Review Article
Austin J Infect Dis. 2014;1(2): 5.
Malaria, Therapeutic Options and Melatonin
Venkatramanujan Srinivasan1*, Rahimah Zakaria2 and Mahaneem Mohamed2
1International Medical Sciences Research Study Center, India
2Department of Physiology, School of Medical Sciences, University Sains Malaysia, Malaysia
*Corresponding author: Srinivasan V, Sri Sathya Sai Medical Educational and Research Foundation, An International Medical Sciences Research Study Center, Prasanthi Nilayam, 40-Kovai Thirunagar, Goldwins, Kovai, Coimbatore, 641014, Tamilnadu, India
Received: July 15, 2014; Accepted: August 15, 2014; Published: August 19, 2014
Abstract
Malaria is one of the most severe global infectious diseases caused by a group of parasites belonging to the genus Plasmodium. Each year it is estimated that over 220 million cases of malarial infections are reported. As drug resistance develops to existing antimalarial drugs, a new artemisinin-based combination therapy has been introduced. The goal of the antimalarial drug therapy should ideally be directed towards the reduction of malarial incidences by targeting the blood stage of the disease and in addition to eliminate all persistent asexual forms of these parasites in their entirety from the blood. As proteases like aspartic proteases and cysteine proteases are involved in the egress phenomenon which is where the parasites emerge from the infected erythrocytes, proteases are also considered as targets for antimalarial drugs and classes of drugs that have been developed along this line have proven most effective in reducing parasitemia. With the establishment of nocturnal melatonin levels in the host's blood acting as the "key signal" regulating Plasmodium life cycles in the erythrocyte, attention is now predominantly more focused on developing melatonin antagonists which will significantly reduce parasitemia and in turn, lessen the incidence of malarial infection. Recently, a number of melatonin antagonists with antimalarial activities have been developed. Melatonin not only acts as the signal for promoting the intraerythrocytic growth of malarial parasites, but similarly serves as an effective antioxidant in the erythrocyte, protecting them from free radical attacks within erythrocytes. Hence, working on the development of antimalarial drugs with anti-melatonergic activity will undoubtedly be extremely effectual in eliminating malaria.
Keywords: Malaria; Antimalarial drugs; Melatonin; Melatonin antagonists
Abbreviations
DDT: Dichloro-diphenyl-trichloroethane; WHO: World Health Organization; DHA: Dihydroartemisinin; UPS: Ubiquitin-proteosome-protein System; PKA: Protein Kinase A; GSK: Glaxo, Smith and Kline; Hz: Hemozoin; RBC: Red Blood Cell; malERA: Malaria Eradication Research Agenda; NK cells: Natural Killer Cells; IP3: Inositol Triphosphate
Introduction
Malaria, a most dreadful infectious disease, is caused by malarial parasites that infect and destroy red blood corpuscles resulting in a high fever, severe anemia and other complications that ultimately result in death in the majority of untreated cases. Recent findings reveal that nearly 40% of the world's population is at a potential risk of suffering from malaria due to being habitants of countries where the disease is endemic. According to a World Health Organization (WHO) report, more than 220 million cases of malarial infections are reported every year and the annual mortality rate due to malaria is 781,000 [1]. The groups of parasites that cause this disease belong to the genus Plasmodium and these species belong mostly to Plasmodium falciparum. Other species include Plasmodium vivax, Plasmodium malariae, Plasmodium ovale and Plasmodium knowlesi. The Global Malaria Eradication Programs launched by WHOrelied solely upon two tools, namely the use of the insecticide dichloro-diphenyl-trichloroethane (DDT) for vector control and the administration of chloroquine for treatment and prevention [2]. Although use of chemical insecticides such as DDT has been successful in eliminating the disease effectively, however, they also produce undesirable side effects on the ecosystem which have resulted in the elimination of non-targeted organisms [3]. The recent availability of genome sequences for Anopheles mosquitoes and Plasmodium parasites has opened up new vistas for novel interventions. A deeper understanding of the Plasmodium biology, vector habitat and proliferation are essential for exploring the discovery and inventions of new interventions that can effectively address malaria eradication programs.
Plasmodium falciparum, growth, development and proliferation
A single bite by an infected female Anopheles mosquito injects 100 or more sporozoites into the bloodstream and within minutes after being introduced into the blood they invade the hepatocytes where they remain either dormant or develop over several days ultimately forming schizonts that give rise to the blood stage infection [4]. The schizonts of the hepatic stage augment into several thousands of merozoites and following the rupture of liver cells these merozoites are released into the blood stream and invade erythrocytes. The intraerythrocytic parasites replicate synchronously and some merozoites develop into male and female gametocytes. The gametocytes are taken up into the gut of female mosquitoes during blood meals. The male gametocytes are activated and form gametes which fuse with the female gametocytes to form diploid ookinetes. These ookinetes migrate into the mid gut of the vector and pass through the gut wall to form oocytes and give rise to sporozoites by meiotic division. These sporozoites then find their way to the salivary glands of the mosquito, where they remain till they are transmitted into human beings by the bite of the mosquitoes [5]. Of the various stages, the intraerythrocytic stage of Plasmodium development is crucial for infecting humans and causing disease. It is the primary cause of malaria morbidity and mortality. All the clinical symptoms of malaria ranging from fever, anaemia and cerebral signs are all associated with blood stage of the Plasmodium lifecycle [6]. The progression of the malarial parasitic life cycle involves interplay between the parasite's changing environment and its own built-in genetic program [7]. The erythrocytic cycle of Plasmodium falciparum occurs over a period of 48 hours and consists of ring, trophozoite, and schizont that ultimately give rise to merozoites that are released into the blood stream at a specific time of the day-night cycle [8]. The role of the host's circadian system and pineal hormone melatonin are essential for the synchronous maturation of Plasmodium and its survival in the host [9]. The emergence of the parasites from the host's erythrocyte involves a coordinated occurrence of the rupture of parasitophorous membranes and erythrocyte membranes as well as the release of many proteases. These proteases, due to their involvement in parasitic development and invasion are targeted mainly by antimalarial drugs [10]. Of the various proteases identified in the Plasmodium, the ubiquitin-proteosome-protein system (UPS) is imperative for regulating the Plasmodium cell cycle. The UPS pathway in Plasmodium falciparum is said to be involved in specific functions related to pathogenesis or virulence [11]. Recent studies conducted by Dr Koyama and his associates have revealed that melatonin up regulates the genes related to UPS and evidence for this has been obtained by using melatonin receptor antagonists, such as luzindole which inhibits this system [12]. Understanding the importance of activation and modulation of kinases in Plasmodium biology is advocated as an essential step for the future development of antimalarial drugs. Kinase inhibitors such as genistein and tyrphostin block the P. falciparum cell cycle and hence have the potential of acting as antimalarial drugs [13].
Recent studies of signaling pathways in P. falciparum have exhibited that Plasmodium has receptors for activated C kinase and is expressed during the intraerythrocytic cell cycle [14]. Although G protein-coupled receptors are absent in Plasmodium, the existence of serpentine or heptahelical receptor genes in its genome and the transcripts of which have been identified in the asexual stages of the parasite [15]. Melatonin acts by inducing cAMP production and increases intracellular Ca2+ activating protein kinase A (PKA) and the protein kinase in turn regulates Plasmodium growth and differentiation. Both cAMP and Ca2+ control different aspects of Plasmodium development and combined action of both are crucial for parasitic function. The blocking of either one of these messengers inhibits PKA and arrests parasitic growth [16].
Malaria control: therapeutic options
Despite the launch of the global malaria eradication programs by WHO in 1955 [2,17] more people are at risk of suffering from malaria now than ever before [18]. Malarial parasites are clever enough to avoid the host immune system as they develop various sophisticated mechanisms [18]. The need for developing effective human malarial vaccines are imperative and will aid in the eradication of malaria at a global level [19] which was indeed developed by Glaxo, Smith and Kline (GSK). The vaccine RTSS developed by GSK, was found effective in reducing the risk of clinical malaria and in preventing the episodes of severe malaria for a period of 18 months [20]. The Bill and Melinda Gates foundation introduced an agenda in 2007 with the final goal of eradicating all species of Plasmodium that caused malaria infection in humans and this was supported by WHO [21]. The antimalarial drugs have been found to be effective in eradicating all four species of malarial parasites that have been identified so far.
Recently the role of malaria pigment has received attention with regards to the control of malarial parasitic development. During the intraerythrocytic stage of development, the trophozoite form feeds on hemoglobin. The toxic heme (to the parasite) is aggregated to form insoluble dark-brown crystals known as hemozoin (Hz) which accumulates inside the parasitized erythrocyte. Phagocytes acquire Hz through phagocytosis of parasitized red blood cells (RBC) or free Hz crystals released by schizont rupture [22]. Hz through a variety of mechanisms causes the suppression of immune mechanisms and is said to be responsible for associated clinical problems. Hz ingestion by mononuclear cells enhances the production of pro-inflammatory mediators such as tumor necrosis factor-a and nitric oxide [23,24]. The blocking of Hz formation has been recommended to increase immune mediated parasitic clearance and thus it will be useful in preventing the formation and transmission of gametocytes [25]. Chloroquine, quinine, and artemisinin are efficient in blocking the formation of Hz from heme. Effective targeting of Hz with these drugs alludes to the reduction of high morbidity and mortality associated with this disease .Since resistance to these drugs has become a major problem, modifications of quinine, chloroquine, or artemether to overcome parasitic resistance will be of immense help in achieving the goal [25]. P. vivax causes intense morbidity in countries of Latin America and Asia. Chloroquine is the drug of choice to treat malaria caused by P. vivax. Therefore, it was felt that monitoring the development and resistant phenotypes and genotypes should be given top priority wherever endemic malaria is present. During a recent study, in vitro methods were implemented in order to examine anti-malarial drug sensitivity towards Plasmodium and to various drugs which were simultaneously assayed. The results of this study indicate that all P. falciparum isolates were revealed to be chloroquine resistance. The majority of P. vivax and P. falciparum isolates were sensitive to mefloquine and artesunate [26]. The Plasmodium parasite has the ability of developing drug resistance at an acutely rapid rate; finding new and effective methods which effectively fight against malaria must henceforth be the top global health priority. Structure-based drug discoveries centered on structural genomics is a newly developed field introduced for the speedy development of anti-malarial programs and it will inevitably assist in accelerating drug development [27]. Recently, the Malaria Eradication Research Agenda (malERA) has been introduced which highlights the importance of both basic science and operational research in its fight against malaria [28]. The malERA emphasizes the need for the development of basic research in fully comprehending malaria parasite biology in addition to the operational research in the global fight against malaria [29]. The role of human natural killer cells (NK cells) and macrophages in the control of malarial parasitic infection was recently evaluated by using the RICH mouse model (RBC supplemented, immune cell-optimized humanized mice) which remains manifest in multiple cycles of P. falciparum infection. By depleting NK cells, a noteworthy (seven-fold) increase of parasitemia was noted at 48 hours post infection in these RICH mice. The removal of macrophages did not result in parasitemia. In vitro studies revealed that NK cells preferentially interacted with infected RBCs causing the activation of NK cells and the elimination of infected RBCs. This study was in favour of the notion that NK cells are essential for controlling malarial infection [30]. Any procedure that activates NK cells can be useful in controlling malarial infection in infected individuals. Based on various effective measures that are currently available, the working group on malaria and the UN Millennium Project have recommended the use of these measures for reducing the malaria morbidity and mortality by 75% in 2015 from its baseline level of 2005 [31].
Melatonin-based therapeutic options: recent studies
Melatonin, the neurohormone, is secreted mainly by the pineal gland and is also formed by multitudinous other organs in the body such as the gastrointestinal tract, thymus, and skin, but the production of melatonin by the pineal gland is circadian in nature and is controlled by the suprachiasmatic nucleus of the hypothalamus. The nocturnal production and release of melatonin either into the blood or into CSF has either very high or very low levels of melatonin which are deducted in the blood during the daytime [32]. The light-dark cycle entrains the melatonin rhythm generated by the suprachiasmatic nucleus of the hypothalamus by acting through the retino-hypothalamic tract. Melatonin secretion existent in humans or other species is used as a "signal essential for the synchronization of Plasmodium falciparum life cycle" [33]. Melatonin found in the blood of the host (human) crosses the erythrocyte membrane, binds with the receptors found on the membrane of P. falciparum and activates phospholipase C. Phospholipase C induces the production of the inositol triphosphate (IP3). IP3 stimulates the release of Ca2+ from the endoplasmic reticulum of the parasite that initiates the synchronous maturation of the malaria parasite [33]. Genomic studies have established that both Ca2++ and cAMP are essential for coordinating various aspects of Plasmodium function and both of these messengers are activated by melatonin [34]. Melatonin activates PKA through these two messengers only [14]. Melatonin modifies the life cycle of P. Falciparum by up regulating the genes involved in the UPS as well as controlling signaling pathways which has been discussed in the earlier section and this effect was blocked by the melatonin receptor antagonist luzindole [12]. The effects of melatonin on Plasmodium cell signaling pathways are also blocked by the phospholipase C inhibitor, U 73122 [35]. Not only malaria parasite Plasmodium but another parasite Trypnasoma cruzi also requires melatonin for regulating its cell cycle but in this case the hormone is synthesized by the parasite itself [36]. Similar to the nocturnal activation of Plasmodium by the host's melatonin levels in the blood, the filarial parasite Wuchereria bancrofti are released in to the host's blood during the night time, and this is triggered by high nocturnal levels of melatonin in the host's blood [37]. The endogenous circadian rhythm in these parasites (both Plasmodium and Wuchereria bancrofti) although self-sustained, they are tuned to environmental time cue, namely the melatonin levels in the host's blood [37]. The above mentioned findings point out that cell cycles of P. falciparum or P. vivax, mainly the development of asexual stages can be blocked by suppressing melatonin actions using the common melatonin receptor (MT1 and MT2) antagonist luzindole [38,39]. As exposure to bright light can suppress nocturnal melatonin levels [40], bright light can be applied at night to remove "key signal" (high plasma melatonin levels in the host) that is essential for synchronizing malaria parasite growth and development. A light mask treatment can deliver light through the subject's eyelids at specific intensities (enough to suppress plasma melatonin levels) without disturbing the subject's sleep in the night [41].
Melatonin derivatives: novel potential antimalarial drugs?
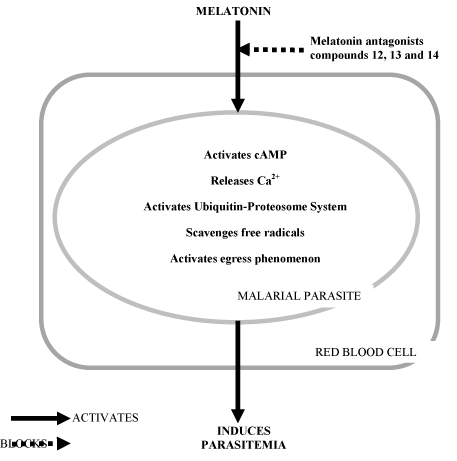
As we discussed in the earlier paragraphs, melatonin plays an imperative role in the control of malaria parasite replication and hence targets and blocks melatonin's action on plasmodium development which will significantly help to curb this infection and disease. Much interest has been provoked in the development of melatonin antagonists as antimalarial drugs leading to the development of new melatonin related compounds 7-11 and 12-16 which may be applied in an attempt to block the parasite's development [42]. These N-heterocyclic derivatives were developed by modifying the structure of the parent compound namely melatonin. Substituting hydrogen in the methoxy group and adding diverse substituents to the carboxamide at the position C3 of the indole ring of melatonin resulted in the synthesis of 7-11 compounds. Compounds 12-16 were developed by the substitution of the amide group. When tested for antimalarial activity, compounds 12, 13, and 14 exhibited antimalarial activity. Among the indole compounds with antimalarial activity which was synthesized earlier NITD 609 belonging to spiroindolone was found to interfere with the transmission of the parasite to the vector Anopheles and this drug is applied in phase I of the clinical trial [43]. The Compounds 7-11 belonging to 2-(indol-3-yl) ethylamine derivatives and compounds 12-16 belonging to (2-(5-methoxy -1H-indol -3yl) amine when tested for antimalarial activities, compounds 9 and 10 were able to block the effect of melatonin in Plasmodium. New derivatives from the compounds 12-14 with antimalarial activities are in progress [42]. As circadian changes in the melatonin levels of the host environment (plasma or blood) acts as a "key signal" for synchronizing the growth, development and differentiation of P. falciparum or P. vivax or P. knowlesi, devices, agents, or drugs that either prevent or block the actions of melatonin on malarial parasites [44] will be beneficial in curbing malaria parasite infection. Besides using the nocturnal melatonin level as a signal for synchronizing its life cycle, malarial parasites also use melatonin as an antioxidant for protecting themselves from free radical attacks in the oxygen rich environment where they have to thrive and multiply (intraerythrocytic stage) [45]. Hence, the usage of melatonin antagonist derivatives will be ideal for blocking the actions of melatonin on malaria parasite growth, differentiation and development. A schematic diagram of the role of melatonin in the activation of the key events which cause malarial parasitemia is demonstrated in Figure 1. Blocking melatonin actions on malaria parasites will be an effective method for inhibiting the malarial life cycle.
Figure 1: Antimalarial actions of melatonin antagonists.
Conclusion
Malaria continues to be one of the major global health problems in developing countries and is the most common cause of death from infectious diseases. More than 3.3 billion people live in areas at risk of malaria [46]. In most cases, infection of the human host with malaria parasites commences with the bite of an infected Anopheles mosquito. Antimalarial drug therapy which was introduced in the early stages was quinine, a natural product and this was followed by synthetic derivatives 8-aminoquinolone, primaquine and the 4-aminoquinolone chloroquine. All these drugs cause parasite death by preventing the polymerization of the toxic byproduct of hemoglobin breakdown namely the heme into non-toxic product Hz. This Hz is the main cause for the occurrence of severe malaria anemia, immunosuppression and cytokine dysfunction apparent in malarial infected individuals, especially in children and pregnant women. Targeting this pigment is considered as essential for the development of new therapeutic tools against malaria. Chloroquine, quinine, and artemisin all block the formation of Hz from heme. As malarial parasites have developed resistance against these drugs, modification of the structures of quinine, chloroquine and artemisinin has become inevitable for targeting the disease. As both intracellular calcium and cAMP are essential for coordinating various aspects of the malarial life cycle, inhibiting the formation or reducing the levels of these agents within the cytoplasm of malarial parasites will be the other possible mechanisms for controlling the growth and spread of malaria. Spiroindole derivatives have been shown to block the entry of Ca2++ into the malarial parasites and the drug was patented in 2013 [47]. With the identification of melatonin as a nocturnal signal essential for synchronizing the Plasmodium life cycle, interest has been diverted to the development of drugs or agents that block the actions of melatonin on Plasmodium and recently melatonin derivatives that are antagonists to melatonin have been developed and found effective in inhibiting malarial parasitic growth. Employing these melatonin antagonists in clinical trials for treating malaria infected patients will benefit not only the patients infested with the disease but will assist in preventing the spread of malaria itself.
References
- World Health Organization. World Malaria report, WHO Press, Geneva, Switzerland. 2010.
- Greenwood BM, Fidock DA, Kyle DE, Kappe SHI, Alonso PL, Collins FH. WHO, Making a difference. The World Health Report. Health Millions.1999; 25: 3-5.
- Rohani A, Chu WL, Zamree I, Abdullah AG, Saadiyah I, Lee HL, et al. Microalgae associated with mosquito breeding grounds in Malaysia. Tropical Bio Med. 2002; 19: 83-96.
- Roberts DR, Manguin S, Mouchet J. DDT house spraying and re-emerging malaria. Lancet. 2000; 356: 330-332.
- Beier JC. Malaria parasite development in mosquitoes. Annu Rev Entomol. 1998; 43: 519-543.
- Schofield L. Intravascular infiltrates and organ-specific inflammation in malaria pathogenesis. Immunol Cell Biol. 2007; 85: 130-137.
- Baker DA. Malaria gametocytogenesis. Mol Biochem Parasitol. 2010; 172: 57-65.
- Garcia CR, Markus RP, Madeira L. Tertian and quartan fevers: temporal regulation in malarial infection. J Biol Rhythms. 2001; 16: 436-443.
- Hotta CT, Markus RP, Garcia CR. Melatonin and N-acetyl-serotonin cross the red blood cell membrane and evoke calcium mobilization in malarial parasites. Braz J Med Biol Res. 2003; 36: 1583-1587.
- Blackman MJ. Malarial proteases and host cell egress: an 'emerging' cascade. Cell Microbiol. 2008; 10: 1925-1934.
- Blackman MJ. Malarial proteases and host cell egress: an 'emerging' cascade. Cell Microbiol. 2008; 10: 1925-1934.
- Koyama FC, Ribeiro RY, Garcia JL, Azevedo MF, Chakrabarti D, Garcia CR, et al. Ubiquitin proteasome system and the atypical kinase PfPK7 are involved in melatonin signaling in Plasmodium falciparum. J Pineal Res. 2012; 53: 147-153.
- Garcia CR, Dluzewski AR, Catalani LH, Burting R, Hoyland J, Mason WT, et al. Calcium homeostasis in intraerythrocytic malaria parasites. Eur J Cell Biol. 1996; 71: 409-413.
- Madeira L, DeMarco R, Gazarini ML, Verjovski-Almeida S, Garcia CR. Human malaria parasites display a receptor for activated C kinase ortholog. Biochem Biophys Res Commun. 2003; 306: 995-1001.
- Madeira L, Galante PA, Budu A, Azevedo MF, Malnic B, Garcia CR, et al. Genome-wide detection of serpentine receptor-like proteins in malaria parasites. PLoS One. 2008; 3: e1889.
- Gazarini ML, Beraldo FH, Almeida FM, Bootman M, Da Silva AM, Garcia CR. Melatonin triggers PKA activation in the rodent malaria parasite Plasmodium chabaudi. J Pineal Res. 2011; 50: 64-70.
- WHO. Global malaria control and elimination. Report of technical review. Global Malaria control and elimination.
- Fidock DA. Drug discovery: Priming the antimalarial pipeline. Nature. 2010; 465: 297-298.
- Saleh JA, Yusuph H, Zailani SB, Aji B. Malaria vaccine: the pros and cons. Niger J Med. 2010; 19: 8-13.
- Guinovart C, Aponte JJ, Sacarlal J, Aide P, Leach A, Bassat Q, et al. Insights into long-lasting protection induced by RTS, S/AS02A malaria vaccine: further results from a phase IIb trial in Mozambican children. PLoS One. 2009; 4: e5165.
- Roberts L, Enserink M. Malaria. Did they really say ... eradication? Science. 2007; 318: 1544-1545.
- Schwarzer E, Turrini F, Ulliers D, Giribaldi G, Ginsburg H, Arese P, et al. Impairment of macrophage functions after ingestion of Plasmodium falciparum-infected erythrocytes or isolated malarial pigment. J Exp Med. 1992; 176: 1033-1041.
- Sherry BA, Alava G, Tracy KJ, Martiney J, Cerami A, Slater AF. Malaria specific metabolite hemozoin mediates the release of several potent endogenous pyrogens. (TNF, MIP-1a, MIP-1β) in vitro and altered thermoregulation in vivo. J Inflammation. 1995; 45: 85-96.
- Keller CC, Kremsner PG, Hittner JB, Misukonis MA, Weinberg JB, Perkins DJ. Elevated nitric oxide production in children and malaria anemia: hemozoin-induced nitric oxide synthase type-2 transcripts and nitric oxide in blood mononuclear cells. Infect Immun. 2004; 72: 4868-4873.
- Ihekwereme CP, Esimone CO, Nwanegbo EC. Hemozoin inhibition and control of clinical malaria. Adv Pharmacol Sci. 2014; 2014: 984150.
- Aguiar AC, Pereira DB, Amaral NS, De Marco L, Krettli AU. Plasmodium vivax and Plasmodium falciparum ex vivo susceptibility to anti-malarials and gene characterization in Rondônia, West Amazon, Brazil. Malar J. 2014; 13: 73.
- Drinkwater N, McGowan S. From crystal to compound: structure-based antimalarial drug discovery. Biochem J. 2014; 461: 349-369.
- Malera. Malaria eradication research agenda.
- Navarro V. The new conventional wisdom: an evaluation of the WHO report, Health Systems: Improving Performance. Int J Health Serv. 2001; 31: 23-33.
- Chen Q, Amaladoss A, Ye W, Liu M, Dummler S, Kong F, et al. Human Natural killer cells control Plasmodium falciparum infection by eliminating infected red blood cells. PNAS. 2014; 111: 1479-1484.
- El-Moamly A. Malaria elimination: needs assessment and priorities for the future. J Infect Dev Ctries. 2013; 7: 769-780.
- Arendt J. Biological Rhythms. International Med. 1983; 3(2): 6-9.
- Hotta CT, Gazarini ML, Beraldo FH, Varotti FP, Lopes C, Markus RP, et al. Calcium-dependent modulation by melatonin of the circadian rhythm in malarial parasites. Nat Cell Biol. 2000; 2: 466-468.
- Beraldo FH, Almeida FM, da Silva AM, Garcia CR. Cyclic AMP and calcium interplay as second messengers in melatonin-dependent regulation of Plasmodium falciparum cell cycle. J Cell Biol. 2005; 170: 551-557.
- Florens L, Washburn MP, Raine JD, Anthony RM, Grainger M, Haynes JD, et al. A proteomic view of the Plasmodium falciparum life cycle. Nature. 2002; 419: 520-526.
- Macías M, Rodríguez-Cabezas MN, Reiter RJ, Osuna A, Acu&nTilde;a-Castroviejo D. Presence and effects of melatonin in Trypanosoma cruzi. J Pineal Res. 1999; 27: 86-94.
- Sack RL. Host melatonin secretion is a timing signal for the release of W. bancrofti microfilaria into the circulation. Med Hypotheses. 2009; 73: 147-149.
- Srinivasan V, Ahmad AH, Mohamed M, Zakaria R. Melatonin effects on Plasmodium life cycle: new avenues for therapeutic approach. Recent Pat Endocr Metab Immune Drug Discov. 2012; 6: 139-147.
- Srinivasan V, Mohamed M, Zakaria R, Ahmad AH. Malaria, anti malarial drugs and the role of melatonin. Infect Disord Drug Targets. 2012; 12: 371-379.
- Lewy AJ, Wehr TA, Goodwin FK, Newsome DA, Markey SP. Light suppresses melatonin secretion in humans. Science. 1980; 210: 1267-1269.
- Ando K, Kripke DF, Cole RJ, Elliott JA. Light mask 500 lux treatment for delayed sleep phase syndrome. Prog Neuropsychopharmacol Biol Psychiatry. 1999; 23: 15-24.
- Schuck DC, Jordao AK, Nakabashi M, Cunha AC, Ferreira VF, Garcia CRS. Synthetic indole and melatonin derivatives exhibit antimalarial activity on the cell cycle of the human malaria parasite Plasmodium falciparum. Eur J Med Chem. 2014; 78: 375-382.
- Rottmann M, McNamara C, Yeung BK, Lee MC, Zou B, Russell B, et al. Spiroindolones, a potent compound class for the treatment of malaria. Science. 2010; 329: 1175-1180.
- Srinivasan V, Zakaria R, Mohamed M, Saleh RM. Effects of Melatonin Derivatives on Human Malaria Parasite Plasmodium falciparum. Recent Pat Endocr Metab Immune Drug Discov. 2014; 8: 102-108.
- Clark IA, Hunt NH. Evidence for reactive oxygen intermediates causing hemolysis and parasite death in malaria. Infect Immun. 1983; 39: 1-6.
- Lal SS, Uplekar M, Katz I, Lonnroth K, Komatsu R, Yesudian Dias HM, et al. Global Fund financing of public-private mix approaches for delivery of tuberculosis care. Trop Med Int Health. 2011; 16: 685-692.
- Mikoshiba K, Enomoto M, Kawazu SI. Method for treating malaria; method for killing malaria and use of methods. 2013. US 20130296230.