-734.
- Baliga V, Sapsford R. Review article: Diabetes mellitus and heart failure--an overview of epidemiology and management. Diab Vasc Dis Res. 2009; 6: 164-171.
- Chinali M, Joffe SW, Aurigemma GP, Makam R, Meyer TE, Goldberg RJ. Risk factors and comorbidities in a community-wide sample of patients hospitalized with acute systolic or diastolic heart failure: the Worcester Heart Failure Study. Coron Artery Dis. 2010; 21: 137-143.
- Greene SJ, Fonarow GC. Insulin resistance in heart failure: widening the divide between reduced and preserved ejection fraction? Eur J Heart Fail. 2015; 17: 991-993.
- Ingelsson E, Sundström J, Arnlöv J, Zethelius B, Lind L. Insulin resistance and risk of congestive heart failure. JAMA. 2005; 294: 334-341.
- Ingelsson E, Arnlöv J, Sundström J, Zethelius B, Vessby B, Lind L. Novel metabolic risk factors for heart failure. J Am Coll Cardiol. 2005; 46: 2054-2060.
- Oka T, Akazawa H, Naito AT, Komuro I. Angiogenesis and cardiac hypertrophy: maintenance of cardiac function and causative roles in heart failure. Circ Res. 2014; 114: 565-571.
- Tsutsui H, Kinugawa S, Matsushima S. Oxidative stress and heart failure. Am J Physiol Heart Circ Physiol. 2011; 301: H2181-2190.
- Shimizu I, Yoshida Y, Katsuno T, Minamino T. Adipose tissue inflammation in diabetes and heart failure. Microbes Infect. 2013; 15: 11-17.
- Tuunanen H., Engblom E., Naum A., Scheinin M, Någren K., Airaksinen J., et al. Decreased myocardial free fatty acid uptake in patients with idiopathic dilated cardiomyopathy: evidence of relationship with insulin resistance and left ventricular dysfunction. J Card Fail. 2006; 12: 644-652.
- Shimizu I, Yoshida Y, Katsuno T, Tateno K, Okada S, Moriya J, et al. p53-induced adipose tissue inflammation is critically involved in the development of insulin resistance in heart failure. Cell Metab. 2012; 15: 51-64.
- Arnlöv J, Lind L, Zethelius B, Andre?n B, Hales CN, Vessby B, et al. Several factors associated with the insulin resistance syndrome are predictors of left ventricular systolic dysfunction in a male population after 20 years of follow-up. Am. Heart J. 2001; 142: 720–724.
- Berezin AE, Kremzer AA, Samura TA, Martovitskaya YV. Circulating endothelial-derived apoptotic microparticles in the patients with ischemic symptomatic chronic heart failure: relevance of pro-inflammatory activation and outcomes. Int Cardiovasc Res J. 2014; 8: 116-123.
- Berezin AE, Kremzer AA. Impaired phenotype of circulating endothelial microparticles in chronic heart failure patients: Relevance to body mass index. Diabetes Metab Syndr. 2015; 9: 230-236.
- Berezin AE, Kremzer AA, Samura TA, Martovitskaya YV, Malinovskiy YV, Oleshko SV, et al. Predictive value of apoptotic microparticles to mononuclear progenitor cells ratio in advanced chronic heart failure patients. J Cardiol. 2015; 65: 403-411.
- Shah KB, Kontos MC. Microparticles and left ventricular assist device complications: a causal association? J Heart Lung Transplant. 2014; 33: 468-469.
- Nozaki T, Sugiyama S, Sugamura K, Ohba K, Matsuzawa Y, Konishi M, et al. Prognostic value of endothelial microparticles in patients with heart failure. Eur J Heart Fail. 2010; 12: 1223-1228.
- Barteneva NS, Fasler-Kan E, Bernimoulin M, Stern JN, Ponomarev ED, Duckett L, et al. Circulating microparticles: square the circle. BMC Cell Biol. 2013; 14: 23.
- Nomura S. Microparticle and Atherothrombotic Diseases. J Atheroscler Thromb. 2015; .
- Wu ZH, Ji CL, Li H, Qiu GX, Gao CJ, Weng XS. Membrane microparticles and diseases. Eur Rev Med Pharmacol Sci. 2013; 17: 2420-2427.
- Tetta C, Bruno S, Fonsato V, Deregibus MC, Camussi G. The role of microvesicles in tissue repair. Organogenesis. 2011; 7: 105-115.
- Martinez MC, Andriantsitohaina R. Microparticles in angiogenesis: therapeutic potential. Circ Res. 2011; 109: 110-119.https://www.ncbi.nlm.nih.gov/pubmed/21852557
- Rautou PE, Vion AC, Amabile N, Chironi G, Simon A, Tedgui A, et al. Microparticles, vascular function, and atherothrombosis. Circ Res. 2011; 109: 593-606.
- Kurtzman N, Zhang L, French B, Jonas R, Bantly A, Rogers WT, et al. Personalized cytomic assessment of vascular health: Evaluation of the vascular health profile in diabetes mellitus. Cytometry B Clin Cytom. 2013; 84: 255-266.
- McMurray JJ, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K, et al. Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Eur J Heart Fail. 2012; 14: 803-869.
- [No authors listed]. Executive summary: Standards of medical care in diabetes--2013. Diabetes Care. 2013; 36 Suppl 1: S4-10.
- Lim MJ, White CJ. Coronary angiography is the gold standard for patients with significant left ventricular dysfunction. Prog Cardiovasc Dis. 2013; 55: 504-508.
- Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American society of echocardiography and the European association of cardiovascular imaging. J Am Soc Echocardiogr. 2015; 28: 1-39.
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985; 28: 412-419.
- Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009; 150: 604-612.
- Orozco AF, Lewis DE. Flow cytometric analysis of circulating microparticles in plasma. Cytometry A. 2010; 77: 502-514.
- Lacroix R, Judicone C, Mooberry M, Boucekine M, Key NS, Dignat-George F. The ISTH SSC Workshop. Standardization of pre-analytical variables in plasma microparticle determination: results of the International Society on Thrombosis and Haemostasis SSC Collaborative workshop. J Thromb Haemost. 2013.
- Tung JW, Parks DR, Moore WA, Herzenberg LA, Herzenberg LA. New approaches to fluorescence compensation and visualization of FACS data. Clin Immunol. 2004; 110: 277-283.
- McGuire DK, Gore MO. Insulin resistance and risk for incident heart failure. JACC Heart Fail. 2013; 1: 537-539.
- Kosmala W, Jellis CL, Marwick TH. Exercise limitation associated with asymptomatic left ventricular impairment: analogy with stage B heart failure. J Am Coll Cardiol. 2015; 65: 257-266.
- Novo G, Pugliesi M, Visconti C, Spatafora P, Fiore M, Di Miceli R, et al. Early subclinical ventricular dysfunction in patients with insulin resistance. J Cardiovasc Med (Hagerstown). 2014; 15: 110-114.
- Vardeny O, Gupta DK, Claggett B, Burke S, Shah A, Loehr L, et al. Insulin resistance and incident heart failure the ARIC study (Atherosclerosis Risk in Communities). JACC Heart Fail. 2013; 1: 531-536.
- Gouya G, Voithofer P, Neuhold S, Storka A, Vila G, Pacher R, et al. Association of nutritional risk index with metabolic biomarkers, appetite-regulatory hormones and inflammatory biomarkers and outcome in patients with chronic heart failure. Int J Clin Pract. 2014; 68: 1293-1300.
- Amabile N, Cheng S, Renard JM, Larson MG, Ghorbani A, McCabe E, et al. Association of circulating endothelial microparticles with cardiometabolic risk factors in the Framingham Heart Study. Eur Heart J. 2014; 35: 2972-2979.
- Cantaluppi V, Biancone L, Figliolini F, Beltramo S, Medica D, Deregibus MC, et al. Microvesicles derived from endothelial progenitor cells enhances neoangiogenesis of human pancreatic islets. Cell Transplant. 2012; 21: 1305-1320.
- Chironi GN, Boulanger CM, Simon A, Dignat-George F, Freyssinet JM, Tedgui A. Endothelial microparticles in diseases. Cell Tissue Res. 2009; 335: 143-151.
- Doehner W, Frenneaux M, Anker SD. Metabolic impairment in heart failure: the myocardial and systemic perspective. J Am Coll Cardiol. 2014; 64: 1388-1400.
- Carvajal K, Balderas-Villalobos J, Bello-Sanchez MD, Phillips-Farfán B, Molina-Muñoz T, Aldana-Quintero H, et al. Ca(2+) mishandling and cardiac dysfunction in obesity and insulin resistance: role of oxidative stress. Cell Calcium. 2014; 56: 408-415.
- Werner N, Wassmann S, Ahlers P, Kosiol S, Nickenig G. Circulating CD31+/annexin V+ apoptotic microparticles correlate with coronary endothelial function in patients with coronary artery disease. Arterioscler Thromb Vasc Biol. 2006; 26: 112-116.
- Blann A, Shantsila E, Shantsila A. Microparticles and arterial disease. Semin Thromb Hemost. 2009; 35: 488-496.
- Pirro M, Schillaci G, Bagaglia F, Menecali C, Paltriccia R, Mannarino MR, et al. Microparticles derived from endothelial progenitor cells in patients at different cardiovascular risk. Atherosclerosis. 2008; 197: 757-767.
- Zhang X, McGeoch SC, Megson IL, MacRury SM, Johnstone AM, Abraham P, et al. Oat-enriched diet reduces inflammatory status assessed by circulating cell-derived microparticle concentrations in type 2 diabetes. Mol Nutr Food Res. 2014; 58: 1322-1332.
- Berezin AE. Impaired Pattern of Endothelial Derived Microparticles in Heart Failure Patients. J Mol Genet Med. 2015; 9: 1.