
Special Article – Medical Microbiology
Austin J Microbiol. 2015; 1(2): 1009.
The Final Stage of Developing Genetically Modified Inactivated Sabin Vaccine for the Eradication of Poliovirus
Pinn Tsin Isabel Yee and Chit Laa Poh*
Virology Research Group, Vice Chancellor’s Office, Sunway University, Bandar Sunway, Kuala Lumpur, Selangor 47500, Malaysia
*Corresponding author: Chit Laa Poh, Virology Research Group, Vice Chancellor’s Office, Sunway University, Bandar Sunway, Kuala Lumpur, Selangor 47500, Malaysia
Received: November 05, 2015; Accepted: December 22, 2015; Published: December 24, 2015
Abstract
The polio vaccine is one of the most successful vaccines which have reduced worldwide poliomyelitis caused by poliovirus infection. The poliovirus is a human enterovirus which belongs to the Picornaviridae family. Formalin- Inactivated Poliovirus Vaccine (IPV) developed by Salk was the first poliovirus vaccine to be licensed in 1955. The IPV was the only poliovirus vaccine available until licensure of the Oral Poliovirus Vaccine (OPV) in1963. The OPV is a live attenuated vaccine that has long-lasting immune response but requires several boosters, is safe and effective. Elimination of poliomyelitis in the developing world was achieved mainly through mass vaccination with the OPV despite its ability to revert to the wild type. However, polio still persistently remained endemic in Afghanistan and Pakistan. One of the most anticipated next generation vaccines is an IPV based on the attenuated Sabin poliovirus strains. An attenuated Sabin IPV (sIPV) has been used in Japan for the past 3 years in their routine immunization program and licensed in China to be introduced in routine immunization in several provinces. The sIPV is produced from poliovirus strains that have an antigenic structure identical to the currently used wild-type strains but were rendered non-pathogenic by genetic manipulations. Of interest would be the manipulations of the IRES within the 5’-NTR to produce geneticallystable OPV strains such as the removal of U-G base-pairs in domain V of the 5’-NTR in the Poliovirus (PV) and insertion of the Cis-acting replication element (cre) from the P2 region to a position near the 5’ end to reduce the risk of loss of this part of the genome through recombination. Other methods include creation of PV strains with high polymerase fidelity, codon pair bias deoptimization and microRNA sequence insertion. Thermostable PV Virus-Like Particles (VLPs) that have similar antigenicity as the wild type PV but are stable enough to allow vaccine production are currently being constructed. The development of such new sIPVs could serve as more promising vaccines to eradicate poliomyelitis from the world.
Keywords: Poliovirus; Poliomyelitis; Vaccine; Genome; sIPV
Introduction
Polio virus vaccines
Inactivated polio vaccine: Poliovirus (PV) is the etiological agent of poliomyelitis and belongs to Enterovirus species C within the Picornaviridae family. Poliomyelitis was a public health scare in the 1950s, even in countries with the best health systems and hygiene practices in place. This thereby led to the raising of funds to support research in the development of a polio vaccine. The Inactivated Poliovirus Vaccine (IPV) was the first poliovirus vaccine to be licensed in 1955. IPV was developed by Salk et al. (1954) and was prepared by formalin-inactivation of three wild-type virulent strains which are the Mahoney (type 1), MEF-1 (type 2) and Saukett (type 3). The United States started using the IPV and it showed such high efficacy that other countries around the world started to follow suit.
Although IPV is considered safe, there is a risk of exposure to the wild type strain during the manufacturing process. Figure 1.1 shows the manufacturing process for the IPV. During monovalent bulk preparation, Vero cells were expanded using two pre-culture steps and cell culture followed by virus culture. The PV was purified using normal flow filtration for clarification, tangential flow filtration for concentration and followed by two chromatography steps involving size exclusion and ion exchange chromatography. Purified virus was inactivated using formaldehyde. Subsequently the IPVs were mixed to obtain trivalent bulk prior to formulation and filling [1]. Due to the need to cultivate large amounts of the live poliovirus which involved complex manufacturing and purification processes, exposure of workers to the live virus must be safely guarded. At the Cutter Laboratories, insufficient inactivation of the IPV led to paralysis in almost 200 vacinees and their contacts [2]. This incident resulted in the temporary halt of the use of IPV and encouraged research groups worldwide to produce a live attenuated polio vaccine. Although IPV has an excellent track record on efficacy, it had poor induction of intestinal immunity, required cold-chain, booster injections and had expensive and potentially dangerous manufacturing processes with the wild type virulent virus. As such, large-scale clinical trials were evaluated using several live attenuated PV strains [3].
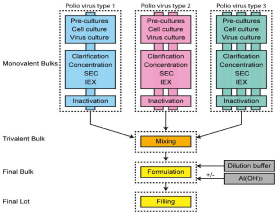
Figure 1: Process overview for preparation of trivalent IPV. Monovalent
bulks are prepared for each poliovirus (type 1, 2 and 3) separately. Virus
was harvested, clarified, concentrated, and purified, first on size using Size
Exclusion Chromatography (SEC) and Ion Exchange Chromatography (IEX).
Purified virus were mixed with buffers and Aluminium Hydroxide Al(OH)3 prior
to filling [1].
Oral polio virus vaccine: The Oral Poliovirus Vaccine (OPV) is an attenuated vaccine which has reduced worldwide poliomyelitis caused by PV infection. The IPV was the only poliovirus vaccine available until licensure of the Oral Poliovirus Vaccine (OPV) in 1963. There was a need for an OPV then as it is a live attenuated vaccine which has long-lasting immune response but requires several boosters, is safe and effective [4]. Elimination of poliomyelitis in the developing world was achieved mainly through mass vaccination with the OPV despite its ability to revert to the wild type in an estimated 1 out of 250, 000 to 1 out of 800, 000 [5,6].
The OPV was produced by micro-carrier technology and passage of the virus in primary monkey kidney cells at sub-physiological temperatures that generated spontaneous mutations in the viral genome [7]. The mutated strains with low virulence were selected and used as vaccines. The process was later scaled up by using 750- L bioreactors and replacing tertiary monkey kidney cells with Vero cells [1]. This has led to successful production of Oral Polio Vaccines (OPV) which have reduced worldwide poliomyelitis by the mid- 1980s.
As such, the World Health Assembly declared in 1988 that polio should be eradicated by the year 2000, aligned with the success of the smallpox eradication program. However, the eradication deadline was repeatedly postponed and has not been met till today due to low vaccination coverage. This is due to the fact that there are several countries where polio persistently remained endemic such as in Pakistan and Afghanistan. Therefore, there is a need to have a greater understanding of the molecular determinants of neurovirulence in PV to develop new and better vaccines based on genetic manipulations to render the virus non-pathogenic, yet containing similar antigenic structures to the wild type polio virus.
Molecular determinants of neurovirulence: The polio virus is an enterovirus from the family Picornaviridae. The PV has a 5’ Non- Translated (NTR) cloverleaf structure and a 3’-poly (A) tail. Domain I is important for virus replication and domains II-VI encompass the Internal Ribosome Entry Site (IRES) that directs translation of mRNA by internal ribosome binding (Figure 1.2). If there are mutations in the 5’-NTR, this decreases multiplication efficiency, alters cell tropism and attenuates virulence [8].
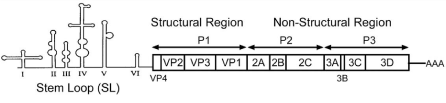
Figure 2: Structure and genome of Poliovirus. All structural proteins:
VP1, VP2 and VP3 are encoded by the P1 region of the genome. The P2 and
P3 genes encode for seven non-structural proteins: 2A-2C and 3A-3D [8].
There are three attenuated strains being used as OPV: Sabin 1 was derived from the Mahoney strain, Sabin 2 was derived from the P172 strain and Sabin 3 was derived from the Leon strain. Identification of the genetic determinants of attenuation of the Sabin OPV strains has been comprehensively reviewed [8]. The complete sequences of the three poliovirus genomes and the development of infectious poliovirus Complementary Deoxyribonucleic Acid (cDNA) clones have led to the systematic investigations of the critical mutations responsible for the attenuated phenotypes of the Sabin OPV strains. From the analysis of Nucleotide (NT) sequences present in the three poliovirus Sabin strains, nucleotide substitutions which were critical in attenuating mutations in the virulent strains isolated from cerebrospinal fluid were identified. There are 57 nucleotide substitutions distinguishing the Sabin 1 strain from its parent strain [9]. Among these nucleotide substitutions, the A480G in the IRES is the most important determinant of the attenuated phenotype of Sabin 1. Their studies strongly suggested that nt. 480 influences the formation of a highly ordered structure in the 5’-NTR that is responsible for neurovirulence [10]. Four other nucleotide substitutions contributing to the attenuated phenotype were mapped to the capsid region. There was one in VP4, one in VP3 and two in VP1. In addition, there was also one substitution that contributed to the temperature-sensitive phenotype mapped to the 3DPol region [11,12].
However, there were only 2 nt. substitutions found in the Sabin 2 strain that appeared at position 481 within the IRES region and position 2909 within VP1 (Figure 1.3). For Sabin 3, a total of 10 nt. substitutions were found to differ from its parent strain, but only 3 substitutions appeared to be the main determinants for the attenuated phenotype (C274U in IRES, C2034U in VP3, and U2493C in VP1) [13]. Sabin 3 strain was also found to be the most genetically unstable of the three Sabin strains. As a result of the analysis of the molecular determinants of attenuation, in vitro construction of piconaviruses with reduced-virulence could be performed via the introduction of mutations in the 5’-NTR to reduce the efficiency of viral replication.
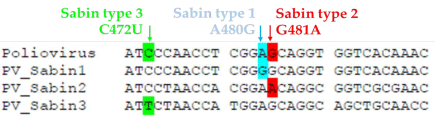
Figure 3: Three genotypes of Poliovirus Sabin vaccine strains. All
contain a determinant of attenuation in the 5’ NTR of ssRNA mutation that
reduces viral replication. They are nt. 480 in Sabin 1, nt. 481 in Sabin 2 and
nt. 472 in Sabin 3 [10].
Recent advances in developing new polio vaccines: In 2000, the Advisory Committee on Immunization Practices (ACIP) has recommended only the use of Inactivated Poliovirus Vaccine (IPV) for childhood polio vaccination in the United States and subsequently, for other countries. All children would receive four doses of IPV at ages 2, 4, 6-18 months and 4-6 years. The ACIP wanted to decrease the risk of Vaccine-Derived Poliovirus (VDPV) and Vaccine Associated Poliomyelitis (VAPP) (ranging from 1 in 700, 000 to 1 in 3.4 million first doses), yet still maintain the benefits of OPV [14,15]. The riskbenefit equation altered after eradication of wild type poliomyelitis in industrialised countries and the only source of poliomyelitis seemed to be derived only from the OPV [16].
Due to this gradual shift from OPV back to IPV, one of the most anticipated next generation vaccines is an IPV based on the attenuated Sabin poliovirus strains, producing a Sabin IPV (sIPV), also known as the enhanced IPV [17]. The sIPV is manufactured from attenuated Sabin virus strains instead of wild-type PVs. In addition, it has already been used in Japan for the past 3 years in their routine immunization program and currently licensed in China to be introduced in routine immunization in several provinces. They would be expected to be in high demand to enhance worldwide production of the sIPV. The cost of sIPV production has increased [18] since the World Health Organisation (WHO) launched the Global Action Plan for Wild Poliovirus Laboratory Containment III (GAPIII) to make biosafety requirements more stringent. As a result of the GAPIII, manufacturing costs for sIPV have increased significantly, leading to challenges in implementation especially in developing countries where they are most needed.
Although there is data to support broad protection of the sIPV at varying efficacies, more studies have to be conducted on issues concerning the quality control, evaluation of the sIPV and strategies to make the sIPV affordable for low-income countries where they are needed the most [19]. Hence, WHO had convened a technical working group on May 2013 participated by world-wide experts from academia and the industry who are involved in the development, manufacture, authorization and testing of sIPV. As a result, WHO has incorporated such concerns into its publication of technical guidelines for the quality, safety and efficacy of the sIPV vaccine to assist in the production and control of sIPV [20].
Hence, alternative vaccine strains that require less stringent biocontainment and manufacturing processes that can be carried out in developing countries are much needed. This approach potentially lowers production costs and is encouraged by the WHO [21]. By the beginning of the 21st century, most developed countries have switched exclusively to IPV. As such, there have been much studies performed to produce a third generation IPV. This new generation of IPV differs from the sIPV as it is produced from poliovirus strains that have an antigenic structure identical to the currently used wild-type strains but were rendered non-pathogenic by genetic manipulations. This was carried out through several methods like modification of the 5'-NTR of the poliovirus genome, mutation of the poliovirus 3D polymerase gene, utilising codon-pair bias de-optimization and microRNA sequence insertion [16].
One of the most important virulence determinants in the poliovirus is in the Internal Ribosome Entry Site (IRES) within the 5’- NTR of the PV RNA. Hence, manipulation of the IRES with multiple nucleotide substitutions, insertions or deletions could be used to create a more stable attenuated PV strain for IPV manufacture [22,23]. Inclusion of weaker base pairing also reduces the stability of the IRES region and thereby, decreases the rate of reversion [24]. Another approach would be to introduce mutations in the 3D Polymerase gene sequence of the PV [25] that can increase replication fidelity and reduce pathogenicity. These mutations could reduce viral virulence, yet retain the antigenic properties of the wild type PV.
An increasingly popular approach would be to carry out codonpair bias deoptimization. This is based on the fact that there are preferred combinations of neighbouring codons. Hence, by swapping different but synonymous codons within the same sequence will result in changed pairs of codons but the original amino acid sequence was retained. This was observed by Coleman et al. who carried out the deoptimization approach that led to decreased viral fitness and the overall attenuation in PV neurovirulence [26]. Who carried out the deoptimization approach that led to decreased viral fitness and an overall attenuation in PV neuro-virulence? They discovered that deoptimization decreased significantly the quantity of infectious PV although the number of virus particles remained the same. This implies that although the infectious viral titre decreased, the amount of antigen required for making the IPV remained sufficient.
Recent studies have demonstrated another emerging approach that could represent an attractive addition to the current research on IPV. Eukaryotic cells produce microRNAs (miRNA) that are 20- 24 nucleotides long, non-coding RNAs that can increase or prevent translation of messenger RNA (mRNA) by binding to it and causing cleavage or translational block, dependent on sequence homology with target mRNA [27]. By manufacturing short RNA pieces that are complementary to mRNA prevalent in tissues that are viral targets, this could reduce pathogenicity. Barnes et al. (2008) engineered poliovirus to carry neuronal-specific miR-124 RNA which prevented viral replication in the central nervous system and thus led to significant attenuation of neurovirulence in infected mice. These viruses could also be grown for IPV production in cultured cells that do not express this particular miRNA [28].
Such modifications have been carried out by various research groups and reviewed by [19]. An investigation by [1]. An investigation by Thomassen et al. produced an IPV at industrial scale that met quality criteria, were immunogenic in rats, showed no toxicity in rabbits and could be released for testing in clinical phase I/IIa [1]. In addition, Japan has developed two IPVs that have already received market authorization [29]. The Chinese Academy of Medical Sciences (CAMS) in Kuming, China had completed clinical Phase III studies for their IPV. The following PV master seeds were used by CAMS: For type 1 PV, Sabin SO+1; for Type 2, Sabin SO+1; for Type 3, Pfizer RS01.The micro-carrier culture method was utilised to cultivate Vero cells and PV in bioreactors. For their Clinical Phase II, a randomized, positive-controlled trial was conducted in 500 infants that were randomly assigned to 5 groups (A, B, C, D, and E). After 3 doses, the seroconversion rates for types 1, 2 and 3 poliovirus were similar to those of the OPV and IPV control groups. They reported seroconversion rates of 100%, 97.8%, 96.6%, 100%, and 90.1%, respectively, in groups A, B, C, D, and E for the type 1 PV [30].
Interestingly, the National Institute for Biological Standards and Control (NIBSC) are developing the production of thermostable PV Virus-Like Particles (VLP) using recombinant mammalian, yeast, bacterial, baculovirus and plant expression systems in collaboration with several UK laboratories. VLPs are devoid of viral RNA but are produced naturally during PV infections. The external surface of a VLP is indistinguishable from that of a PV particle but several internal amino acid chains are disordered (Figure 1.4). NIBSC have identified several amino acid substitutions that stabilise VLPs without altering antigenicity and these VLPs appear are as thermostable as the current IPV.
Figure 4: The difference between a VLP and virus particle. The external
surface of a VLP is indistinguishable from that of a PV particle but several
internal amino acid chains are disordered [31].
In addition, NIBSC is also trying out an approach to produce genetically-stable OPV strains. The 5’-NTR of the PV genome which contains domain V, forms part of a larger structure that controls initiation of translation. The level of attenuation is determined by its thermodynamic stability. They have modified domain V to make it genetically stable by removing all the U-G base-pairs (Figure 1.5). Any single mutation in base-paired stems would weaken the domain V and generate a more attenuated virus. In order to increase the genetic stability of this attenuated strain, NIBSC researchers have also moved an essential Cis-acting replication element (cre) from the P2 region to a position near the 5’ end to reduce the risk of loss of this part of the genome through recombination (Figure 1.5). In this way, two recombination events would be required to replace domain V which is highly unlikely. As the 3D Polymerase also plays an integral role in attenuation of viruses, NIBSC scientists have collaborated with researchers at the University of California to introduce mutations that would increase the fidelity of the 3D polymerase and decrease its ability to catalyse homologous recombination [31].
Figure 5: Production of genetically-stable OPV strains. All the U-G
base-pairs in domain V was removed to make it more genetically stable and
the Cis-acting replication element (cre) was moved from the P2 region to a
position near the 5’ end to reduce the risk of loss of this part of the genome
through recombination [31].
Conclusion
After eradication of wild type poliomyelitis in industrialised countries, the only source of polio seemed to be derived from cases of VAPP and VDPVs that inevitably could lead to potential outbreaks. Due to a gradual shift of polio vaccination from OPV back to IPV, one of the most anticipated next generation vaccines is an IPV based on the attenuated Sabin poliovirus strains, producing a Sabin IPV (sIPV). The sIPV has good clinical trial data by research groups in USA, Japan and China to support the use of the vaccine. In addition, there have been several studies performed to produce a third generation IPV. This new generation of IPV differs from the sIPV as it is produced from poliovirus strains that have an antigenic structure identical to the wild-type strains but were non-pathogenic by genetic manipulations like modification of the 5' -NTR of the poliovirus genome, mutation of the poliovirus 3D polymerase gene, utilising codon-pair bias de-optimization and microRNA sequence insertion. However, technological advances in the development of the sIPV or 3rd generation IPV will not result in universal global protection against poliomyelitis unless they are supported by a sustainable public health infrastructure for vaccine distribution, coverage and use. There should also be mobilization of efforts from both the public and private organizations in order to achieve success of the polio eradication initiative.
References
- Thomassen YE, van ’t Oever AG, van Oijen MGCT, Wijffels RH, van der Pol LA. Next generation inactivated polio vaccine manufacturing to support post polio-eradication biosafety goals. PLoS One. 2013; 8: 83374.
- Offit PA. The Cutter incident: How America's first polio vaccine led to the growing vaccine crisis. New Haven, CT. Yale University Press. 2005.
- Melnick JL, JC. Brennan. Monkey neurovirulence of attenuated poliovirus vaccines being used in field trials in live poliovirus vaccines. First International Conference on Live Poliovirus Vaccines. Pan American Sanitary Bureau, Washington, D.C. 1959; 44: 65-100.
- Sutter RW, Kew OM, Cochi SL, Aylward RB. Poliovirus vaccine-live. Plotkin SA, Orenstein WA, Offit PA, editors. In: Vaccines, 6th edition. Elsevier Sanders. 2008; 598-645.
- WHO Meeting of the Strategic Advisory Group of Experts on immunization. Weekly Epidemiological Record. 2011; 86: 205-220.
- WHO Sabin-IPV Development in IMB, CAMS, China. 2011.
- Van Wezel AL. Growth of cell-strains and primary cells on micro carriers in homogeneous culture. Nature. 1967; 216: 64-65.
- Kew OM, Sutter RW, de Gourville EM, Dowdle WR, PM. Vaccine-derived polioviruses and the endgame strategy for global polio eradication. Annu Rev Microbiol. 2005; 59: 587–635.
- Nomoto A, Omata T, Toyoda H, Kuge S, Horie H, Kataoka Y, et al. Complete nucleotide sequence of the attenuated poliovirus Sabin 1 strain genome. Proc Natl Acad Sci USA. 1982; 79: 5793–5797.
- Kawamura N, Kohara M, Abe S, Komatsu T, Tago K, Arita M, et al. Determinants in the 5’ noncoding region of poliovirus Sabin 1 RNA that influence the attenuation phenotype. J Virol. 1989; 63: 1302–1309.
- Bouchard MJ, Lam DH, Racaniello VR. Determinants of attenuation and temperature sensitivity in the type 1 poliovirus Sabin vaccine. J Virol. 1995; 69: 4972–4978.
- Paul AV, Mugavero J, Yin J, Hobson S, Schultz S, van Boom JH, et al. Studies on the attenuation phenotype of polio vaccines: poliovirus RNA polymerase derived from Sabin type 1 sequence is temperature sensitive in the uridylylation of VPg. Virology. 2000; 272: 72–84.
- Westrop GD, Wareham KA, Evans DM, Dunn G, Minor PD, Magrath DI, et al. Genetic basis of attenuation of the Sabin type 3 oral poliovirus vaccine. J Virol. 1989; 63: 1338–1344.
- WHO Information Sheet on observed rate of vaccine reactions: Polio vaccines. Global Vaccine Safety Essential Medicines & Health Products. 2014.
- WHO Background and Technical Rationale for Introduction of one dose of Inactivated Polio Vaccine (IPV) in Routine Immunization Schedule. 2014.
- Chumakov K, Ehrenfeld E. New generation of inactivated poliovirus vaccines for universal immunization after eradication of poliomyelitis. Vaccine. 2008; 47: 1587-1592.
- Bakker WA, Thomassen YE, van't Oever AG, Westdijk J, van Oijen MG. Inactivated polio vaccine development for technology transfer using attenuated Sabin poliovirus strains to shift from Salk-IPV to Sabin-IPV. Vaccine. 2011; 29: 7188-7196.
- WHO. WHO global action plan to minimize poliovirus facility associated risk after eradication of wild polioviruses and cessation of routine OPV use? 2009.
- Ehrenfeld E, Modlin J, Chumakov K. Future of polio vaccines. Expert Rev Vaccines. 2009; 8: 899-905.
- WHO. WHO Technical Working Group Meeting on Revision of the WHO Recommendations for the Production and Control of Inactivated Poliomyelitis Vaccines: TRS No. 910, Annex 1. Geneva, Switzerland. 2013; 14-15.
- WHO. Recommendations for the production and control of poliomyelitis vaccine (Inactivated). Technical Report Series No. 910 World Health Organization. 2002.
- Agol VI, Pilipenko EV, Slobodskaya OR. Modification of translational control elements as a new approach to design of attenuated picornavirus strains. J. Biotechnol. 1996; 44: 119-128.
- Toyoda Hidemi, Yin Jiang, Mueller Steffen, Wimmer Eckard, Cello Jeronimo. Oncolytic treatment and cure of neuroblastoma by a novel attenuated poliovirus in a novel poliovirus-susceptible animal model. Cancer Res. 2007; 67: 2857-2864.
- MacadamAJ, Ferguson G, Stone DM, Meredith J, Knowlson S, Auda G, et al. Rational design of genetically stable, live-attenuated poliovirus vaccines of all three serotypes: relevance to poliomyelitis eradication. J Virol. 2006; 80: 8653-8663.
- Pfeiffer Julie K, Kirkegaard Karla. A single mutation in poliovirus RNA-dependent RNA polymerase confers resistance to mutagenic nucleotide analogs via increased fidelity. Proc Natl Acad Sci USA. 2003; 12: 7289–7294.
- Coleman JR, Papamichail D, Skiena S, Futcher B, Wimmer E, Mueller S. Virus attenuation by genome-scale changes in codon pair bias. Science. 2008; 320: 1784-1787.
- Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004; 116: 281-297.
- Van Rij RP, Andino R. The silent treatment: RNAi as a defense against virus infection in mammals. Trends Biotechnol. 2006; 24: 186-193.
- Tanimoto T, Murashige N, Hosoda M, Kusumi E, Ono S, Kami M, et al. Vaccination for whom? Time to reinvigorate Japanese vaccine policy. Lancet. 2012; 380: 1647.
- Liao G, Li R, Li C, Sun M, Li Y, Chu J, et al. Safety and immunogenicity of inactivated poliovirus vaccine made from Sabin strains: A phase II, randomized, positive-controlled trial. J Infect Dis. 2012; 205: 237-243.
- National Institute for Biological Standards and Control. New Polio Vaccines. 2015.