
Review Article
Austin J Nanomed Nanotechnol. 2015; 3(1): 1039.
An Advanced Approach of NFID - Nanopatch Technology
Pallavi B, Thofeeq MD* and Venkat Reddy B.Ch*
Department of Biotechnology, Jawaharlal Nehru Technological University, India
*Corresponding authors: Thofeeq MD, Department of Biotechnology, Sri Indu Engineering College, Jawaharlal Nehru Technological University, Hyderabad
Venkat Reddy B.Ch, Department of Biotechnology, Sri Indu Engineering College, Jawaharlal Nehru Technological University, Hyderabad, India
Received: May 05, 2015; Accepted: August 05, 2015; Published: August 10, 2015
Abstract
Needle-free injection systems are novel ways to introduce various medicines into patients without piercing the skin with a conventional needle. Needle-free technology offers the very obvious benefit of reducing patient concern about the use of needle. According to Food and drug administration [FDA]: A needle-less or needle free injection is a device used for the parenteral administration of a medicament is disclosed. They can take in the form of power sprays, edible products, inhalers, and skin patches. Needle-free systems are designed to solve these problems making them safer, less expensive, and more convenient. The purpose of current article is to enlighten the advantages and uses of drug delivery using Nanopatch technology. This technology is used to deliver drugs, vaccines, etc. Today, Nanopatch is a developing technology that promises to make the administration of medicine more efficient and there is the obvious attraction that being needle-free, a patch is painless, and does not run the risk of needle-stick injuries, can use easily of its small size, temperature stability also introduces the option of distributing vaccines to parts of the world where “cold chain” infrastructure is unreliable or non-existent.
Keywords: Nanopatch Technology; NFID; Needle-free
Introduction
Approximately 95% of people who die from infectious diseases reside in resource-limited areas [1]. Now a days we can treat or prevent many diseases using different vaccines available in market. Effective and widespread distribution of vaccines in these areas is limited by breakdowns in refrigeration (‘cold chain’), lack of trained personnel for safe vaccine administration, needlestick injuries resulting from cross-contamination or needle sharing, and the phobia associated with needle pain. Indeed, even in developed countries, so many people have needle phobia because of that people avoid taking medicaments, studies shows that in developing countries almost 10% of people of total population are afraid of injections and needles. While development of new and improved vaccines will continue into the future for a range of diseases, the only way to meet the resource challenges worldwide is to engineer better ways of administering vaccines in a simple and effective way to more people than is currently the case [2].
The hypodermic syringes were first developed by French surgeon, Charles Gabriel pravaz, in 1853s and since then, intramuscular injection of vaccines has been the most popular delivery method due to its ease of administration [3]. However, this technology has several disadvantages. Firstly, muscle contains relatively few APCs, which are important in generating protective immune responses. Secondly, the stability of vaccine formulations is heavily dependent upon environmental factors such as temperature and humidity, with costs in maintaining the cold chain potentially sufficient to vaccinate a further 10 million children if refrigeration were not required [4].
Finally, needle-based vaccine delivery in low-resource regions requires qualified medical practitioners to reduce needlestick injuries, unsafe injections and biological waste exposure [5]. Clearly, there is a pressing need for new vaccine delivery tools that are needlefree, pain-free, simple and inexpensive to distribute and use, with improved efficacy or reduced dose per person, in order to vaccinate more people per less cost.
To overcome all these problems Needle-free injection devices (NFID) have been available for humans since the 1930s. According to the International Organization for Standardization (ISO) needle-free injection is defined as the injection of medicinal products through the skin by pressure without penetrating the skin with a needle [ISO/ TC 84/WG 4 2004] [6]. Even though some people use needle syringe injections due to its less cost and lack of awareness about needle free injection systems. People are given injections to protect them from different infectious diseases. When a needle is inserted through the skin, the vaccine (or drug) it carries provides systemic immunity. This is because the vaccine gets into the bloodstream and provokes the body to create antibodies that are carried throughout the entire body. Nanopatch is the advanced Needle-free injection system, and novel way to introduce various medicines into patients without piercing the skin with a conventional needle [7].
Background
A scientific study shows that, Over 13 million people die from infectious diseases every year [1]. Whilst public and private research initiatives continue to develop novel vaccines for many diseases [8], the issue of how best to formulate, package, distribute and administer these vaccines across the world remains a significant unsolved problem. Most vaccines have been delivered by the needle and syringe, however this technology has several important disadvantages including needlestick injuries, disease transmission through needle reuse [5], limited thermostability, the need for training/ expertise for administration, lack of targeting to immune rich regions of the body, and the issues of pain/phobia that result in avoidance of medical care in ~10% of the population. Micro Projection Arrays (MPAs) can overcome some of these challenges and have thus far shown promising results in terms of immunogenicity and protection both in preclinical and Phase 1 human trials. As described below, recently extended the field with an ultra-high density projection array- the Nanopatch – to deliver vaccine into the epidermis and dermis, the skin layers rich in antigen presenting cells (APCs). Key advantages of its design result in improved immune responses (with 100th of the dose required by the needle and syringe into muscle) and greatly improved thermostability in comparison to needle/ syringe delivery, enabling better suitability for applications in developing countries and broad applicability across a range of different vaccine types.
Skin as an Alter Native Target Organ for Vaccine Delivery
Structure of human skin
The skin is the largest organ in the human body and the first line of defence against most opportunistic pathogens. Skin is essential for successful administration of drugs through needle free injection systems as these drugs are administered underneath the skin [9]. It has a thickness of approximately 2000 – 3000 μm. In addition to serving as a mechanical barrier to pathogen entry, the skin is also a potent immune organ [10] (Figure 1).
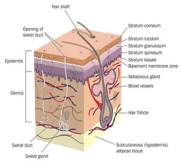
Figure 1: Structure and layers of Skin.
Epidermis: It is the outermost layer of the skin. It forms the waterproof, protective wrap over the body’s surface and is made up of stratified squamous epithelium with an underlying basal lamina. The epidermis contains no blood vessels, and cells in the deepest layers are nourished by diffusion from blood capillaries extending to the upper layers of the dermis. The main type of cells which make up the epidermis are Merkel cells, keratinocytes, with melanocytes and Langerhans cells also present. The epidermis can be further subdivided into the following strata (beginning with the outermost layer): corneum, lucidum (only in palms of hands and bottoms of feet), granulosum, spinosum, basale.
Dermis: The dermis is the layer of skin beneath the epidermis that consists of connective tissue and cushions the body from stress and strain. The dermis is tightly connected to the epidermis by a basement membrane. It also harbors many Mechanoreceptors (nerve endings) that provide the sense of touch and heat. It contains the hair follicles, sweat glands, sebaceous glands, apocrine glands, lymphatic vessels and blood vessels. The blood vessels in the dermis provide nourishment and waste removal from its own cells as well as from the Stratum basale of the epidermis.
Hypodermis: The hypodermis is not part of the skin, and lies below the dermis. Its purpose is to attach the skin to underlying bone and muscle as well as supplying it with blood vessels and nerves. It consists of loose connective tissue and elastin. The main cell types are fibroblasts, macrophages and adipocytes (the hypodermis contains 50% of body fat). Fat serves as padding and insulation for the body. Another name for the hypodermis is the subcutaneous tissue.
The combination of physical barrier and potent immune responses provides the basis of the front-line defence functions of the skin. Both of these aspects also present a unique challenge for vaccination: the need for shallow and precise targeting of vaccine antigens to the APC-rich regions while accounting for the complex mechanical properties of the individual skin layers.
Physical Methods for Targeting Vaccine to the Skin
There are many emerging approaches to deliver vaccines through the skin, taking advantage of the abundance of resident APC populations. Passive methods like topical application of creams/ lotions are effective for small, lipophilic compounds that traverse the SC. However, delivery of vaccines (typically » 0.1 MDa) is ineffective via this route [11]. Intradermal injections are known to elicit improved antibody responses in comparison to the intramuscular route. Difficult to administer accurately, even for highly trained clinicians, this is not in widespread use in vaccine administration [12]. Needlefree techniques include liquid- or gas-powered jet injectors that ballistically deliver vaccine (either in liquid or solid particle form) at > 100m/s into the skin, achieving comparable immune responses (and in some cases, improved responses) to the intramuscular route [13]. While these techniques have taken important steps in using the skin as a site for vaccination, issues including pain, targeting variability due to different skin type and other complexities remain as challenges.
Nanopatch: Origin and Methodology
While early results in pre-clinical and clinical trials investigating MPA technology are encouraging, key challenges of achieving significant dose reduction (leading to significant improvements in vaccine distribution and availability) and significantly improved thermostability remain. Design of the Nanopatch (Figure 2), an ultrahigh density MPA with dry-coated vaccine, to target the dendritic cell populations within the epithelia with the aim to meet this need. Nanopatches indeed targeted ~50% of available APCs (both in the viable epidermis and dermis), requiring less than 100th of the standard intramuscular dose required for equivalent protective immune responses using an influenza-based mouse model [14]. We then sought to explore broader applications of the Nanopatch by investigating a range of different vaccines, developing novel coating technologies and formulations to support long-term thermostability (Figure 2,3).

Figure 2: NANOPATCH – Design.
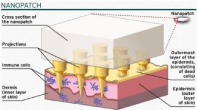
Figure 3: NANOPATCH - Mechanism of Working.
Nanopatch Technology
The Nanopatch approach consists of an array of thousands of vaccine-coated microprojections that perforate into the outer layers of the skin when applied with an applicator device [15]. The tips of Nanopatch’s microprojections are coated with a vaccine material and release this material directly to the large numbers of key immune cells which are immediately below the skin surface. The nanopatch is created through deep reactive ion etching [16]. The central element of this technology is the Nanopatch array itself which consists of a 1 cm2 square of silicon with ~20,000 microprojections on its surface - invisible to the naked eye (Figure 4). The Nanopatch array penetrates through the protective outer skin layer (stratum corneum) and targets immune-activating material to the immune-cell rich layers just beneath the outermost skin layer utilising the microprojections with optimised spacing and length. The result, demonstrated in a mouse model, is an effective increase in immunogenicity, which can be leveraged for two different purposes: either reducing the dose required to achieve efficacy (100-fold reduction has been achieved in the mouse model when delivering Fluvax®), and for amplifying the vaccine efficacy. Pre-clinical experiments have also shown the ability of the Nanopatch to remove or significantly reduce the amount of adjuvant required for effective vaccination.

Figure 4: The Nanopatch, a needle-free vaccine delivery system, magnified
under scanning electron microscopy at A. 200-fold, B. 500-fold and C. 1200-
fold magnification. These images show the individual projections of the
Nanopatch, arranged in an ultra-high density array (>20,000 projections/cm2),
with high aspect ratio and tips with sub-μm sharpness [15].
Traditionally, microneedle delivery systems have been held back from commercialisation due in part to challenges in manufacture scaling. Even in early research programs within Professor Kendall’s laboratory, this consideration was always at the forefront of new process developments. At Vaxxas today personnel are confident that we have a technology which is inherently simple and feasible for high throughput, cost effective manufacture.
Formulation and coating
To ensure that the Nanopatch device delivers an effective volume of vaccine to the desired targets, it is essential that microprojection arrays are not only fabricated to the correct geometry, but also coated uniformly and consistently. The manufactured patches are coated with a formulation containing the vaccine. Coating methods have been optimised to be scalable and efficiently apply coating formulations rapidly with minimal wastage. The coatings are stable at ambient temperature (avoiding refrigeration requirements) and strong so they remain intact when inserted through the outer skin layer, but release quickly (often seconds) once in contact with moisture within the skin.
Applicator
The outer layer of the skin varies its properties considerably depending on age, gender, health and even environmental humidity. This introduces variability which must be overcome to achieve consistent and repeatable vaccine delivery. The Nanopatch approach achieves this by combining the coated Nanopatch arrays with a carefully considered applicator. The applicator addresses variations in the skin by exploiting our knowledge of the skin’s mechanical properties to achieve uniform penetration and delivery across the natural variation in a patient population. The applicator design overcomes the inconsistency that plagues other transdermal vaccine delivery approaches (Figure 5).
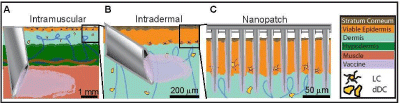
Figure 5: Comparison between three key modes of vaccine delivery via the
skin.
A. Intramuscular injection, in which payload is delivered intramuscularly
below the skin’s epidermis/dermis.
B. Intradermal injection, in which payload is delivered into the dermal cavity
via the Mantoux method.
C. Nanopatch, which rapidly releases dry-coated vaccine payload in the
vicinity of APCs in the epidermis/dermis.
LC: Langerhans cells; dDC: Dermal dendritic cells; APC: Antigen-presenting
cell.
The Nanopatch technology has been used to deliver a range of different vaccines, including
• inactivated whole virus vaccines (e.g., FluVax® – commercially available seasonal influenza vaccine [14]),
• virus-like particles (e.g., Gardasil® – commercially available tetravalent human papilloma virus vaccine [17]),
• DNA plasmids (e.g., preclinical herpes simplex virus 2 vaccine [18]) and other compositions. Long-term thermostability of our dry-coated Nanopatch vaccines, showing comparative immunogenicity with freshly coated devices or those coated and stored for over six months at 23oC prior to skin application [19].
Nanopatch Benefits
The inherent features of the Nanopatch delivery technology provide some key benefits that include [20]:
Improved immunogenicity
By direct delivery of the vaccine to key immune cells, the Nanopatch can potentially either enhance the immune response generated by a vaccine, or allow the generation of an effective immune response with fraction of a full vaccine dose. Indeed, the Nanopatch has been shown in preclinical studies to result in a protective immunogenic response using as little as one hundredth of the dose required by conventional needle and syringe.
No cold chain
The coating formulations used to coat the patches can provide for ambient temperature stability of the vaccine. As a result, vaccine distribution would not need to rely on costly cold distribution chain that is otherwise required to prevent temperature damage which can render conventional vaccines ineffective or potentially harmful. Temperature stability also introduces the option of distributing vaccines to parts of the world where cold chain infrastructure is unreliable or non-existent.
Needle free
The array of Nanopatch microprojections rely on the use of an applicator to allow them to penetrate through the protective outer layer of the skin to deliver a vaccine [21]. This contrasts the traditional needle and syringe where needle stick injuries are common and can lead to serious consequences due to the transmission of infectious diseases as a result. The Nanopatch projections are invisible to the naked eye and therefore are not anticipated to cause distress to people that dislike needles (around 10% of the population are considered to have a phobia against needles); this is expected to help improve patient compliance.
Pain free
The microprojection array of the Nanopatch has been designed to deliver vaccine directly to the key immune cells just below the skin surface. These projections do not reach a depth where they meet nerve endings, and so the Nanopatch delivery device is anticipated to be pain free.
Cost effective
The Nanopatch vaccine delivery system is being developed with high volume, low cost manufacture in mind, using well established manufacturing techniques. As a result of some of the other benefits listed above, there may be further cost savings due to using less vaccine to achieve an effective immunisation, elimination of cold chain costs and a significant reduction in the costs associated with needle stick injuries.
With its strong and unique benefits, we envisage this platform technology may be suitable for delivering a vast majority of vaccines.
Conclusion
Novel Applications for the Future Delivery of drugs to the skin is only one potential application of Nanopatch technology. The collection and processing of whole blood prior to immunoassays contributes to the high cost, complexity, long turnaround time and expertise required to perform diagnostic tests for diseases. Rapid screening in infectious diseases is a key concern, and in the future, rapid screening to aid personalized treatment of complex diseases (e.g., cancer, diabetes, and cardiovascular disease) is likely to benefit from advances in this area. The skin is highly vascularized, with a blood vessel density ranging from 20–40 vessels/mm2 at a depth of 58-65 μm [10]. New technologies are urgently required to overcome challenges in distributing vaccines to those who need them, especially in the developing world. Needle-free vaccination via the skin offers key advantages in comparison to standard needle and syringe methods. These include targeting of APCs in the skin dermis and epidermis and enhanced thermostability through dry coating technology and reduced doses per application, enabling more people to be vaccinated. The Nanopatch has shown ~100-fold dose reduction for vaccinations in comparison to standard intramuscular injections within less cost. The technology is the size of a postage stamp, and has thousands of small projections designed to deliver the vaccine to abundant immune cells in the skin. “It is designed for thermostability, making it cheaper to produce, more convenient to transport because of its small size and easier to access in developing countries [22].”
References
- Mabey D, Peeling RW, Ustianowski A, Perkins MD. Diagnostics for the developing world. Nat Rev Microbiol. 2004; 2: 231-240.
- Hickling JK, Jones KR, Friede M, Zehrung D, Chen D, Kristensen D. Intradermal delivery of vaccines: potential benefits and current challenges. Bull World Health Organ. 2011; 89: 221-226.
- Mitragotri S. Immunization without needles. Nat Rev Immunol. 2005; 5: 905-916.
- Giudice EL, Campbell JD. Needle-free vaccine delivery. Adv Drug Deliv Rev. 2006; 58: 68-89.
- Ekwueme DU, Weniger BG, Chen RT. Model-based estimates of risks of disease transmission and economic costs of seven injection devices in sub-Saharan Africa. Bull World Health Organ. 2002; 80: 859-870.
- ISO/TC 84/WG 4 2004.
- Houser TA, Sebranek JG, Thacker BJ, Baas TJ, Nilubol D, Thacker EL, et al. Effectiveness of transdermal, needle-free injections for reducing pork carcass defects. Meat Sci. 2004; 68: 329-332.
- https://www.madehow.com/Volume-6/Needle-free-Injection-System.html.
- Tortora GJ, Grabowski SR. Principles of Anatomy and Physiology. HarperCollins College Publishers. 1993.
- Huzaira M, Rius F, Rajadhyaksha M, Anderson RR, González S. Topographic variations in normal skin, as viewed by in vivo reflectance confocal microscopy. J Invest Dermatol. 2001; 116: 846-852.
- Güere&nTilde;a-Burgue&nTilde;o F, Hall ER, Taylor DN, Cassels FJ, Scott DA, Wolf MK, et al. Safety and immunogenicity of a prototype enterotoxigenic Escherichia coli vaccine administered transcutaneously. Infect Immun. 2002; 70: 1874-1880.
- Lambert PH, Laurent PE. Intradermal vaccine delivery: will new delivery systems transform vaccine administration? Vaccine. 2008; 26: 3197-3208.
- Kis EE, Winter G, Myschik J. Devices for intradermal vaccination. Vaccine. 2012; 30: 523-538.
- Fernando GJ, Chen X, Prow TW, Crichton ML, Fairmaid EJ, Roberts MS, et al. Potent immunity to low doses of influenza vaccine by probabilistic guided micro-targeted skin delivery in a mouse model. PLoS One. 2010; 5: e10266.
- https://www.vaxxas.com/nanopatch-technology/overview.
- https://www.engineering.com/ArticleID/6961/The-Nanopatch-Delivers-Vaccines-without-Needles--A-Moonshot-Project.aspx.
- Corbett HJ, Fernando GJ, Chen X, Frazer IH, Kendall MA. Skin vaccination against cervical cancer associated human papillomavirus with a novel micro-projection array in a mouse model. PLoS One. 2010; 5: e13460.
- Chen X, Kask AS, Crichton ML, McNeilly C, Yukiko S, Dong L, et al. Improved DNA vaccination by skin-targeted delivery using dry-coated densely-packed microprojection arrays. J Control Release. 2010; 148: 327-333.
- Chen X, Fernando GJ, Crichton ML, Flaim C, Yukiko SR, Fairmaid EJ, et al. Improving the reach of vaccines to low-resource regions, with a needle-free vaccine delivery device and long-term thermostabilization. J Control Release. 2011; 152: 349-355.
- https://www.vaxxas.com/nanopatch-technology/benefits.
- Rakesh Tiwle, Sanghi DK. An update: on needle free injections. JPCBS. 2014; 4: 129-138.
- https://www.uq.edu.au/news/article/2015/02/nanopatch-vaccine-technology-company-attracts-25-million.