
Special Article - Nicotine Addiction and Withdrawal
Austin J Drug Abuse and Addict. 2016; 3(1): 1008.
Smoking and Lifestyle-Related Diseases
Kamide K* and Kabayama M
Division of Health Science, Osaka University Graduate School of Medicine, Japan
*Corresponding author: Kei Kamide, Division of Health Science, Osaka University Graduate School of Medicine, Osaka, Japan
Received: December 26, 2015; Accepted: May 19, 2016; Published: May 20, 2016
Abstract
Smoking is a well-known strong risk factor for various malignant neoplasms, chronic obstructive pulmonary disease, and Cardiovascular Diseases (CVD) including stroke, ischemic heart disease based on the progression of atherosclerosis. Although smoking is one of the risk factors for CVD, lifestylerelated diseases such as hypertension, diabetes, dyslipidemia, obesity, and metabolic syndrome commonly advance atherosclerotic CVD. This review summarizes the current topics about the relationship between smoking and lifestyle-related diseases and the effect of smoking cessation on this relation and makes use of health promotion to prevent atherosclerotic CVD induced by smoking.
Keywords: Smoking; Lifestyle-related diseases; Cardiovascular diseases; Cessation
Introduction
Smoking is a well-known strong risk factor for various malignant neoplasms, Chronic Obstructive Pulmonary Disease (COPD), and Cardiovascular Diseases (CVD) including stroke, Ischemic Heart Disease (IHD), and Atherosclerosis Obliterans (ASO) based on the progression of atherosclerosis. Mechanisms of the strong association between smoking and CVD are thought to be due to induced oxidative stress, vascular inflammation, platelet coagulation, vascular endothelial dysfunction with reduced Nitric Oxide (NO) bioavailability and increased expression of adhesion molecules [1], and impaired serum lipid profiles including low HDL cholesterol [2] by cigarette smoking. Although smoking is one of the risk factors for CVD, lifestyle-related diseases such as hypertension, diabetes, dyslipidemia, obesity, and metabolic syndrome commonly advance atherosclerotic CVD. Smoking may also influence these lifestylerelated diseases. Therefore, smoking not only directly induces the progression of atherosclerosis but it may also progress lifestyle-related diseases and consequently cause atherosclerosis in both current and past smokers [3,4]. Smoking cessation is very important as a healthpromoting measure to reduce malignant neoplasms, COPD, and CVD. Various recent approaches including drug intervention for smoking cessation are useful. This review summarizes the current topics about the relationship between smoking and lifestyle-related diseases and the effect of smoking cessation on this relation.
Smoking and Lifestyle-Related Diseases
Hypertension
It is well-known that smoking acutely increases Blood Pressure (BP) mainly by the sympathetic nerve activation induced by nicotine exposure [5,6]. In contrast, chronic influences of smoking are controversial. Most studies before 1990 indicated a lower BP among smokers compared with nonsmokers after adjustment for the body weight and alcohol drinking [5]. However, recent epidemiological studies showed that smoking was a risk for the incidence of hypertension both in women [7] and men [8]. What is the cause of this discrepancy? Bowman TS et al. reported the risk of incident hypertension in women by a large-scale prospective study.
In that study, they followed 28,236 women for an average of 9.8 years, and demonstrated that women smoking >15 cigarettes/day had a significant risk of incident hypertension [7]. Halperin RO et al. also reported a similar large-scale prospective study about smoking and incident hypertension for middle-aged and elderly men. They followed 13,529 middle-aged and elderly men for an average of 14.5 years, and both current and past smokers had a significant risk of incident hypertension in this study [8]. In contrast, past studies were reviewed in Green’s paper [5]. Cross-sectional designed studies and studies including small samples reported results that current and past smokers showed a lower BP compared with non-smokers; however, a few prospective studies with a short follow-up duration in this paper showed inconsistent results regarding the association of smoking with BP [5]. Therefore, we suggest that the study design, cross-sectional or prospective, sample size, and follow-up duration are probably playing key roles in the investigation of the relationship between smoking and hypertension. Most of the studies on the effects of smoking cessation on the BP indicated that the BP was significantly lower several days after smoking cessation and the lower BP compared with the previous BP in the smoking period was maintained [9,10]. From a pathophysiological point of view, since smoking proceeds the progression of atherosclerosis and arteriosclerosis, increased peripheral vascular resistance by these vascular changes may cause BP elevation. We can conclude that smoking is clearly a risk factor for elevation of the BP, especially systolic BP, and incidence of hypertension.
Diabetes
Smoking is known as a risk of type 2 diabetes based on some prospective cohort studies [11-13]. Rimm EB et al. reported that 114,247 women without diabetes or cancers were followed for 12 years in the Nurse Health Study and 2,333 subjects developed newly diagnosed diabetes (1.8 /1,000 persons/year), in which those smoking >25 cigarettes/day showed a Hazard Ratio (HR) for the incidence of diabetes of 1.37 (95% CI: 1.16-1.62) compared with non-smokers [11]. Similar results were found in male medical professionals in a 6-year follow-up study [12]. A significant risk of diabetes was noted in male smokers smoking 14-24 cigarettes /day with HR 2.38 (95% CI: 1.57-3.59) and those smoking >25 cigarettes /day showed HR 1.94 (95% CI: 1.25-3.03) compared with non-smokers [12]. In a Japanese prospective cohort study, male smokers had a significant risk of diabetes, with HR 3.27 (95% CI: 1.18-9.09) in subjects smoking 15- 25 cigarettes/day and HR 3.21 (95% CI: 1.05-9.83) in those smoking >25 cigarettes/day [13]. Since there were contradictory reports about the relationship between smoking and diabetes using information on only the smoking history, smoking more than 15 cigarettes /day may be very important to develop new onset diabetes. Concerning the progression of diabetic complications, it was reported that diabetic nephropathy more rapidly progressed in smokers than in nonsmokers in some studies [14,15]. Regarding the mechanisms of the progression of smoking-induced diabetes, it is possible that Metabolic Syndrome (MetS) induced by smoking, as mentioned below, will progress to diabetes. Insulin resistance promoted by smoking may be associated with MetS and diabetes [16].
Lipid abnormality
Smoking increases the rate of CVD via alterations in the lipid profile, and particularly through its impact on HDL. Craig WY et al. reported based on a comprehensive meta-analysis that individuals who smoked cigarettes when compared with non-smokers had significantly higher Total Cholesterol (TC) (3%), Triglycerides (TG) (9.1%), and VLDL (10.4%), higher but not significant LDL (1.7%), and lower concentrations of HDL (-5.7%) and Apolipoprotein AI (Apo A-I) (-4.2%) [17]. They also indicated a dose-response relationship between the number of cigarettes per day and the extent of lipid abnormalities.
A study by McCall MR et al. showed that lecithin cholesterol acyl-transferase, the enzyme to maintain adequate levels of HDL, is very sensitive to cigarette smoking and that this could be directly responsible for the HDL changes in smokers [18].
Freeman DJ et al. suggested that reduced Lipoprotein Lipase (LPL) activity induces impaired TG clearance, which was commonly seen in smokers due to a slower metabolism of TG-rich lipoproteins [19]. Also, LPL activity in skeletal muscle is affected by insulin, and several studies have shown insulin resistance among smokers as one possible mechanism, which we discussed in the previous section [20]. The amount of LPL activity is negatively correlated with the amount of released insulin; the amount of released insulin may be positively correlated with the amount of nicotine consumed per day. Since LPL release is not stimulated, the circulating TG concentration is increased; this perturbation causes the excess VLDL as a major risk factor for CVD [21].
Obesity, metabolic syndrome
Obesity and MetS are well-known strong risks for CVD. Obese individuals with smoking have a 14-year reduction in life expectancy at the age of 40 years in the Framingham study [22]. A large prospective study showed that smoking coupled with obesity contributes substantially to all-cause mortality with a 3.5-5 fold greater risk for severely obese current smokers than for normal weight nonsmokers [23]. Especially, central obesity based on visceral fat accumulation is a big issue clinically because of a major cause of MetS. Recently, there has been some evidence indicating a close relationship between smoking and MetS. Sun K, et al. reported that active smoking was a clear risk (Hazard Ratio (HR) 1.26) of MetS by meta-analysis of prospective studies including 13 studies [24]. About the close relationship between smoking and MetS, it is reported that waist circumference is significantly higher in obese subjects with smoking than in those without smoking [25]. Smoking may accelerate visceral fat accumulation and promote obesity-related disorders. Waist circumference is strongly associated with visceral fat mass [26], which is influenced on the plasma cortisol concentration [27]. Smokers have higher fasting plasma cortisol concentrations than nonsmokers [28,29]. Higher cortisol concentrations may stimulate sympathetic nervous system activity, which is induced by smoking [30,31]. Other possible mechanisms of smoking-induced MetS may be explained by insulin resistance and glucose impairment due to smoking [16]. This insulin resistance induced by smoking will result in the development of DM, as mentioned above.
About amounts of smoking exposure, it is reported that subjects who habitual smoking had a 1.07-1.66 fold greater risk of developing MetS than subjects who are not smoker [32]. Weitzman M et al. [33] have demonstrated for the first time a dose-responsive, cotinineconfirmed relationship between smoking and the severity of MetS. Weitzman’s study also demonstrated the positive association between smoking and MetS in adolescents. In this study, it is reported that exposure to smoking, whether through active or passive smoking, is associated with 4 fold increase in the risk of the MetS among adolescents who are overweight or at risk of overweight. The association between adolescent smoking and overweight or abdominal obesity in adulthood investigated by Saarni SE et al. [34]. In consequence, smoking is a risk factor for abdominal obesity in both sexes and for overweight in women. Furthermore, it has been reported that smoking aggravates the risk of CVD associated with MetS based on a Japanese prospective epidemiological study [35]. From this report, smokers without MetS have a significant risk of CVD (HR: 2.07 in men, 2.67 in women) compared with non-smokers without MetS, and smokers with MetS have a significantly higher risk of CVD (HR: 3.56 in men, 4.84 in women) compared with nonsmokers without MetS [35]. Therefore, smoking cessation will be more important for people with than without MetS.
Meanwhile, numerous cross-sectional studies have indicated that BMI is lower in smokers than in nonsmokers [36-38]. However, Kim BJ et al. [39] reported that continuous smokers show a greater weight gain than nonsmokers and they speculated that continuous smokers are less physically active. These findings indicate that greater emphasis should be placed on the risk of central obesity, insulin resistance, and associated conditions among smokers, especially for female whose major reasoning for smoking is weght control [40]. Smokers should be made aware that smoking is not an efficient way to control body weight, does not help prevent obesity, and will be inducing visceral fat accumulation and increase the risk of MetS, diabetes and incidence of CVD.
Smoking Cessation as an Important Health Promotion Strategy
From here, we will discuss smoking cessation. Previous studies showed that many physiologic, psychological, and sociological benefits are associated with smoking cessation (summary of benefits in Table 1) [41]. Current data, however, show that fewer than half of smokers ever achieve long-term abstinence, despite 70-80% wanting to quit and one-third having made at least three serious attempts to do so [42]. Part of the reason for continued smoking is the addictive nature of nicotine, a substance found in all types of tobacco product, which makes smoking cessation challenging. In the Surgeon General’s Report in the Health Consequences of Smoking (1988), nicotine was declared an addictive drug similar to heroin or cocaine [43].
Short-Term Benefits
Long-Term Benefits
Physiological benefit
• Blood pressure returns to pre-smoking levels (within 20 minutes).
• Carbon monoxide drops within hours.
• The ratios of HDL-C : TC and HDL -C : LDL-C alter (promote enhanced clearance of cholesterol from the circulation).
• Risk of coronary heart disease is reduced by 50% after 1 year.
• Risk of stroke is similar to that of a nonsmoker within 5 to 15 years.
• Risk of lung cancer is reduced.
• Risk of COPD is reduced.
• Lung function improves up to 30% within 2 to 3 months.
Psycho - social benefit
• Sense of smell and taste improve within days.
• Money is saved each day by not buying cigarettes.
• Patient earns greater self-respect because of a real sense of accomplishment in quitting.
• Patient enjoys increased self-esteem due to quitting smoking.
Table 1: Benefits of smoking cessation.
Despite nicotine’s strong addictive nature, various effective treatments to assist people quit smoking exist [44]. The guideline released by the US Public Health Service reported clear scientific evidence that 5 different medications are helpful in assisting people who wish to quit smoking (bupropin, sustained-release, nicotine gum, nicotine inhaler, nicotine nasal spray, and nicotine patch) [45,46]. They also found evidence that certain types of counseling increased a person’s likelihood of quitting permanently. The guideline recommended that smokers be offered both counseling and medication to increase their chance of success.
The potential impacts of treatments for tobacco dependence not only depend on their efficiency but also the extent to which these treatments reach those who might benefit from them. Today, there is little evidence to support the idea that therapies for treating nicotine dependence have significantly reduced the rates of smoking at a population level. The main reason for this small effect is the generally low utilization of these therapies, which may be due in part to the reluctance to prescribe smoking cessation medications to patients with diabetes because of fear of post-cessation weight gain. Recent evidence has indicated an increase in the risk of diabetes post-cessation. Potential weight gain continues to be a key barrier to quitting smoking, and the increased risk of developing diabetes following cessation may elevate this concern further. Considering the physiologic, psychological, and sociological benefits of smoking cessation, interventions for smokers that combine smoking cessation interventions with weight gain prevention should be needed among patients with life-related diseases [47-49]. Another potential reason for the low utilization of the smoking cessation medications may be the insufficient awareness of health-care workers who actively assist their tobacco-using patients in quitting [50]. While many clinical trials have proved the effectiveness of smoking cessation medications, few smokers use them when they try to quit. Previous studies reported that the most effective interventions (such as faceto- face counseling or medication) also have the lowest use rate, and interventions with the highest use rate have low efficiency (e.g., mass media campaigns) [51,52]. It would be desirable for medical workers, public health community members, and governments to do more to reduce smoking on considering the evidence indicating that smoking cessation has major and immediate health benefits. The extensive research provides strong support for the cessation of smoking as a means to reduce on excess risk of lifestyle-related diseases.
Effects of Smoking Cessation on CVD via Protecting Against Lifestyle-Related Diseases
Early studies reported that individuals who quit smoking had a substantial decrease in risk for acute myocardial infarction as compared to those who continued to smoke [53,54]. It has been observed that smoking cessation can delay the prevalence of atherosclerosis by 10 years as compared to individuals who continue to smoke [55]. Previous study clarified smoking cessation could be inducing a significant increase in the HDL-C levels in randomized controlled study [41]. Besides, smoking is a clear risk of lifestyle-related diseases such as hypertension, diabetes, dyslipidemia, and MetS, as we have reviewed in this paper, smoking cessation will definitely be helpful to improve these lifestyle-related diseases and consequently reduce the incidence of CVD. Although smoking cessation is not easy, as mentioned above, Nicotine Replacement Therapy (NRT) is helpful to promote the success of smoking cessation. Regarding the relationship between NRT and lifestyle-related diseases, previous studies also investigated the physiologic effects of the usage of NRT [56]. Some early studies indicated that the administration of a nicotine patch does not alter lipids or lipoproteins. However, a more recent study by Moffat RJ et al. suggested that nicotine prevents the normalization of HDL-C and its subfractions, and that effects are acute and persistent only as long as the patch is in place [57]. Based on this study, we suggest that NRT is a desirable way for successful smoking cessation, assuming that the use of NRT is temporary. Approaches to smoking cessation are progressing. We need to clarify the effects of smoking cessation on each lifestyle-related disease. Since there are few studies that clarified the smoking cessation can directly improve the hypertension or diabetes, future studies are desirable to prove it.
Conclusion
We have reviewed the process of smoking-related CVD focusing on the links between smoking and lifestyle-related diseases (Figure 1). Smoking directly promotes atherosclerosis and arteriosclerosis in small vessels and increases the risk of CVD. In addition, smoking causes the incidence and progression of lifestyle-related diseases, hypertension, diabetes, dyslipidemia, and MetS. These smoking-related diseases also promote the incidence of CVD via atherosclerosis. Therefore, smoking cessation is definitely important not only to prevent malignant neoplasm and lung diseases, but also CVD and lifestyle-related diseases induced by smoking.
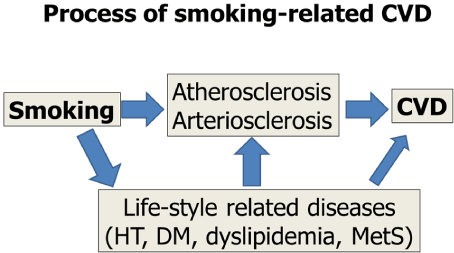
Figure 1: Process of smoking-related CVD.
CVD: Cardiovascular Diseases; HT: Hypertension; DM: Diabetes Mellitus;
MetS: Metabolic Syndrome.
References
- Aida J, Kondo K, Hirai H, Nakade M, Yamamoto T, Hanibuchi T, et al. Association between dental status and incident disability in an older Japanese population. Journal of the American Geriatrics Society. 2012; 60: 338-343.
- Stewart R, Hirani V. Dental health and cognitive impairment in an English national survey population. J Am Geriatr Soc. 2007; 55: 1410-1414.
- Fukutomi E, Okumiya K, Wada T, Sakamoto R, Ishimoto Y, Kimura Y, et al. Importance of cognitive assessment as part of the “Kihon Checklist” developed by the Japanese Ministry of Health, Labor and Welfare for prediction of frailty at a 2-year follow up. Geriatrics & gerontology international. 2013; 13: 654- 662.
- Mitnitski AB, Graham JE, Mogilner AJ, Rockwood K. Frailty, fitness and latelife mortality in relation to chronological and biological age. BMC Geriatr. 2002; 2: 1.
- Crimmins EM, Saito Y. Trends in healthy life expectancy in the United States, 1970-1990: gender, racial, and educational differences. Soc Sci Med. 2001; 52: 1629-1641.
- Manton KG. A longitudinal study of functional change and mortality in the United States. J Gerontol. 1988; 43: 153-161.
- Bowman TS, Gaziano JM, Buring JE, Sesso HD. A prospective study of cigarette smoking and risk of incident hypertension in women. J Am Coll Cardiol. 2007; 50: 2085-2092.
- Halperin RO, Gaziano JM, Sesso HD. Smoking and the risk of incident hypertension in middle-aged and older men. Am J Hypertens. 2008; 21: 148- 152.
- Mattos IE, do Carmo CN, Santiago LM, Luz LL. Factors associated with functional incapacity in elders living in long stay institutions in Brazil: a crosssectional study. BMC geriatrics. 2014; 14: 47.
- Avila-Funes JA, Helmer C, Amieva H, Barberger-Gateau P, Le Goff M, Ritchie K, et al. Frailty among community-dwelling elderly people in France: the three-city study. J Gerontol A Biol Sci Med Sci. 2008; 63: 1089-1096.
- Ogawa KFY, Yoshida H, Nishi M, Fukaya T, Kim M, et al. The validity of the “Kihon Checklist” as an index of frailty and its biomarkers and inflammatory markers in elderly people. Nihon Ronen Igakkai Zasshi. 2011; 48: 545-552.
- Kabayama MKK, Sakakibara K, Hayakawa K. The Role of public health nurses in Japanese long-term care prevention projects in the community. J Nurs Care. 2014; 3:166.
- Kawakami N, Takatsuka N, Shimizu H, Ishibashi H. Effects of smoking on the incidence of non-insulin-dependent diabetes mellitus. Replication and extension in a Japanese cohort of male employees. Am J Epidemiol. 1997; 145: 103-109.
- Ministry of Health Law. Manual for the long-term care prevention.
- Umegaki H, Suzuki Y, Yanagawa M, Nonogaki Z, Nakashima H, Kuzuya M, et al. Cognitive impairments and functional declines in older adults at high risk for care needs. Geriatrics & gerontology international. 2013; 13: 77-82.
- Piatti P, Setola E, Galluccio E, Costa S, Fontana B, Stuccillo M, et al. Smoking is associated with impaired glucose regulation and a decrease in insulin sensitivity and the disposition index in first-degree relatives of type 2 diabetes subjects independently of the presence of metabolic syndrome. Acta Diabetologica. 2014; 51: 793-799.
- Craig WY, Palomaki GE, Haddow JE. Cigarette-Smoking and Serum-Lipid and Lipoprotein Concentrations - an Analysis of Published Data. Brit Med J. 1989; 298: 784-788.
- Mc Call MR, Van den Berg JJ, Kuypers FA, Tribble DL, Krauss RM, Knoff LJ, et al. Modification of LCAT activity and HDL structure. New links between cigarette smoke and coronary heart disease risk. Arterioscler Thromb. 1994; 14: 248-253.
- Freeman DJ, Griffin BA, Murray E, Lindsay GM, Gaffney D, Packard CJ, et al. Smoking and Plasma-Lipoproteins in Man-Effects on Low-Density- Lipoprotein Cholesterol Levels and High-Density-Lipoprotein Subfraction Distribution. Eur J Clin Invest. 1993; 23: 630-640.
- Facchini FS, Hollenbeck CB, Jeppesen J, Chen YD, Reaven GM. Insulin resistance and cigarette smoking. Lancet. 1992; 339: 1128-1130.
- Eliasson B, Attvall S, Taskinen MR, Smith U. The insulin resistance syndrome in smokers is related to smoking habits. Arterioscler Thromb. 1994; 14: 1946- 1950.
- Peeters A, Barendregt JJ, Willekens F, Mackenbach JP, A Mamun A, Bonneux L, et al. Obesity in adulthood and its consequences for life expectancy: a lifetable analysis. Ann Intern Med. 2003; 138: 24-32.
- Freedman DM, Sigurdson AJ, Rajaraman P, Doody MM, Linet MS, Ron E. The mortality risk of smoking and obesity combined. Am J Prev Med. 2006; 31: 355-362.
- Sun K, Liu J, Ning G. Active smoking and risk of metabolic syndrome: a metaanalysis of prospective studies. PLoS One. 2012; 7: 47791.
- Mizuno O, Okamoto K, Sawada M, Mimura M, Watanabe T, Morishita T. Obesity and smoking: relationship with waist circumference and obesityrelated disorders in men undergoing a health screening. J Atheroscler Thromb. 2005; 12: 199-204.
- Han TS, Sattar N, Lean M. ABC of obesity. Assessment of obesity and its clinical implications. BMJ. 2006; 333: 695-698.
- Pasquali R, Vicennati V. Activity of the hypothalamic-pituitary-adrenal axis in different obesity phenotypes. Int J Obes Relat Metab Disord. 2000; 24: 47-49.
- Cryer PE, Haymond MW, Santiago JV, Shah SD. Norepinephrine and Epinephrine Release and Adrenergic Mediation of Smoking-Associated Hemodynamic and Metabolic Events. New Engl J Med. 1976; 295: 573-577.
- Friedman AJ, Ravnikar VA, Barbieri RL. Serum Steroid-Hormone Profiles in Postmenopausal Smokers and Nonsmokers. Fertil Steril. 1987; 47: 398-401.
- Williamson DF, Madans J, Anda RF, Kleinman JC, Giovino GA, Byers T. Smoking cessation and severity of weight gain in a national cohort. N Engl J Med. 1991; 324: 739-745.
- Yoshida T, Sakane N, Umekawa T, Kogure A, Kondo M, Kumamoto K, et al. Nicotine induces uncoupling protein 1 in white adipose tissue of obese mice. Int J Obes Relat Metab Disord. 1999; 23: 570-575.
- Nakanishi N, Takatorige T, Suzuki K. Cigarette smoking and the risk of the metabolic syndrome in middle-aged Japanese male office workers. Ind Health. 2005; 43: 295-301.
- Weitzman M, Cook S, Auinger P, Florin TA, Daniels S, Nguyen M, et al. Tobacco smoke exposure is associated with the metabolic syndrome in adolescents. Circulation. 2005; 112: 862-869.
- Saarni SE, Pietilainen K, Kantonen S, Rissanen A, Kaprio J. Association of smoking in adolescence with abdominal obesity in adulthood: a follow-up study of 5 birth cohorts of Finnish twins. Am J Public Health. 2009; 99: 348- 354.
- Higashiyama A, Okamura T, Ono Y, Watanabe M, Kokubo Y, Okayama A. Risk of Smoking and Metabolic Syndrome for Incidence of Cardiovascular Disease-Comparison of Relative Contribution in Urban Japanese Population: The Suita Study. Circ J. 2009; 73: 2258-2263.
- Bouros D, Tzouvelekis A, Anevlavis S, Doris M, Tryfon S, Froudarakis M, et al. Smoking acutely increases plasma ghrelin concentrations. Clin Chem. 2006; 52: 777-778.
- Klesges RC, Winders SE, Meyers AW, Eck LH, Ward KD, Hultquist CM, et al. How much weight gain occurs following smoking cessation? A comparison of weight gain using both continuous and point prevalence abstinence. J Consult Clin Psych. 1997; 65: 286-291.
- La Rowe TL, Piper ME, Schlam TR, Fiore MC, Baker TB. Obesity and Smoking: Comparing Cessation Treatment Seekers with the General Smoking Population. Obesity. 2009; 17: 1301-1305.
- Kim BJ, Kim BS, Sung KC, Kang JH, Lee MH, Park JR. Association of Smoking Status, Weight Change, and Incident Metabolic Syndrome in Men: A 3-Year Follow-Up Study. Diabetes Care. 2009; 32: 1314-1316.
- Vidrine JI, Anderson CB, Pollak KI, Wetter DW. Gender differences in adolescent smoking: mediator and moderator effects of self-generated expected smoking outcomes. American journal of health promotion: AJHP. 2006; 20: 383-387.
- Nilsson P, Lundgren H, Soderstrom M, Fagerstrom KO, Nilsson-Ehle P. Effects of smoking cessation on insulin and cardiovascular risk factors--a controlled study of 4 months’ duration. J Intern Med. 1996; 240: 189-194.
- WHO. Action for Tobacco Control. Guideline for Controlling and Monitoring the Tobacco Epidemic. 1998.
- Henningfield JE, Higgins ST. The influence of behavior analysis on the surgeon general’s report, the health consequences of smoking: Nicotine addiction. The Behavior analyst / MABA. 1989; 12: 99-101.
- Jorenby DE. Smoking cessation strategies for the 21st century. Circulation. 2001; 104: 51-52.
- Fiore MC, Jaen CR, Baker TB, Bailey WC, Benowitz NL, Curry SJ, et al. Treating tobacco use and dependence: 2008 update US Public Health Service Clinical Practice Guideline executive summary. Resp Care. 2008; 53: 1217-1222.
- Waugh JB, Lorish CD. New guideline on treating tobacco use and dependence: 2008 update is an opportunity to advance. Respir Care. 2008; 53: 1166-1168.
- Fiore MC, Baker TB. Should clinicians encourage smoking cessation for every patient who smokes? Jama. 2013; 309: 1032-1033.
- Oba S, Noda M, Waki K, Nanri A, Kato M, Takahashi Y, et al. Smoking Cessation Increases Short-Term Risk of Type 2 Diabetes Irrespective of Weight Gain: The Japan Public Health Center-Based Prospective Study. Plos One. 2012; 7.
- Luo J, Rossouw J, Margolis KL. Smoking cessation, weight change, and coronary heart disease among postmenopausal women with and without diabetes. Jama. 2013; 310: 94-96.
- Paoletti L, Jardin B, Carpenter MJ, Cummings KM, Silvestri GA. Current status of tobacco policy and control. J Thorac Imaging. 2012; 27: 213-219.
- Raw M, McNeill A. The prevention of tobacco-related disease. Addiction. 1994; 89: 1505-1509.
- Shahab L, Mc Ewen A. Online support for smoking cessation: a systematic review of the literature. Addiction. 2009; 104: 1792-1804.
- Hammond EC, Horn D. Smoking and death rates-report on forty-four months of follow-up of 187,783 men. By E. Cuyler Hammond and Daniel Horn, 1958. CA: a cancer journal for clinicians. 1988; 38: 28-58.
- Doll R, Hill AB. The mortality of doctors in relation to their smoking habits; a preliminary report. British medical journal. 1954; 1: 1451-1455.
- Feeman WE Jr. The role of cigarette smoking in atherosclerotic disease: an epidemiologic analysis. J Cardiovasc Risk. 1999; 6: 333-336.
- Allen SS, Hatsukami D, Gorsline J. Cholesterol Changes in Smoking Cessation Using the Transdermal Nicotine System. Preventive medicine. 1994; 23: 190-196.
- Moffatt RJ. Effects of cessation of smoking on serum lipids and high density lipoprotein-cholesterol. Atherosclerosis. 1988; 74: 85-89.