
Research Article
J Drug Discov Develop and Deliv. 2017; 4(1): 1026.
A Review of Acute Opiate Poisoning in the South West of Iran
Jalali A1¹*, Hasan Rahmani A², Moosavi M¹, Mehrabi L², Saki A³ and Rowan EG4
¹Department of Pharmacology and Toxicology, School of Pharmacy and Toxicology Research Center, Ahvaz Jundishapur University of Medical Science, Iran
2Department of Clinical Toxicology, Razi Hospital and Toxicology Research Center, Ahvaz Jundishapur University of Medical Science, Iran
3Department of Statistics, School of Health, Ahvaz Jundishapur University of Medical Science, Iran
4Institute of Pharmacy and Biomedical Sciences, University of Strathclyde, UK
*Corresponding author: Jalali A, Department of Pharmacology and Toxicology, School of Pharmacy and Toxicology Research Center, Ahvaz Jundishapur University of Medical Science, Iran
Received: February 02, 2017; Accepted: March 13, 2017; Published: March 21, 2017
Abstract
Background: Evidences on the exact burden and pattern of acute opiate poisoning in the southwest of Iran (Ahvaz, Khuzestan province) are limited.
Objectives: We aimed to characterize acute opiate poisoning with regard to demographic and epidemiological factors of cases registered at Razi General Teaching Hospital, Jundishapur University; main referral poisoning ward in the southwest of Iran.
Methods: This was a retrospective collection of data on all acute opiate poisoning cases recorded from January to December 31 2007.
Results: Of 264 cases (84.8% male, 15.2% female, mean age of 28.5±14 years), 62.5% were aged between 4-25, 22% between 26-40 and 15.5% of patients were 40 years and over. The main drugs implicated in opioid overdosing were tramadol in 69.3% (183 cases), opium in 27.3% (72 cases), and heroin in 2.7% (7 cases). The most common symptom of the intoxication was, decreased consciousness (54.5%) followed by nausea and vomiting (17.8%), drowsiness (15.2%) and seizure (12.5%). Seizure was more common in the group of patients who were taking tramadol and were less than 25 years of age. Most cases were referred to hospital within six hours; the death rate was 1.9%. Acute opiate poisoning was associated with not high mortality rate than reported elsewhere. Analysis of cases with seizure (male 90.9% vs. female, 9.1%) showed that most seizures happened in age group less than 25.
Conclusion: This demonstrates a pressing need from policy-makers for awareness that tramadol using may increase the mortality in young adults - the most affected group and further calls for the use family approaches in raising this awareness.
Keywords: Opioid; Tramadol; Age-dependent; Seizure; Poisoning
Introduction
Acute opiate poisoning remains a significant public health problem in many developing countries. Furthermore, there are almost a couple of millions suicides each year, and a significant number of deaths are related to opiate poisoning. Overdose deaths from acute opiate poisoning become more prevalent in developing countries [1-6]. In the southwest of Iran, Khuzestan province, acute opiate poisoning has not been identified well as a significant cause of both morbidity and mortality with hospital referral. Tramadol is a centrally acting opioid-receptor agonist that is extensively dispensed analgesic in more than 100 countries. Tramadol has been marketed in Iran since 1995. This drug was employed to accelerate opioid detoxification by displacing opiate and as a maintenance agent for detoxified formerly opiate-dependent patients who want to remain opioid-free [1]. It is being abused by opioid addicted subjects. Its main effects including multimodal antinociceptive and analgesic via two mechanisms: opioid through μ-opioid receptors and nonopioid component related to inhibition of neuronal 5-hydroxytryptamine (5-HT; serotonin) [2]. Tramadol like other opioid drugs (opiates) is an agonist of the μ receptor. Its affinity is moderate and weaker than morphine. This low affinity at receptor causes relieving properties are about 10 times less than morphine. Tramadol prevents the reuptake of noradrenaline and serotonin in the synapses like antidepressants and act consumer awareness and active unlike morphine. This synthetic drug has two significant adverse reactions: seizure and serotonin syndrome [3-6]. These adverse reactions may occur even during normal doses in animals and humans [4,6].
Seizure is a serious complication that may associate with medication or drug use [7]. Clinical factors for complications of drug-induced seizures are stimulant exposure, suicide attempt, initial hypotension, and admission acidosis or hypoglycemia [8]. Drug overdose or ethanol which is responsible for 18% cases in seizure occurrence has fatality rate over 20% [9].
The lack of up-to-date information concerning acute opiate poisoning in the southwest of Iran, Khuzestan province can be attributed to the unavailability of published data in accessible databases, a deficiency in national surveillance systems, including the non-mandatory notification of poisoning cases. This lack of information is a barrier to effective poisoning prevention and targeted intervention programmers. The aim of the present study was twofold. Firstly, this study sought to evaluate the frequency of signs and symptoms of opium and opioid poisoning patients. The study also aimed to identify the risk factors of opioid-induced seizures among all hospitalized opioid poisoning cases. So, all consecutive referred to center involving seizures were forwarded to more investigations.
Materials and Methods
Patients and sample
This retrospective observational study was performed at Razi General Hospital over three years period (from 1 January 2005 to 31 December 2007) on 264 opioid overdose cases whether the intention was accidental or recreational. Center of Toxic Emergency in Razi General Hospital has the largest caseload of seriously admission of Acute Opiate Overdose (AOO) in the southwest of Iran, Khuzestan. This teaching hospital is the main referral center for poisoning in the metropolitan city of Ahvaz, and serves population of about 2 million. This ward receives cases from Ahvaz city and health centres within and outside Khuzestan. All cases have been registered due to acute opiate poisoning recorded in hospital covering the period of the study were listed and included. The patients diagnosed with any form of epilepsy and other types of seizures one year before admission were excluded.
Opiates included heroin, opium, tramadol, morphine, methadone and tamjizak. Tamjizak is sometimes used to treat and prevent withdrawal symptoms. This drug was made for the first time by a pharmaceutical company and is a combination of industrial morphine hydrochloride and other synthetic drugs. All subjects completely informed to take part in this study. The criteria for diagnosis of acute opiate overdose, was defined as altered level of consciousness, miotic pupils, decreased respiration and symptoms responding to naloxane. Altered level of consciousness was defined according to physiologic depression grading (Table 1) [10]. The primary outcome of the study was seizure attack and secondary outcomes were the demographic characteristics, history of drug use, poisoning agents, intent of poisoning, route of exposure, place of poisoning, admission status and hallmarks of overdose. All the information was obtained from hospital files and recorded in a standardized form.
Grade
State of awareness
I
Confused, drowsy, lethargic, indifferent and/or uncooperative; does not
lapse into sleep when undisturbed
II
Stuporous; may be disoriented to time, place, and person; will lapse
into sleep when not disturbed; or belligerent and uncooperative
III
Deep stupor; requires strong pain to evoke movement
IV
Exhibits decorticate or decerebrate posturing to a deep pain stimulus
V
Does not respond to any stimuli; flaccid
Table 1: The used methodical evaluation of altered level of consciousness in cases.
Statistical analysis
Characteristics of all cases and the signs were reported by relative frequency. Chi square test was used as an univarite analysis for revealing the significant associated factors with seizure of overdose patients. Binary logistic regression was performed as a multivariate analysis to evaluate the simultaneous impact of different risk factors.
Significant risk indicators (P<0.1) from univariate analysis were chosen as candidates to enter the multivariate analysis. Odds Ratio (OR) (and its 95% CI) was considered as the effective size of interest. In the multivarite step, p-values less than 0.05 were considered as significant. For more investigating the results of the binary logistic regression, path analysis was performed. This analysis is often useful to describe the direct and indirect dependencies between a set of variables. Data were analyzed using SPSS 16.0 and LISREL 8.0 Statistical Software.
Results
A total of 264 subjects met the inclusion criteria which included 224 (84.8%) men and 40 (15.2%) women. The most common age group was the 14-25 years old (165 cases, 65.2%) followed by 26- 40 years old (58 cases, 22%) years old. Most poisoning occurred unintentionally (94.3%, n=249). In the cases of intentional overdose, attempted suicide was the most common cause of poisoning (5.7%, n=15). Overall 184 (69.7%) of total patients had been poisoned with a single opiate-product and the most frequent agent in single opiate group was tramadol (n=120, 65.2%), followed by opium (n=58, 31.5%) and heroin (n=5, 2.7%). Tramadol was main drug used by patients poisoned with a multi opiate-product (n=64, 80%) (Table 2).
Variable
No. (%)
Sex
Male
224 (84.8%)
Female
40 (15.2%)
Marital status
Married
72 (27.3%)
Single
192 (72.7%)
Age groups
14-25
165(62.5%)
26-40
58 (22%)
> 41
41(15.5%)
Life area
Urban
225 (85.2%)
Rural
39 (14.8%)
Sample drugs
Tramadol
183(69.3%)
Opium
72 (27.3%)
Heroin
7 (2.7%)
Tamjizak
1(0.4%)
Metadon
1 (0.4%)
Complications
Decreased Level of Consciousness (LOC)
144(54.5%)
Nausea and vomiting
47 (17.8%)
Drowsiness
40 (15.2%)
Seizure
33 (12.5%)
Route of usage
Ingestion
234(88.6%)
Smoking
24(9.1%)
Injection
6 (2.3%)
Table 2: Characteristics of study patients (n=264).
Combination of opium and alcohol was seen in 10 (3.78%) cases. In order to investigate the agents that were used with opiates, data on patients consuming multiple agents was examined in detail. Benzodiazepines, antidepressants and alcohol were the most often involved substances. Most of the patients were referred to hospital within 6 hours (85.98%; n=237) and 30 (11.4%) were admitted to the Intensive Care Unit (ICU). The mean length of stay in hospital was 2.68 ± 5.06 days with a median of one day.
Out of total of 264 cases, 5 (1.9%) patients died following hospitalization and being placed in ICU. The mean age among patients who suffered from a fatal sign was 48.6 years old with a median of 17 years. The mortality was found lesser in heroin consumers. Only 2.8% of opium and 1.1% of tramadol cases died. Ingestion was the main route of overdose in 4 (80%) of fatal cases. Table 2 includes other characteristic of study patients.
Based on univariate analysis for cases characteristics (Table 3), age, marital status and taking tramadol were definitely related to the seizure (P < 0.1) but ethnicity wasn’t significant (P > 0.1) (Table 3). In the next step, significant variables in the univariate analysis were entered in the multivariate analysis. The results of multivariate analysis (Table 4) show taking tramadol was significant risk factor of seizure (P <0.05). Actually patients who used tramadol relative to others were more likely to experience seizure attack (OR: 10.4, 95%CI: 1.2-90.4, P=0.035). The risk of seizure in single and marital cases was almost same (OR: 0.98, 95%CI: 0.24-3.9, P=0.9).
Patient with seizure (%)
Patient without� seizure
Chi-Square test
P-value
male
30(90.9)
194(84%)
gender
female
3(9.1)
37(16%)
1.08
0.44
14-25
29(87.9)
136(58.9)
age
26-40
3(9.1)
55(23.8)
10.53
<0.001
4.37
0.037
Married
4(12.1)
68(29.4)
Referring
6-Jan
11(33.3)
54(23.4)
time
24-Jun
13(39.4)
105(45.5)
1.54
0.49
24-72
9(27.3)
72(27.3)
Suicide
1(3)
14(6.1)
0.5
0.7
Cause
Unintentional
32(97)
217(93.9)
Ingestion
31(93.9)
203(87.9)
Route of use
Smoking
1(3)
23(10)
1.74
0.42
Injection
1(3)
5(2.1)
Poised with single opiates
157(68)
27(81.8)
Poisoning
2.6
0.12
agent
Poised with multi opiates
74(32)
6(18.2)
Tramadol
32(97)
152(65.8)
Opium
0(0)
71(30.7)
opiates
Metadon
0(0)
1(0.4)
14.48
0.006
Heroin
1(3)
6(2.6)
Tamjizak
0(0)
1(0.4)
Table 3: Characteristics of the study participants and results of univariate analysis characteristic.
Also, multivariate results show that patients with 14-25 years old were likely to onset seizure (OR=2.3) but it was not statistically significant (p > 0.05). In order to investigate this result, more analysis performed, since it’s due to co linearity problem and it automatically removed from analysis. So the relationships between the variables were assessed using chi square test. The result of this analysis showed the strong association between tramadol intake and the age (chi- square = 107.93 p<0.001). Actually 91.5% (151 of 165) of younger cases use tramadol and 97% of onset seizure observed in the tramadol consumers. Age affects the seizure indirectly via its effects on taking tramadol (Figure 1).
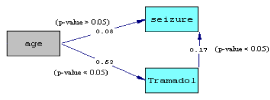
Figure 1: Diagram of path analysis showed the indirect age effect through the
significant path between age and tramadol.
Discussion
However the burden of death following acute opiate poisoning in this study was low but the seizure occurring following tramadol poisoning serves as an important call for public health concern, taking into account cases of the general population that is not captured in hospitals.
High-doses of tramadol induce seizures in animals and human [11,12] and some studied have shown seizures to occur even at therapeutically recommended doses [12]. A dose-dependent occurrence a seizure of tramadol has been reported in animals [13] and humans [14]. The present study showed that tramadol-induced seizures were significantly correlated to age. The seizure occurrence was different significantly and seizures may occur more often in different age groups (Figure 1 and Table 3). The age-dependent occurrence of a seizure of the present study is hallmark, remarkable and not confirmed previously. This relation is consistent with earlier not confirmed reports [15,16] and inconsistent with tagadossinejad et al study [14].
Serotonin syndrome is the most suspected causes of seizureinducing effect of tramadol [13]. The main signs of the tramadol toxicity are agitation, tachycardia, confusion and hypertension suggest a possible serotonin syndrome [11]. This association was highlighted in Mehrpour report [17]. In earlier reports, suggested that tramadol may induce seizures to a greater extent than do other opioid [6]. This hypothesis is still controversial [12,16]. We report the seizure occurrence among patients with an acute opiate overdose diagnosis is caused only by tramadol. Thus, the seizure-inducing effect of tramadol is most likely mediated through the serotonergic pathway.
The main cause of poisoning was tramadol followed by opium. Tramadol is a synthetic opioid analgesic used in treating moderate and severe pain. Tramadol has been marketed officially as a new generic prescription drug in Iran by the office of controlled drugs and substances during 1999-2007 in retail pharmacies. It has been abused, especially by the young population [19]. Increasing medical use of tramadol may have inadvertently contributed to the rate of increase in available poisoning endemic. During this period, tramadol overdose has become one of the most common causes of poisoning admissions to emergency departments in Iran [19-22]. It might because opiates like tramadol were more accessible and inexpensive than other agents.
These findings show increased risk of seizure (18% of cases) due to an appropriate sample size in patients with a tramadol poisoning diagnosis. Only a few studies have been published elsewhere. The seizure occurrence was reported in 1% of adult users in the range of 25-54 years and in alcoholic persons [16,18]. The alcoholism may have been involved in the age-dependent occurrence of seizure at the age below about 25 years. But information on alcohol consumption among our subjects was not available because these kinds of beverage are prohibited totally in Iran, thus was not reported by patients if even consumed.
Most of the patients were intoxicated unintentionally. Prevalence of opium poisoning was assessed 84.8% in men and 15.2% in women. The prevalence of opium use by men is similar as shown previously [23]. The prevalence of opium poisoning was previously assessed 91% in men and most of overdoses occurred unintentionally [24], which is similar to this study. The reason is males are more prone to drug dependency, abuse, and poisoning. The prevalence of opium use in urban (85.2%) was significantly higher than rural areas. This result may show that opium is not a common form of addiction in rural areas. The most cases were intoxicated due to opiate as a single agent and mostly by ingestion. In Thailand has shown that the most common symptoms in opium and opiate intoxication were decreased level of consciousness and vomiting which are similar to present study [25]. Benzodiazepines and antidepressants followed by ethanol and other opioids were the most commonly used with opiates agents in this study. This finding is consistent with previous reports [19,26-28]. Combined use of central nervous system depressant (such as alcohol and benzodiazepines) is well documented as a significant risk factor for opiate overdose [27-28]. This combination pattern of addiction may increase the risk of overdose and opium toxicity [29]. It is likely toxic effects of tramadol are exacerbated following consumption of CNS (central nerve system)-depressant drugs [21]. The rate of fatality was 5 death cases in 264 patients (1.9%). The fatality rate in Tehran was higher (47 deaths of 534 patients or 8.8%) and close to that of another study conducted in Switzerland (15 deaths among 190 cases or 7.9%) [30]. Most of the patients were referred to Razi hospital earlier than six hours. This suitable time of referral makes improving patient management with earlier contact and specialist care. It might be a reason for low mortality.
The most cases were reported with tramadol poisoning (183) followed by opium (72 case). Seizures and loss of consciousness are the most common adverse neurological signs among the 14-25 age group. This comparison was significant with other age groups (Table 4). All of the seizure cases were caused by tramadol poisoning. Occurrence of seizure has a peak in the 14-25 age group and was significant between the classified age groups. Reports show that the seizure may occur in individual or abusers consuming tramadol for reduction of pain or induction of complete euphoria, but it is more common when patients exceed the usual doses [31,32]. The frequency of seizure in the present study is remarkably comparative with presented studies [11,18,20,33]. Talaie et al., (2009) found an increased risk of seizure (46.2%) in patients less than 25 years old [20]. Among the identified patients, 61.3% were in 25-40 years old, so a low risk of seizure in patients in the age above 25 years was seen. This result is not consistent with other studies that showed increased risk of seizure in patients in the range above 25 years [18,21].
Characteristic
OR
95% CI
P-value
age
14-25
2.3
0.22-24.14
0.49
26-40
1.1
0.099-12.1
0.94
41-80
1a
----
----
Marital Status
Single
0.98
0.24-3.9
0.9
Married
1a
----
----
opiates
Tramadol
10.4
1.2-90.4
0.035
Other opiates
1a
----
----
a Reference category;
Table 4: Results of multivariate model for significant risk factors from univariate analysis.
There was no reported use of heroin and tramadol substances in the northern Iranian province [23]. It seems there is a significant difference in the pattern of substance use in the northern and the southern regions of Iran. The leading cause of poisoning among opium derivatives may show the risk increasing medical use of tramadol. This warning should be considered in its prescription to manage chronic, severe and non management pain.
In conclusion, the seizure was more frequent with tramadol than other opioid. Tramadol-induced seizure is age-dependent. Ages less than 25 are playing a significant role. These findings may indirectly implicate on non- related opioid mechanisms causing seizures. This is a suggestion that needs answered by following studies.
Recommendation
Establishing and implementing of inflexible rules to ban physician prescription or over the counter selling of tramadol is an obligation. The most common route of overdose was ingestion, highlighting ingestion as a potential cause of fatal overdose as opposed to the popular belief that only smoking and injection of opium is harmful [34].
Financial disclosure: The authors have not any financial support in this submission.
Acknowledgement
The research was extracted from a G.P. thesis that was performed at Ahvaz Jundishapur University of Medical Sciences with the collaboration of Razi hospital (registration number A/219).
References
- Nikfar S, Kebriaeezadeh A, Majdzadeh R. Monitoring of National Drug Policy (NDP) and its standardized indicators: conformity to decisions of the national drug selecting committee in Iran. BMC Int Health Hum Rights. 2005; 5: 5.
- Wu, McKown LA, Liao S. Metabolism of the analgesic drug ULTRAM (tramadol hydrochloride) in humans: API-MS and MS/MS characterization of metabolites. Xenobiotica. 2002; 5: 411-425.
- Sansone RA, Sansone LA. Tramadol: seizures, serotonin syndrome, and coadministered antidepressants. Psychiatry (Edgmont). 2009; 6: 17-21.
- Potschka H, Friderichs E, Loscher W. Anticonvulsant and proconvulsant effects of tramadol, its enantiomers and its M1 metabolite in the rat kindling model of epilepsy. British J Pharma. 2000; 131: 203-212.
- Sen H, Ozkan S, Dagli. Epileptic seizure during patient-controlled analgesia with tramadol. Eur J Anesthesiology. 2009; 35: 361-364.
- Kahn LH, Alderfer RJ, Graham DJ. Seizures reported with tramadol. J Am Med Association. 1997; 278: 1661.
- Thundiyil JG, Kearney TE, Olson KR. Evolving epidemiology of drug induced seizures reported to a poison control center system. J Med Tox. 2007; 3: 15-19.
- Thundiyil JG, Rowley F, Papa L. Risk factors for complications of druginduced seizures. J Med Tox. 2011; 7: 16-23.
- DeLorenzo RJ, Sun DA, Deshpande LS. Erratum to “Cellular mechanisms underlying acquired epilepsy: the calcium hypothesis of the induction and maintenance of epilepsy”. Pharma Therapeutics. 2006; 111: 288-325.
- Walker HK, Hall WD, Hurst JW. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd ed. Boston: Butterworths. 1990.
- Spiller HA, Gorman SE, Villalobos D. Prospective multicenter evacuation of tramadol exposure. J Tox Clin Tox. 1997; 35: 361-364.
- Marquardt KA, Alsop JA, Albertson TE. Tramadol exposures reported to statewide poison control system. Ann Pharmacother. 2005; 39:1039-1044.
- Manocha A, Sharma KK, Mediratta PK. Tramadol, a centrally acting opioid: anticonvulsant effect against maximal electroshock seizure in mice. Ind J Physiology Pharma. 1998; 42: 407-411.
- Taghaddosinejad F, Mehrpour O, Afshari R, Seghatoleslami A, Abdollahi M, Dart RC. Factors related to seizure in tramadol poisoning and its blood concentration. J Med Tox. 2011; 7: 183-188.
- Jovanovic-Cupic V, Martinovic Z, Nesic N. Seizures associated with intoxication and abuse of tramadol. Clin Tox (phila). 2006; 44: 143-146.
- Gardner JS, Blough D, Drinkard CR. Tramadol and seizure: a surveillance study in a managed care population. Pharmacotherapy. 2000; 20: 1423-1431.
- Mehrpour M. Intravenous tramadol-induced seizure: two case report. Iran J Pharmaco Therapy. 2005; 4: 146-147.
- Gasse C, Derby L, Vasilakis-Scaramozza C, Jick H. Incidence of first-time idiopathic seizures in users of tramadol. Pharmacotherapy. 2000; 20: 629- 634.
- Shadnia S, Soltaninejad K, Heydari K, Sasanian G, Abdollahi M. Tramadol intoxication: a review of 114 cases. Hum Exp Tox. 2008; 27: 201-205.
- Talaie H, Panahandeh R, Fayaznouri M, Asadi Z, Abdollahi M. Doseindependent occurrence of seizure with tramadol. J Med Tox. 2009; 5: 63-67.
- Talaie H, Panahandeh R, Fayaznouri M. Seizures associated with tramadol use/abused. J Clin Tox. 2008; 46: 628.
- Afshari R, Tashakori A, Shakiba AH. Tramadol overdose induced CPK rise, haemodynamic and electrocardiographic changes and seizure. Clin Tox. 2008; 46: 369.
- Meysamie A, Sedaghat M, Mahmoodi M, Ghodsi SM, Eftekhar B. Opium use in a rural area of the Islamic Republic of Iran. East Mediterranean Health J. 2009; 15: 425-431.
- Karbakhsh M, Salehian, Zandi N. Acute opiate overdose in Tehran: The forgotten role of opium. Addictive Behaviors. 2007; 32: 1835-1842.
- Jittiwutikarn J, Ali R, White JM, Bochner F, Somogyi AA, Foster DJ. Comparison of tincture of opium and methadone to control opioid withdrawal in a Thai treatment centre. British J Clinical Pharma. 2004; 58: 536-541.
- Clarot F, Goulle JP, Vaz E, Proust B. Fatal overdose of tramadol: Is Benzodiazepine a risk factor of lethality? Forensic Science Int. 2003; 134: 57-61.
- Oliver P, Keen J. Concomitant drugs of misuse and drug using behaviours associated with fatal opiate-related poisoning in Sheffield, UK, 1997–2000. Addiction. 2003; 98: 191-197.
- Warner-Smith M, Darke S, Lynskey M, Hall W. Heroin overdose: Causes and consequences. Addiction. 2001; 96: 1113-1125.
- Darke S, Ross J. The use of antidepressants among injecting drug users in Sydney, Australia. Addiction 2000; 95: 407-417.
- Cook S, Moeschler O, Michaud K, Yersin B. Acute opiate overdose: Characteristics of 190 consecutive cases. Addiction. 1998; 93:1559-1565.
- Grond S, Sablotzki A. Clinical pharmacology of Tramadol. Clin Pharmacokinetic. 2004; 43: 879-923.
- McDiarmid T, Mackler L, Schneidar DM. Clinical inquiries. What is the addiction risk associated with tramadol? J Family Practice. 2005; 54: 72-73.
- Gholami K, Shalviri G, Zarbakhsh A, Daryabari N, Yousefian S. New guideline for tramadol usage following adverse drug reaction reported to the Iranian pharmacovigilance Center. Pharmacoepidemiology Drug Safety. 2007; 16: 229-237.
- Kalant H. Opium revisited: A brief review of its nature, composition, nonmedical use and relative risks. Addiction. 1997; 92: 267-277.