
Special Article - Oral Administration of Leptin
Austin J Endocrinol Diabetes. 2016; 3(3): 1050.
Control of Food Intake by Oral Administration of Leptin to Dogs
Bendayan M* and Cammisotto PG
Department of Pathology and Cell Biology, University of Montreal, Canada
*Corresponding author: Bendayan M, Department of Pathology and Cell Biology, University of Montreal, C.P. 6128 Succ. Centre Ville 2900 Edouard Montpetit Montreal, Quebec, H3C 3J7, Canada
Received: July 08, 2016; Accepted: September 09, 2016; Published: September 14, 2016
Abstract
Upon demonstrating that long term oral administration of leptin to mice led to reducing food intake and control of body weight, we decided in the present study to evaluate the effect of short term oral administration of leptin to dogs. An optimal vehicule was designed in order to administer orally leptin to dogs. One dose consisted in a number of components and 1mg of leptin encapsulated. The pill was easily swallowed by the dog. Food was presented to the animals one hour after and food intake was measured. The experiments were performed either early in the morning or early in the afternoon. Results demonstrated systematically that upon oral administration of leptin the animals reduced their food intake. Reduction in the amount of food ingested varied from 15% to 55% depending mainly on the time of the day. Oral administration of leptin was more efficient in the mornings that in the afternoons. The success of the treatment was related to the efficiency of leptin absorption. A high correlation was found between circulating levels of exogenous leptin and amounts of food intake. Based on these results and on those obtained previously on rodents we propose that oral administration of leptin represents an excellent approach for the control of food intake.
Keywords: Obesity; Leptin; Oral administration; Food intake; Dogs
Introduction
In previous studies we have established that gastric chief cells secrete leptin [1]. Release of leptin by the gastric mucosa is a regulated exocrine secretion that occurs together with several gastric enzymes such as lipase and pepsinogen [1]. Gastric leptin secretion is triggered by and regulates food intake [2].
Being secreted in an exocrine fashion, leptin, a small peptide of 146 amino acids with a molecular weight of 16kDa [3,4], finds itself in the gastric juice and as such exposed to very harsh conditions. In order for this peptide to survive the gastric environment, it is complexed to a soluble isoform of its own receptor [1,2,5,6]. Indeed, the chief cells synthesise an isoform of the leptin receptor which becomes soluble along the RER-Golgi-granules secretory pathway [5]. This soluble receptor isoform gets associated to the leptin peptide and the complex leptin-leptin receptor packaged in the secretory granules is released [2,5]. The leptin-leptin receptor complex appears to be quite resistant to the acidic conditions of the gastric juice [5]. From the gastric cavity, the leptin-leptin receptor complex is vehiculated towards the duodenum where leptin interacts with leptin-transmembrane receptors located on the apical membrane of the intestinal cells. Upon binding to its membrane receptor, leptin is internalized by the enterocytes and through an intricate trans-cellular pathway, crosses the intestinal cells to be released on baso-lateral side and reach through blood circulation its target hypothalamic cells [2,6].
The fact that leptin is normally present in the gastric juice, elicited the proposal that leptin could be administered orally. Once in the gastric cavity, exogenous orally administered leptin could take its physiological path to get to the duodenal lumen, interact with its receptor present at the luminal epithelial cell membrane, get internalized, transported and released into the basal connective tissue to reach blood circulation and thus its target hypothalamic cells.
A first series of experiments were performed in order to assess this hypothesis. Free leptin dissolved in an appropriate buffer was force-fed to mice. We have demonstrated that from the gastric cavity, the exogenous leptin is indeed able to reach blood circulation very efficiently in a matter of minutes [7]. It then interacts with its own receptors located in the hypothalamic cells. We have demonstrated that oral leptin is able to control food intake and to regulate body weight gain in mice [7]. Several experiments were performed with different strains of mice. Normal mice forced-fed with oral leptin, exhibit a very high efficiency in the transport of the exogenous leptin from the gastric cavity to the duodenal lumen and from there to cross the duodenal mucosa entering the blood [7]. In normal mice, the transported leptin reaches a maximum within the first five minutes after oral administration. We found that the oral leptin is able to decrease amounts of food intake by the animal and consequently influences gain in body weight [8]. Oral administration of leptin to ob/ob mice was also efficient [8]. The ob/ob mice characterized by a genetic deficiency of leptin [9] have no satiety feelings and when exposed to food ad libitum, the animals eat non-stop to become rapidly obese [10]. Oral administration of leptin was able to control the amounts of food intake and consequently to restrain the animal gain of weight. It even allowed for body weight decrease when treatment was carried out for a long period of time [8]. Compared to the normal mouse, the ob/ob one handles exogenous leptin less efficiently [8]. Oral leptin reached its highest blood concentration only 60 minutes after administration, compared to the five minutes registered in the normal mouse [8]. This must be due to the fact that since ob/ob mice have no leptin, the trans-cellular transport system of leptin across the epithelial cells may not be as active as in normal mice. Further to this, we also tested the efficiency of oral leptin in db/db mice to find out that in spite of oral leptin reaching circulation, no major leptin action was observed [8]. This is due to the fact that the membrane bound leptin receptor in hypothalamic cells is deficient [11,12]. Thus in spite of large amounts of leptin in circulation, the hormone is unable to trigger the hypothalamic response and to control food intake.
In view of the efficiency demonstrated by oral leptin in reaching the hypothalamic cells and triggering a physiological response in mice [8], we evaluated in the present study the ability for oral leptin to control food intake in large animals. Experiments were performed in dogs. For this we designed a tablet containing the different elements that allow for oral leptin to reach blood circulation. Once the nature of the tablet designed and its composition determined, we provided young dogs with one tablet prior feeding. We determined the time required for the oral leptin to reach blood circulation and further evaluated its efficiency to control amounts of food intake.
Results demonstrate that a leptin pill taken by the dogs an hour prior feeding is able to reduce significantly the amount of food intake. The efficiency of the treatment appears to be proportional to the amount of exogenous leptin reaching circulation. These results together with previous ones obtained with rodents [8], demonstrate that leptin is a powerful factor that regulates food intake and that once given orally, it does play its roles very efficiently. Oral leptin administration appears as a potent approach for the management of certain cases of obesity.
Material and Methods and Results
Animals
Four beagles (2 males and 2 females) were acquired by and housed at the Animal Facility of the Sacré-Coeur University Hospital in Montreal. Animals were about one year old and weighed between 8 and 10kg. Experiments were carried out following the guidelines of the Canadian Committee of Animal Care and those of the Research Center of the Sacré-Coeur Hospital. Animals were kept in individual cages and acclimated for one week prior starting the experiments. They were trained to get their food twice a day at fixed hours (9AM and 3:30PM). 250g of standard dog food was provided for 30 minutes at each feeding time. Food intake was measured mornings and evenings. Body weight was registered at 8AM each day. Work was carried out following the guidelines of the Canadian Committee for Animal care and those of the Animal facility of the Sacré-Coeur Hospital.
The leptin capsule
Several recipes and combinations of components were tested. Composition and concentration of factors composing the capsule were tested in order to achieve a rather optimal situation in which oral leptin was able to trigger significant results. After trying several protocols and strategies, we came up with the following protocol. 75 min prior feeding, one pill of the following composition was administered to each dog: 1g of sodium bicarbonate (Sigma St- Louis, Mo, USA) and 100mg of trypsin inhibitor (Sigma type 2ST91228). This pill was washed down with 20 ml of H2O. 15 min later (60 min prior feeding) one pill of the following composition was administered to each dog: 0.8g sodium bicarbonate (Sigma); 0.5g sodium deoxycholate (Sigma); 100mg trypsin inhibitor (Sigma); 50mg Aprotinin (Cedarlane CLPRO-285-3, Burlington, Ontario) and 1mg of human leptin (about 100μg/Kg) (R&D System OB-398LP, Minneapolis, MN, USA). This pill was also washed down with 20ml of water. All components were inserted into hard gelatin capsules (# 00, 3901-2001; Galenova, Saint-Hyacinthe, Qc, Canada).
Internalization of oral leptin
Animals receiving the leptin capsule containing the above mentioned composition, were bled (0.3ml) from the front leg artery at time 0, 15min, 30min, 60min and then at 2 and 3 hours after the oral leptin administration. The decision to administer human leptin was made in order to be able to follow the outcome of the exogenous leptin. Indeed, the Elisa (R & D Systems) test carried out to measure exogenous leptin is specific for human leptin and does not cross-react with dog leptin. Endogenous dog leptin did not interfere with our measurements. Results reported on Figure 1 show that upon oral administration of human leptin, the peptide appears in the bloodstream of the dogs 15min after administration. Blood concentrations of human leptin continue to rise, reaching a peak at 60 minutes (Figure 1). This was followed by a return towards basal values after 3 hours (Figure 1).
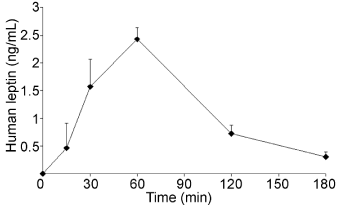
Figure 1: Levels of human leptin in the bloodstream of dogs receiving 1mg
of human leptin. Blood was sampled at regular time points and assessed
with an Elisa test specific for human leptin. Human leptin blood levels reach
a peak 60 minutes after its oral administration and returns to basal values
after 3 hours.
Treatment of the dogs with oral leptin
At time of the experiments, the animals were given one leptin capsule 60 minutes prior feeding. The animals were trained to swallow the capsule without difficulties. Food was presented to the animals 60 minutes later and the animals were allowed to eat for a period of 30 minutes. Food was then removed and food intake measured. Experiments were performed either in the morning or in the afternoon.
Upon receiving the leptin capsule, food intake by the dogs was significantly reduced. No major variations were detected in relation with the sex of the animals, male as well as the female dogs being sensitive to leptin action. On the other hand, the reduction in food intake differed significantly between experiments performed in the morning or in the afternoon. Indeed the animals appeared to be more sensitive to the treatment in the morning. Figures 2,3 and 4 illustrate the differences in food intake by individual dogs upon receiving 1mg of human leptin orally. The average decrease in food intake in the mornings varied from -26% to -54% (Figure 2). Changes in the afternoon ranged only between -11% and -19% (Figure 3). These values are averages of 6 to 11 experiments. Male dogs responded to the oral treatment by decreasing their food intake by -46% and -52% while female dogs decrease their food intake by -26% and -54% (Figures 2,3&4). The experiments also demonstrate that upon discontinuing the leptin treatment the eating behavior returned immediately (at the time of the next feeding) back to normal with a 100% recovery of their food intake (Figures 2,3&4).
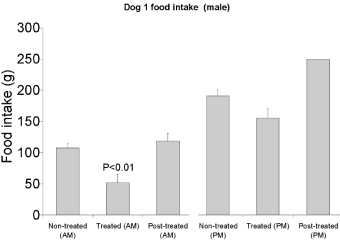
Figure 2: Food intake by male dog #1 receiving 1mg of human leptin either
in the morning or in the afternoon an hour prior meals. The effect is more
significant in mornings than in afternoons.
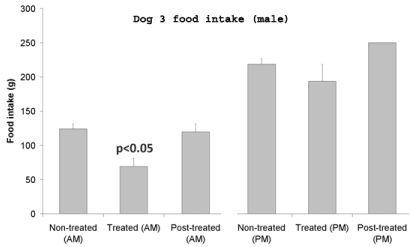
Figure 3: Food intake by male dog #3 upon receiving 1mg of human leptin
either in the morning or in the afternoon, an hour prior meals. The effect is
more significant in mornings than in afternoons.
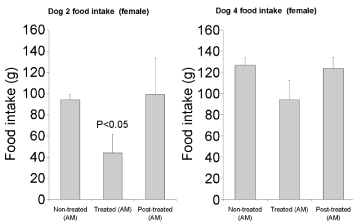
Figure 4: Food intake by female dogs #2 & #4 receiving 1mg of human leptin
one hour prior meals.
Dog body weights were daily measured and found to remain stable. This may be explained by the fact that the leptin administration was usually limited to once a day and not consequently. Another possible explanation was to find out that absorption of leptin was not always consistent, one day better that others and certainly more efficient in the morning than in the afternoon. To verify this point, we carried out a correlation assessment between circulating levels of oral human leptin and the percentage of food intake in the same experiment. Figure 5 illustrates such evaluation and demonstrates that the amount of food ingested is directly related to the circulating levels of oral leptin. The correlation factor is highly significant R2= 0.75 (Figure 5).
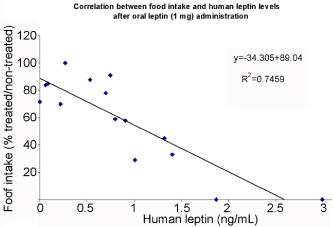
Figure 5: Correlation between food intake and circulating levels of human
leptin. Food intake by the animals is directly proportional to the levels of
human leptin (n=15).
Discussion
We have previously demonstrated with normal and ob/ob mice, that oral administration of leptin is highly efficient in controlling food intake and once given on a long term basis, in reducing the animal body weight [8]. In view of this efficient action of oral leptin in mice, we herein evaluated if larger animals such as dogs, would also respond to oral leptin by reducing their food intake. The present study demonstrates that leptin given orally is efficient in controlling food intake in large animals. Provided that the oral leptin is protected from early degradation in the stomach, it does reach the intestinal lumen to be transported through the intestinal mucosa and to reach through blood circulation its hypothalamic target cells triggering a response. We designed an oral formulation which allows for leptin reaching blood circulation in dogs. Upon giving leptin orally in combination with certain reagents, leptin survives the harsh conditions of the gastric juice; it is vehiculated to the duodenum where, interacting with its transmembrane receptor it is internalized by the epithelial cells and transferred to the blood circulation. Once reaching its target hypothalamic cells, oral leptin suppresses food intake in dogs.
The oral formulation established for dogs is similar to the one used for rodents [8]. However, aprotinin used for rodents, was replaced by trypsin inhibitor which exhibits a better peptide protection from degradation by the gastric juice in dogs [13]. We decided to administer human leptin since physiologically it is very efficient in dogs and presents the advantage of being evaluated by a specific Elisa test without interfering by endogenous dog leptin during measurement [14].
Oral leptin given to dogs was found to reach circulation shortly after administration and reached a maximum within one hour, decreasing gradually thereafter and reaching very low values at three hours. Thus, we designed the experiment accordingly to take advantage of the maximum effect. The leptin capsule was given to the dogs early in the morning or afternoon and food was presented to the animals an hour later. Once oral leptin in circulation and reaching its target hypothalamic cells, it controlled food intake. Thus food intake by dogs receiving a leptin pill was significantly reduced. Amounts of food intake were drastically different from those ingested a day before or a day after the experiment. Experiments were repeated several times with the same animals on different days and results remained consistent. However, we found out that efficiency of the leptin administration differed between morning and afternoon. In fact we found a very high correlation between levels of circulating exogenous leptin and decreases in amounts of food intake. This indicates that the effectiveness of the experiments relates to the efficiency of leptin to cross the intestinal wall rather than leptin action itself. Levels of circulating exogenous leptin were higher when the oral leptin was given in the morning than in the afternoon. One possible explanation may lay on the efficiency of leptin protection in the gastric cavity. Morning leptin administration may be confronted to smaller amounts of gastric juice while in the early afternoon gastric conditions may be different and probably harsher [15].
Amounts of food intake were significantly reduced upon oral administration of leptin. However, body weight of the animals did not demonstrate any significant changes. This result differs somehow from those obtained with mice [8]. Upon long term administration of oral leptin to normal mice the animals display losses of body weight. This loss was even more significant when the experiments were performed on ob/ob mice [8]. Experimental protocol applied to dogs differs somehow from that on mice. Administration of leptin to dogs was carried out once a day and not consistently every day, days after days. Another issue is the fact that the dogs used for the experiment, were young lean animals not having reached their stable adult weight and not having significant fat deposits, situation that differs totally from the experiments carried out on mice and on ob/ob mice in particular which were adult and obese [8].
In summary the study confirms on large animals previous results obtained on rodents. Oral administered leptin survives the harsh conditions of the gastric cavity, is vehiculated by the gastric juice to the duodenal lumen, transferred through the epithelial cells to circulation and acts on target hypothalamic cells to reduce food intake. Oral administration of leptin appears powerful for the management of food intake.
Acknowledgment
The authors would like to express their gratitude to Daniel Hofmann and Doctor Ilan Hofmann from I-Med Pharma Inc. for their support and enthusiasm. Doctor Grégoire Leclair & Mihaela Friciu from the Faculty of Pharmacy, University of Montreal, for their help in designing the pill and Caroline Bouchard from the Sacré- Coeur Hospital for her help in running the dog facility. This work was supported by a grant from I-Med Pharma Inc.
References
- Cammisotto PG, Renaud C, Gingras D, Delvin E, Levy E, Bendayan M. Endocrine and exocrine secretion of leptin by the gastric mucosa. J Histochem Cytochem. 2005; 53: 851-860.
- Cammisotto PG, Levy E, Bukowiecki LJ, Bendayan M. Cross-talk between adipose and gastric leptins for the control of food intake and energy metabolism. Prog Histochem Cytochem. 2010; 45: 143-200.
- Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature. 1994; 372: 425-432.
- Cohen SL, Halaas JL, Friedman JM, Chait BT, Benet L, Chang D, et al. Human leptin characterization. Nature. 1996; 382; 589.
- Cammisotto PG, Gingras D, Renaud C, Levy E, Bendayan M. Secretion of soluble leptin receptors by exocrine and endocrine cells of the gastric mucosa. Am. J. Physiol. Gastrointest. Liver Physiol. 2006; 290: 242-249.
- Cammisotto PG, Gingras D, Bendayan M. Transcytosis of gastric leptin through the rat duodenal mucosa. Am J Physiol Gastrointest Liver Physiol. 2007; 293: 773-779.
- Bendayan M, Cammisotto PG. Leptin secretion by adipose tissue and gastric mucosa for the control of food intake: A review. Endocrinol. Diabetes In press.
- Bendayan M, Cammisotto PG. Control of food intake and body weight through oral administration of leptin. Endocrinol. Diabetes In press.
- Friedman JM, Leibel RL, Siegel DS, Walsh J, Bahary N Molecular mapping of the mouse ob mutation. Genomics. 1991; 11: 1054-1062.
- Pelleymounter MA, Cullen MJ, Baker MB, Hecht R, Winters D, Boone T, et al. Effects of the obese gene product on body weight regulation in ob/ob mice. Science. 1995; 269: 540-543.
- Tartaglia LA, Dembski M, Weng X, Deng N, Culpepper J, Devos R, et al. Identification and expression cloning of a leptin receptor, OB-R. Cell. 1995; 83: 1263-1271.
- Cammisotto PG, Bukowiecki LJ. Mechanisms of leptin secretion from white adipocytes. Am J Physiol Cell Physiol. 2002; 283: 244-250.
- Ziv E, Kidron M, Raz I, Krausz M, Blatt Y, Rotman A, et al. Oral administration of insulin in solid form to non-diabetic and diabetic dogs. J Pharm Sci. 1994; 83: 792-794.
- LeBel C, Bourdeau A, Lau D, Hunt P. Biologic response to peripheral and central administration of recombinant human leptin to dogs. Obes Res. 1999; 7: 577-585.
- Moore JG, Englertjun E. Circadian rhythm of gastric acid secretion in man. Nature. 1970; 226: 1261-1262.