
Special Article - Hearing Loss
Gerontol Geriatr Res. 2020; 6(1): 1040.
Overview of Hearing Loss in the Elderly
Molinier CE*, Gallois Y, Marx M and Fraysse B
Department of Otoneurology-Head and Neck Surgery, Pierre-Paul Riquet University Hospital Center of Toulouse, France
*Corresponding author: Charles-Edouard Molinier, Department of Otoneurology-Head and Neck Surgery, Pierre-Paul Riquet University Hospital Center, France
Received: December 11, 2019; Accepted: January 03, 2020; Published: January 10, 2020
Introduction
Deafness is a major public health issue worldwide. According to the World Health Organization (WHO), of all diseases it is the 4th leading cause of morbidity. The WHO defines hearing loss as a loss of more than 25dB in the ear with best hearing. Deafness is considered disabling when hearing loss exceeds 40dB in adults and 30dB in children in the best ear. In their March 2019 report, the WHO estimated that more than 466 million people are affected by disabling hearing loss, which is 6.6% of the world population. The vast majority (93%) is adults and the people most at risk are the elderly, since 1/3 of those over 65 years of age are reported to suffer from deafness. The number of people with disabling hearing loss is expected to rise in the future to 630 million in 2030 and over 900 million in 2050 [1].
In addition, the link between deafness and cognitive decline is now corroborated by robust data and is the subject of much research [2-4]. The economic impact of deafness is estimated to be $750 billion per year [5,6].
Adult Hearing Loss
Overview and epidemiology
Strictly speaking, presbycusis or Age-Related Hearing Loss (ARHL) is not a disease but rather a normal aging process involving all the neurosensory structures of the hearing system.
The pathophysiology is complex because it involves several structures and depends on several factors. Damage can be localized at the cochlear, neural and central level. The intrinsic factors involved are mitochondrial alterations, genetic and cellular disorders, micro-vascularization damage due to cardiovascular risk factors (hypertension, diabetes, dyslipidemia), and other systemic diseases. Extrinsic factors are mainly related to exposure to noise, ototoxic drugs and diet.
ARHL is characterized by a decrease in auditory perception and speech comprehension, especially in noisy environments. Despite a normal hearing threshold, some subjects have difficulty understanding speech. This is referred to as a Central Auditory Processing Disorder (CAPD), which is a deficiency in central auditory pathways such as neural transmission, and an extraction deficit in auditory information processing.
The genetics of presbycusis is still poorly defined due to the polygenicity [7]. At present, a genetic examination is not systematically conducted in the context of classical presbycusis.
Screening for hearing loss
There is no universal screening system for adults as hearing loss is currently defined by general practitioners, ENT specialists or hearing aid acousticians on request by the patient or family.
WHO-sponsored awareness days such as World Hearing Day, held on March 3rd each year, provide free screening for patients at participating centers in many countries.
The gold standard in the diagnosis of presbycusis is still tonal and vocal audiometry. Therefore, presbycusis appears to be bilateral symmetrical sensorineural hearing loss that is predominant in high frequencies on tonal audiometry. Speech audiometry identifies difficulties in speech discrimination with an increase in the intelligibility threshold, which corresponds with the dB threshold required to recognize 50% of the words presented(Figure 1).
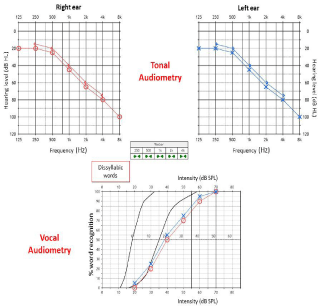
Figure 1: Tonal et vocal audiometries of Age-Related Hearing Loss.
Interviewers can ask simple questions that rule out or point to presbycusis:
- Do you have problems understanding in noisy environments such as restaurants, family meals, etc.?
- Do you understand male voices better than female voices?
- Do people around you complain about your tendency to increase the volume of the TV or radio?
- Have you become intolerant to certain loud sounds?
Many questionnaires related to presbycusis are also available. The most consensual is the Hearing Handicap Inventory for the Elderly (HHIE) and the simplified version which is more suitable for screening, the Hearing Handicap Inventory for the Elderly - Screening Version (HHIE-S) [8,9] (Table 1).
TABLECREATED
Table 1: Hearing Handicap Inventory for Elderly - Screening Version (HHIE-s).
Finally, smartphone applications have been developed to screen adult hearing loss. HearZao (De Sousa et al., 2018) or HearWHOo (De Wet et al., 2019) applications consist of repeating a series of 3-digit numbers presented on the headset or earphones in antiphasic conditions. This application can be used to test bilateral hearing in noise in less than 3 minutes and achieves a sensitivity and specificity of more than 80% for the diagnosis of a hearing impairment and the severity [10].
Current limitations that are being evaluated are related to digital access, particularly for older populations or those living in rural areas.
Risk factors for deafness
Risk factors for deafness should be systematically explored during an interview:
- Family history of early deafness
- Acute or repeated noise trauma (working in noise without protection)
- Otological pathologies: chronic otitis, otospongiosis, sudden deafness and Meniere’s disease
- Ototoxic drugs (aminoglycosides, chemotherapies, highdose aspirin)
- Severe infections (meningitis, severe sepsis)
- Autoimmune or systemic diseases
- Cardiovascular predisposition (diabetes, high blood pressure, dyslipidemia, obesity, etc.).
The impact of hearing loss
Classically, 3 “social” stages are described in presbycusis:
- The “subclinical” stage during which only high frequencies are affected has little clinical impact. At this stage the diagnosis is based on the audiometry.
- The “social impact” stage where conversational level frequencies begin to be affected. The first symptoms are often difficulty understanding speech in noise. The patient then gradually adapts their lifestyle by limiting certain social situations.
- The “advanced” stage where hearing loss is evident even in silence. The subject then begins to reduce their social interactions and gradually withdraws.
The impact of deafness on young and active populations is also significant.
Among children, there is a strong association between hearing loss and academic difficulties. Similarly, among adults, poorly cared for hearing loss is a major obstacle to hiring, especially in service or communication sectors which are experiencing rapid growth. As of the age of 60, early retirement motivated by hearing difficulties occurs more often than one might think.
Apart from this academic or professional “social” effect, a link has recently been identified between hearing loss and cognitive decline. A study published in Le Lancet in 2017 (3) identifies modifiable and non-modifiable risk factors for developing dementia. Hearing loss appears to be the most significant modifiable risk factor due to the high prevalence and strong association with cognitive decline. The mechanisms are still unknown and although angiopathies could be a confounding factor since the micro-vascularization of the inner ear and central nervous system are very similar, it now seems obvious that hearing loss accelerates cognitive decline.
Finally, pure central hearing loss is thought to be a prodrome of Alzheimer’s disease. The relative risk of developing Alzheimer’s disease in affected patients is estimated to be 4.2 [2].
This is a rare type of hearing loss. However, it is suspected in discordances revealed by audiometry, with preservation of tonal thresholds while spoken word recognition, i.e. speech comprehension plummets. A battery of specific behavioral and electrophysiological tests (GAP test, Tone Decay test, Auditory-Evoked Potential, etc.) carried out by a specialist confirms central hearing loss.
In a 25-year longitudinal study, Amieva et al. found a decrease in the MMSE score, an increase in cognitive decline and an increase in the risk of depression in elderly patients who reported hearing loss [11].
Under all circumstances, early use of a hearing aid in patients with presbycusis remains an important means of preventing dementia in the elderly.
The economic impact of presbycusis
Reed et al. estimated the overall additional cost of care in 10 years for a subject with untreated hearing loss to be $22,434 [5]. Unmanaged hearing loss could result in a 46% increase in the cost of care unrelated to the management of hearing loss itself. For example, there could be a 47% increase in hospitalizations or a 44% increase in readmissions within 30 days of hospitalization. These additional costs are not found in patients who wear a hearing aid [5].
The Geriatrician: An Essential Actor in the Management of Deafness
A screening role
As noted above, due to the fact that there is no systematic screening for deafness in the adult population, often the geriatrician is the one in a position to identify the signs of hearing loss, even if this is not the initial reason for consultation.
With simple questions or an adapted questionnaire the geriatrician can identify hearing loss, assess the severity, and most of all, the impact.
• Finally, in case of doubt concerning presbycusis with associated cognitive decline, the Codex test [12]can be performed in 3 minutes:Presentation of 3 words to the patient
• The clock drawing test
• Recall of the 3 words
• If the person fails the previous tests: 5 spatial orientation questions
A preventive role
Geriatricians should provide patients with information on the risk factors for hearing loss and how to protect against it. Protection against sound trauma, knowledge of ototoxic drugs and close monitoring of patients at risk are all ways to prevent early hearing loss in adults. Finally, controlling cardiovascular risk factors is also crucial to limit early sensorineural degeneration.
A diagnostic role
With an otoscope and a tuning fork, the geriatrician can make an excellent assessment. Recently, recommendations have been made in Great Britain by the National Institute for Health and Care Excellence [13]and in the USA by the American Family Physician [14].
An abnormal otoscopy alone can indicate hearing loss. In most instances it is related to earwax plugs (see Box 1), tympanic membrane perforations, seromucous otitis and chronic otitis media with cholesteatoma (Figure 2).
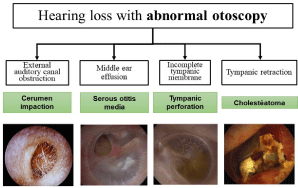
Figure 2: Principal causes of hearing loss with abnormal otoscopy.

Box 1: Earwax plugs.
When the otoscopy is normal, a Rinne test (tuning fork placed on the mastoid bone and then the still vibrating fork is presented in front of the outer ear) accompanied by a Weber test (tuning fork placed on the vertex) makes it possible to qualify hearing loss. Unilateral or bilateral damage then points to different pathologies (Figure 3).
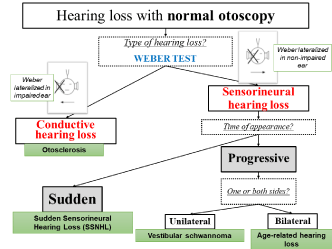
Figure 3: Principal causes of hearing loss with normal otoscopy.
There are 3 types of hearing loss that are urgent or worrying and require specialized ENT advice as soon as possible:
- Sudden unilateral sensorineural hearing loss
Oral corticosteroid treatment can be initiated by the referring physician and an ENT consultation within the next few days is recommended so that an audiometry can be performed to quantify the loss and to plan complementary treatments (hemodilution, hyperbaric oxygen therapy, transtympanic corticosteroid injections).
- Hearing loss with persistent otitis externa in diabetic or immunocompromised patients
Otitis externa is severe and resistant to standard treatments and can develop into otitis externa maligna, a real osteitis of the base of the skull, which is an extremely serious condition. Emergency hospitalization in the ENT department for appropriate localized care, cranial imaging and long-term IV antibiotic therapy are essential. Bacterial culture of the external auditory canal can be collected early by the general practitioner before any treatment because identification of the germ in question (often Pseudomonas Aeruginosa) and the antibiotic susceptibility test are essential for treatment.
- Unilateral sensorineural hearing loss, possibly associated with vertigo, facial paralysis or unilateral tinnitus
It should raise suspicion of a retrocochlear pathology, among which acoustic neuroma is at the top of the list. A contrast MRI and an ENT consultation within the next few weeks are necessary(Box 1).
An additional assessment could be suggested by the geriatrician, in particular imaging, before referral to a specialist in the following situations:
- CT scan without contrast of the petrous part of temporal bone if a middle ear pathology is suspected (chronic otitis media, conductive hearing loss with a normal tympanic membrane leading to a suspicion of otosclerosis)
- MRI with contrast focused on the cerebellopontine angles if retrocochlear pathology is suspected (unilateral sensorineural hearing loss, associated symptoms such as vertigo, facial paralysis or unilateral tinnitus).
A monitoring role
By interviewing and use of the otoscopy, geriatricians can ensure that the hearing aid is well tolerated. Similarly, they can monitor compliance with wearing the hearing aid. Finally, their role is to monitor the progression of hearing loss, including for patients who wear a hearing aid, by ensuring that any development of severe hearing loss despite the use of an appropriate hearing aid is detected (Box 2).

Box 2: When to consider a cochlear implant?
Hearing Aids
When should hearing aids be recommended?
Hearing aids should be routinely recommended to any patient who complains of hearing loss and for whom an audiometric test shows a hearing loss.
Hearing loss on tonal and vocal audiometry is an essential prerequisite for hearing aids. However, it is difficult to suggest the use of a hearing aid to a patient who complains of hearing loss but who has perfectly normal hearing test results in silence. In this particular case, noise tests or a central hearing loss assessment should be carried out by the ENT specialist.
On the other hand, not all hearing losses on audiometry necessarily require a hearing aid. In fact, a discussion with the patient about their hearing loss, lifestyle, associated symptoms (tinnitus and hyperacusia in particular) and expectations is necessary prior to broaching the subject of a hearing aid trial.
For presbycusis in particular, it will sometimes be necessary to “convince” the patient, who in most instances has been referred at the request of their family, of the need for a hearing aid even though they have no complaints, as the progressive and insidious onset of the disease sometimes makes the patient blind (and deaf) to their health problem.
Benefits and limitations
The benefits of hearing aids in patients with hearing loss are sometimes obvious to everyone except the person affected.
Hearing aids help to protect against:
- Social isolation
- Linguistic impoverishment
- Cognitive decline and especially the alteration in short term memory
- Auditory fatigability
- Tinnitus and hyperacusia, which are sometimes associated
A hearing aid does not instantly restore hearing to that of a 20-year-old individual. There is a habituation phenomenon that usually takes several months for the patient’s hearing system to “get back on track”. When this is added to the initial discomfort of the earpiece, learning how to handle the device and the initial adjustments, it is not uncommon for the patient to be “disappointed” when fitting starts if they are not sufficiently prepared. Therefore, it is important to inform the patient that this is a long process that will require patience in the beginning before the benefits can be reaped after several weeks to months.
Most hearing aid failures are related to a poor understanding of the patient’s expectations (sometimes unrealistic) and above all, to the fact that the practitioner has not adequately informed the patient.
The ACHIEVE study, which should be completed in 2022, is expected to provide irrefutable evidence of the effect of hearing aids on cognitive decline in older adults with hearing loss [4].
The different types of hearing aids
There are many types of hearing aids (Figure 4) but they all have the same basic structure of one or more microphones, more or less complex signal processing, a loudspeaker outlet, all powered by a battery.
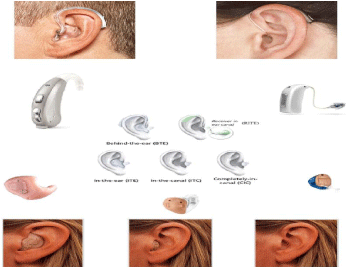
Figure 4: Different types of conventional hearing aids.
In general, the more miniaturized they are, the more performance suffers (in terms of power as well as technology or battery). In-the-canal hearing aids, which are dear to patients because of their “aesthetic” features, often require occlusion of the external auditory canal, which can cause discomfort. In addition, the loss of low frequencies due to the occlusive effect can have a negative impact on the perception of melody or the spatial localization of sounds. Therefore, careful consideration should be given before advising patients. Behind-The- Ear (BTE) hearing aids should be recommended as they are more efficient and versatile and at the same time, more and more discreet.
The choice of hearing aid depends on several factors:
- The patient’s degree of hearing loss and the frequencies involved
- The patient’s anatomy and comorbidities (Parkinson’s disease, rheumatoid arthritis, etc.)
- The patient’s aesthetic taste, as much as possible
- Associated symptoms (tinnitus or hyperacusia)
- The patient’s financial means or the reimbursement allocated by social security
Assessment and Speech and Language Therapy
Speech and language therapy assessment is the cornerstone of the speech and language therapist’s intervention. Using standardized tests, the difficulties as well as the strengths of each patient can be described, in order to sketch out an individualized rehabilitation project. This assessment allows the speech therapist to prescribe rehabilitation sessions that they will run with the patient’s agreement.
One of the major compensation strategies is the use of lip reading. By virtue of their certification of skills, speech and language therapists are authorized to provide lip reading training sessions. The work of mental compensation is also important to maintain “auditory fitness” and promote communication.
Adapting to hearing aids is not always easy: new and sometimes embarrassing perceptions can result, and the entire world of sound is left to be rediscovered. Speech and language therapy rehabilitation to accompany the equipment might be indicated. It supports and complements the work of hearing aid acousticians: it is up to them to find the best possible adjustment for the patient, and to the speech and language therapist to help the patient to become accustomed to the device and to optimize the hearing perception that it enables. For cochlear implants, speech therapy is all the more essential.
Conclusion
Until recently, ARHL was considered to be one of the inevitabilities related to aging. The prevention, diagnosis and management were certainly not the main concern for geriatricians who have many other age-related health issues on their hands. Recent awareness of the extent of this disability has been supported by numerous studies that examine the relationship between hearing loss and cognitive decline or the major economic impact of untreated hearing loss. Of all the diseases, hearing loss is the 4th leading cause of morbidity and will continue to increase worldwide according to the WHO. Prevention, screening, diagnosis, management and follow-up should be priority issues in the future, especially among the elderly.
References
- Deafness and hearing loss fact sheet from WHO.
- Quaranta N, Coppola F, Casulli M, Barulli MR, Panza F, Tortelli R, et al. The prevalence of peripheral and central hearing impairment and its relation to cognition in older adults. Audiol Neurootol. 2014; 19: 10-14.
- Livingston G, Sommerlad A, Orgeta V, Costafreda SG, Huntley J, Ames D, et al. Dementia prevention, intervention, and care. Lancet. 2017; 390: 2673- 2734.
- Deal JA, Goman AM, Albert MS, Arnold ML, Burgard S, Chisolm T, et al. Hearing treatment for reducing cognitive decline: Design and methods of the Aging and Cognitive Health Evaluation in Elders randomized controlled trial. Alzheimers Dement (NY). 2018; 4: 499-507.
- Reed NS, Altan A, Deal JA, Yeh C, Kravetz AD, Wallhagen M, et al. Trends in Health Care Costs and Utilization Associated With Untreated Hearing Loss Over 10 Years. JAMA Otolaryngol Head Neck Surg. 2019; 145: 27-34.
- Wilson BS, Tucci DL, Merson MH, O’Donoghue GM. Global hearing health care: new findings and perspectives. Lancet. 2017; 390: 2503-2515.
- Ciorba A, Hatzopoulos S, Bianchini C, Aimoni C, Skarzynski H, Skarzynski PH. Genetics of presbycusis and presbystasis. Int J Immunopathol Pharmacol. 2015; 28: 29-35.
- Ventry IM, Weinstein BE. Identification of elderly people with hearing problems. ASHA. 1983; 25: 37-42.
- Weinstein BE, Spitzer JB, Ventry IM. Test-retest reliability of the Hearing Handicap Inventory for the Elderly. Ear Hear. 1986; 7: 295-299.
- De Sousa KC, Swanepoel W, Moore DR, Myburgh HC, Smits C. Improving Sensitivity of the Digits-In-Noise Test Using Antiphasic Stimuli. Ear Hear. 2019.
- Amieva H, Ouvrard C, Giulioli C, Meillon C, Rullier L, Dartigues JF. Self- Reported Hearing Loss, Hearing Aids, and Cognitive Decline in Elderly Adults: A 25-Year Study. J Am Geriatr Soc. 2015; 63: 2099-2104.
- Belmin J, Pariel-Madjlessi S, Surun P, Bentot C, Feteanu D, Lefebvre des Noettes V, et al. The cognitive disorders examination (Codex) is a reliable 3-minute test for detection of dementia in the elderly (validation study on 323 subjects). Presse Med. 2007; 36: 1183-1190.
- Ftouh S, Harrop-Griffiths K, Harker M, Munro KJ, Leverton T. Guideline Committee, Hearing loss in adults, assessment and management: summary of NICE guidance. BMJ. 2018; 361: k2219.
- Michels TC, Duffy MT, Rogers DJ. Hearing Loss in Adults: Differential Diagnosis and Treatment. Am Fam Physician. 2019; 100: 98-108.
- Schwartz SR, Magit AE, Rosenfeld RM, Ballachanda BB, Hackell JM, Krouse HJ, et al. Clinical Practice Guideline: Earwax (Cerumen Impaction). Otolaryngol Head Neck Surg. 2017; 156: S1-S29.