
Case Report
Austin J Otolaryngol. 2015; 2(9): 1064.
Uncommon Sinonasal Pathologies with Common Clinical Presentations
Stephen MCS¹*, Raman EV¹ and Mishra S²
¹Department of ENT and Head & Neck Surgery, Manipal Hospitals, India
²Department of Pathology, Manipal Hospitals, India
*Corresponding author: Stephen MCS, Department of ENT and Head & Neck Surgery, Manipal Hospitals, Bengaluru, No: E906, Alpine Eco Apartments, Doddenakundi, Marathahalli, Bengaluru–560037, India
Received: December 02, 2015; Accepted: December 21, 2015; Published: December 24, 2015
Abstract
Sinonasal neoplasms form a diverse group in the routine clinical otolaryngology practice. The uncommon pathologies in guise of common clinical presentations require meticulous diagnostic evaluation and timely intervention. Hemato/lymphoid neoplasms are infrequent in the maxillary sinus and mimic common infective and inflammatory conditions. Here, we report two patients who presented with usual symptoms and imaging findings, histopathology following surgery revealed: plasmablastic lymphoma and plasmacytoma. We report these cases for their rarity, clinical dilemma, atypical presentation and better quality of life with adjuvant therapy.
Keywords: Maxillary; Sinonasal; Lymphoma; Plasmacytoma; Plasmblastic lymphoma
Abbreviations
DLBCL: Diffuse Large B-cell Lymphoma; ISH: In-situ Hybridization; EBER: Epstein-Barr Virus Related Expression RNA; NHL: Non-Hodgkins Lymphoma; LCA: Leukocyte Common Antigen; CD: Cluster Differentiation; MUM: Multiple Myeloma oncogene; ALK: Anaplastic Lymphoma Kinase; EPOCH: Etoposide/ Prednisolone/Vincristine/Cyclophosphamide/Doxorubicin; PBL: Plasmablastic Lymphoma; CHOP: Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone; CODOX-M/IVAC: Cyclophosphamide, Vincristine, Doxorubicin, High-dose Methotrexate/Ifosfamide, Etoposide, High-dose Cytarabine; DA-EPOCH: Dose-adjusted Etoposide, Prednisone, Vincristine, Cyclophosphamide, and Doxorubicin; MM: Multiple Myeloma; EMP: Solitary Extramedullary Plasmacytoma; EBV: Epstein Barr Virus; HHV: Human Herpes Virus
Introduction
With a spectrum of disease pattern common to maxillary sinus, neoplasms deserve a special mention. Early diagnosis and timely intervention cannot be over emphasized. The symptom presentation depends largely on the volume of the tumor mass and site of involvement.
Sinonasal neoplasms are rare with an annual incidence of ~1 per 100,000 cases worldwide [1]. They account for 0.2-0.8% of all malignant neoplasms and 3% of all head and neck neoplasms [2]. Maxillary sinus houses 77% of all paranasal sinus malignant tumors [2]. Tumors of epithelial cell origin are the commonest with 80% being squamous cell and adenocarcinoma [1].
However, hemato/lymphoid neoplasms of maxillary sinus are an important concern as treatment options and their natural clinical course vary. The etiology of these neoplasms unlike the epithelial tumors remains largely unknown. The most common lymphoid neoplasm in Asians are DLBCL (29%) and plasma cell neoplasms (17%) with a male predominance [3,4]. However, their presentation may cause a clinical dilemma.
Here, we report 2 patients with common clinical presentation and diagnosis was revealed only on surgical histopathology that directed further adjuvant treatment.
Case 1
A 57 year old female presented to the ENT outpatient with complaints of first episode unilateral epistaxis. There were no other significant positive histories. Clinical examination showed right deviated nasal septum. X-ray paranasal sinuses was suggestive of acute sinusitis with fluid level in the right maxillary sinus (Figure 1). Following a week of appropriate antibiotics, she continued to have self-limiting intermittent episodes of epistaxis and clinical findings were non-contributory.
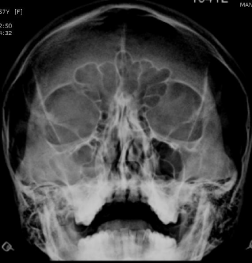
Figure 1: Plain x-ray paranasal sinus – clinically correlated to acute sinusitis.
Plain CT paranasal sinuses revealed soft tissue density in the right maxillary sinus, obliterating the right osteomeatal unit (Figure 2). Findings were opined as suggestive of sinusitis. As her symptoms did not resolve and she had developed dull aching right facial pain, she was planned for endoscopic sinus surgery.
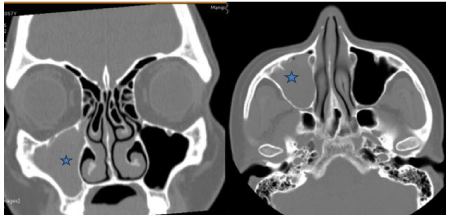
Figure 2: Non contrast enhanced CT paranasal sinuses coronal and axial
sections showing right maxillary sinus opacity (*) obliterating the right
osteomeatal unit.
Intraoperatively, she was found to have a fleshy, friable, vascular mass in the right maxillary sinus. The histopathology revealed Non- Hodgkin’s lymphoma, plasmablastic type and immunohistochemistry showed tumor cells diffusely positive for CD-45 (LCA), CD138, MUM- 1 and negative for CD 20, pan-ck, synaptophysin, chromogranin, desmin, CD-99, CD 3, CD5, CD10, ALK 1, K & λ. ISH for EBER was negative Ki-67 index >90% suggestive of high cellular proliferation.
She was diagnosed with right maxillary plasmablastic lymphoma (Figure 3). Further systemic work-up with bone marrow aspiration/ biopsy revealed norm cellular marrow (40%) with tri-lineage hematopoiesis and no morphologic evidence of NHL. Her peripheral blood parameters were within normal limits. Blood borne viral screen was negative.
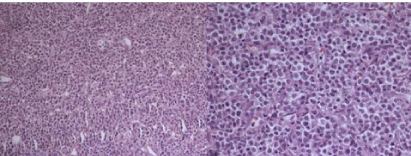
Figure 3: Histopathology sections showing sheets of malignant round
cells arranged in irregular masses divided by thin walled vascular strands.
Individual cells have small round to oval nuclei, many with nuclear molding.
Nucleus is vesicular with small nucleoli.
Whole body PET-CT showed residual disease in the right maxillary sinus extending into the right ethmoid sinus. In view of this, she was planned for EPOCH based chemotherapy for 6 cycles.
Following completion of 3 cycles of chemotherapy, contrast enhanced CT paranasal sinuses showed features of radiological remission (Figure 4). She tolerated the chemotherapy well and had a good quality of life. She was taking adjuvant radiotherapy with 30.6Gy over 3.5 weeks in 17 fractions, at the time of this write-up.
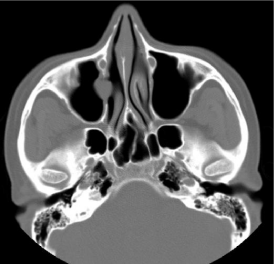
Figure 4: Disease remission in right maxillary sinus following 3 cycles of
chemotherapy.
Case 2
A 50 year old male presented to the Otolaryngology outpatient with complaints of left nasal obstruction, nasal discharge associated with moderate self-limiting epistaxis for 2 months. Clinical examination revealed a fleshy, pale mass in the left nasal cavity that appeared to arise from the lateral nasal wall causing septal deviation to the right and extended posteriorly. The nasal mucosa looked normal. Rest of his local & systemic examination including ophthalmological examination and cranial nerves did not reveal any positive finding. Clinically, he was suspected to have left antrochoanal polyp.
CT paranasal sinuses showed opacification of the left maxillary sinus with expansion and scalloping of the superior and lateral walls. A soft tissue density was seen extending from the left maxillary sinus, widening the left maxillary ostium, extending into the left nostril, impinging on the left bony nasal septum and protruding slightly into the nasopharynx. However, there was also focal bony thinning of the left inferior orbital wall (Figure 5).
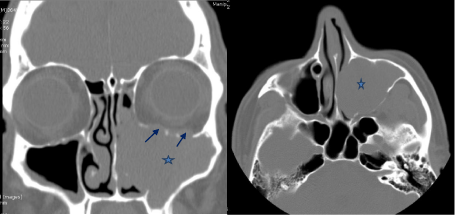
Figure 5: Non contrast enhanced CT paranasal sinuses, coronal and axial
sections showing a soft tissue density in the left maxillary sinus (*) extending
into the nasal cavity and till the nasopharynx, with 2 focal areas of inferior
orbital wall thinning (arrows).
His biochemical parameters were within normal limits. Blood borne viral screen was negative. Sonogram of the abdomen revealed mild enlarged prostate, gall bladder polyp, fatty liver.
Echocardiogram revealed normal cardiac function.
He underwent endoscopic sinus surgery and debridement of the mass. Intraoperatively, he was found to have a pale, fleshy, speckled, friable mass in the left nasal cavity, arising from the left maxillary sinus with focal areas of bony thinning over the inferior orbital margin, although there was no orbital involvement.
The histopathology revealed plasma cell neoplasm with kappa chain restriction, confirmed by immunohistochemical markers (Figure 6). Ki-67 was >80% suggestive of cellular proliferation. Systemic and skeletal survey along with bone marrow examination ruled out any multifocal or disseminated disease. He was diagnosed to have sinonasal solitary extra-medullary plasmacytoma. Thus he was initiated on radiotherapy at the time of this write-up.
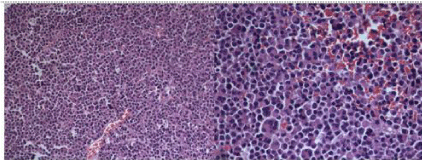
Figure 6: Monomorphic infiltrate of plasma cells with a variable degree of
cellular atypia in a sparse, delicate and reticular stroma. The plasma cells
show a round eccentric nuclei and dense chromatin clumps that are arranged
along the nuclear membrane.
Discussion
While primary sinonasal lymphomas are non-Hodgkin lymphoma arising in the sinonasal region (express B-cell/T-cell phenotype), plasma cell neoplasms are monoclonal plasma cell neoplastic proliferation, producing monoclonal immunoglobulins and present as either disseminated-multiple myeloma (MM) or localized (solitary plasmacytoma)- medullary and extramedullary forms.
Plasmablastic lymphoma (PBL), first reported by Delecluse et al. (1997) is a recently recognized aggressive subtype of non-Hodgkin B-cell lymphoma, now considered a separate diagnostic entity, distinct from DLBCL, not otherwise specified, as per the 2008 WHO classification of lymphoid neoplasms [5]. Predominantly involves the oral cavity of HIV seropositive patients, and exhibits a immuno blastic morphology with plasma cell phenotype [3]. Common extraoral sites include GIT, lymph nodes, skin, omentum, lung, sinonasal region, testes, bones, soft tissue, bone marrow and CNS [5]. With male predominance (4-5:1), majority present in the middle age. (Mean 39 years -HIV positive and 59 years in HIV negative patients) [5]. Dual infection with EBV and HHV8 has been demonstrated in PBL [5,6]. PBL in HIV-negative patients is commonly associated with immunosuppression in the form of steroid therapy or solid organ transplantation. However, HIV-negative immunocompetent patients have been reported [7]. Our patient was a middle aged female, immunocompetent, seronegative for HIV with maxillary sinus involvement.
PBL is typified by early dissemination to a fulminant disease with poor response to therapy causing limited survival and death in the first year after diagnosis (mean survival 3 months).
It remains a therapeutic challenge in view of its peculiar morphology and immunohistochemistry profile. The minimum morphological criteria required to diagnose include (a) predominant Monomorphic cellular proliferation of plasmablasts with either centrally or eccentrically placed nuclei, high nuclear-cytoplasmic ratio and a moderate amount of eosinophilic cytoplasm; (b) high mitotic and/or apoptotic index & (c) absence of neoplastic plasma cells in the background. Further, essential diagnostic criteria on immunophenotyping consisted of CD20 negativity, LCA +/-, CD138/ VS38c diffuse positivity, light chain restriction and high MIB-1 index (>60 %) [8]. The treatment options include a combination of chemotherapy, radiotherapy and/ or surgery, however with grave prognosis (60% mortality at 1 year) [6]. There is no standard chemotherapy protocol followed. CHOP, CODOX-M/IVAC or DAEPOCH have been the available options. Lately, the potential value of the proteasome inhibitor bortezomib and thalidomide has been reported.
Our patient obeyed the morphological and immunophenotyping characteristics. Following chemotherapy (EPOCH), she was found to have disease reduction and was further continuing adjuvant radiotherapy, already 6 months following diagnosis. She has so far tolerated well.
Plasmacytoma was first described by Schridde in 1905 [9]. Solitary extra-medullary plasmacytoma (EMP) account for ~3% of all plasma cell neoplasms with a worldwide annual incidence of 3 per 100,000 populations [10]. 75-80% of them originate in the submucosa of the upper aero-digestive tract owing to rich lymphatics, of which 75% occur in the upper respiratory tract (nose, nasopharynx, paranasal sinuses). There is a strong male predilection (male: female ratio 3:1) and commonly occurred during the 5th-7th decade [11].
Patients usually present with localized disease that has an insidious clinical course. Clinical findings may include nasal obstruction and soft-tissue swelling (80%), epistaxis, nasal discharge, pain, proptosis, cervical lymphadenopathy and neurologic deficits observed in decreasing order of frequency [12]. These non-specific clinical findings may mimic other malignancies of the head and neck of epithelial origin. Patients may present with advanced disseminated systemic form, owing to the slow-growing tumor. 15-20% of EMP progress into MM with a poorer prognosis [13]. Our patient presented with non-specific clinical findings, within 2 months of symptoms.
The diagnosis includes a combination of pathological, radiological and clinical features. The diagnostic criteria include: (a) pathological tissue evidence of monoclonal plasma cells involving a single extramedullary site; (b) no bone marrow involvement; (c) negative skeletal survey results; (d) no anemia, hypercalcemia or renal impairment caused by plasma cell dyscrasia; (e) low serum or urinary levels of monoclonal immunoglobulin [9,14]. Our patient obeyed the above diagnostic criteria.
CT and MRI are complementary in assessing the local disease, though are non-specific. The tumor may be locally aggressive with destruction of the adjacent structures [15]. Isointense images on T1- weighted and iso- to hyperintense images on T2-weighted sequences are well demonstrated in MRI assessment. Obtaining biochemical evidences before biopsy is challenging as only 25% of the patients show increased levels of M-protein in their blood or urine [15]. Our patient had similar findings of a maxillary mass that was locally aggressive and his biochemical tests were inconclusive.
Pathological evidence is crucial to diagnosis. Sinonasal EMPs originate in the submucosa and the sinus mucosa may be thickened by inflammation which warrants deep biopsy specimens for frozen section analysis [12]. The gross tumor appear as fleshy, yellowish gray to dark red, sessile, polypoid or pedunculated lesion. Microscopy reveals a florid monomorphic infiltrate of plasma cells with variable cellular atypia in a sparse, delicate and reticular stroma. The plasma cells demonstrate a ‘cart-wheel’ appearance with round eccentric nuclei and dense chromatin clumps arranged along the nuclear membrane. Immunophenotyping with CD15, CD20, CD45RB, CD45RA, epithelial membrane, IgA, IgD, IgG, IgM, IgE, kappa light chain, and lambda light chain antibodies is helpful [12]. The demonstration of a monoclonal pattern of Immunoperoxidase staining for kappa and lambda immunoglobulin light chains provides for definitive diagnosis and thus distinguishes plasmacytomas from reactive plasmacytosis.
The use of gene rearrangement molecular techniques has been advocated to demonstrate the monoclonal pattern and predict progression to MM.
Concurrent multiple myelomas should be ruled out and local recurrence or disseminated disease can recur years after treatment which warrant long term regular follow-up [16].
There has been controversy over the optimal treatment of EMP. The modes of treatment include radiotherapy alone, surgery alone or a combination of both. The tumor is extremely radiosensitive; however, sinonasal EMP with bony destruction should not be treated with surgery alone or radiation alone, rather with a combination of surgical resection with radiotherapy [17]. Combined intervention resulted in a longer overall survival and recurrence free survival. Surgery may suffice in limited, easily resectable disease and nonupper aerodigestive lesions [17].
Conclusion
Besides the common conditions of the maxillary sinus, the uncommon pathologies should be borne in mind. Optimal treatment with multi-disciplinary intervention in the correct set of patient lead to prolonged survival and better quality of life.
Points to Learn
Case 1: Plasmablastic lymphoma, an aggressive variety of the NHL, is usually found in HIV seropositive or other immunocompromised patients with male predominance and show a fulminant natural course with death in first few months of diagnosis. Our patient is a female, middle-aged, immunocompetent, seronegative, with limited disease (maxillary sinus involvement), tolerated chemotherapy well and taking adjuvant radiotherapy, quality of life so far good.
Case 2: Plasma cell neoplasms are uncommon. Extra-medullary solitary sinonasal plasmacytoma is rare. The atypical clinical evolution and aggressiveness of this neoplasm warrant meticulous evaluation of all suspected sinonasal neoplasms for timely intervention.
References
- Llorente JL, López F, Suárez C, Hermsen MA. Sinonasal carcinoma: clinical, pathological, genetic and therapeutic advances. Nat Rev Clin Oncol. 2014; 11: 460-472.
- Suzuki M, Ishikawa H, Kawakami M, Nakahara T, Tanaka A, Mataga I. Establishment and characterization of METON myoepithelioma cell line derived from human palatal myoepithelioma: apical reference to the diverse differentiation potential. Hum Cell. 2013; 26: 170-176.
- Montes-Moreno S, Gonzalez-Medina AR, Rodriguez-Pinilla SM, Maestre L, Sanchez-Verde L, Roncador G, et al. Aggressive large B-cell lymphoma with plasma cell differentiation: immunohistochemical characterization of plasmablastic lymphoma and diffuse large B-cell lymphoma with partial plasmablastic phenotype. Haematologica. 2010; 95: 1342-1349.
- Gaur S, Padilla O, Nahleh Z, Gaur S, Padilla O, Nahleh Z. Clinical Features and Prognosis of CD20 Negative Aggressive B-Cell Non-Hodgkins Lymphoma, Clinical Features and Prognosis of CD20 Negative Aggressive B-Cell Non-Hodgkins Lymphoma. Lymphoma Lymphoma. 2013; e290585.
- Elyamany G, Al Mussaed E, Alzahrani AM. Plasmablastic Lymphoma: A Review of Current Knowledge and Future Directions. Adv Hematol. 2015; 2015: 315289.
- Rafaniello Raviele P, Pruneri G, Maiorano E. Plasmablastic lymphoma: a review. Oral Dis. 2009; 15: 38-45.
- Kim JE, Kim YA, Kim WY, Kim CW, Ko YH, Lee GK, et al. Human immunodeficiency virus-negative plasmablastic lymphoma in Korea. Leuk Lymphoma. 2009; 50: 582-587.
- Kane S, Khurana A, Parulkar G, Shet T, Prabhash K, Nair R, et al. Minimum diagnostic criteria for plasmablastic lymphoma of oral/sinonasal region encountered in a tertiary cancer hospital of a developing country. J Oral Pathol Med. 2009; 38: 138–144.
- Soutar R, Lucraft H, Jackson G, Reece A, Bird J, Low E, et al. Guidelines on the diagnosis and management of solitary plasmacytoma of bone and solitary extramedullary plasmacytoma. Br J Haematol. 2004; 124: 717–726.
- Grover N, Chary G, Makhija P, Rout P. Extramedullary plasmacytoma of the nasal cavity: treatment perspective in a developing nation. Ear Nose Throat J. 2006; 85: 434-436.
- Patel TD, Vázquez A, Choudhary MM, Kam D, Baredes S, Eloy JA. Sinonasal extramedullary plasmacytoma: a population-based incidence and survival analysis. Int Forum Allergy Rhinol. 2015; 5: 862–869.
- Verim A, Sheidaii S, Bilaç O, Tepe Karaca C, Naiboglu B. Extramedullary plasmacytoma of the frontal sinus: case report and Turkish literature review. Turk J Haematol. 2014; 31: 301-306.
- Du RC, Li HN, Huang W, Tian XY, Li Z. Unusual coexistence of extramedullary plasmacytoma and nasopharyngeal carcinoma in nasopharynx. Diagn Pathol. 2015; 10: 170.
- Vincent Rajkumar S. Multiple myeloma: 2014 Update on diagnosis, risk-stratification, and management. Am J Hematol. 2014; 89: 999-1009.
- Ching AS, Khoo JB, Chong VF. CT and MR imaging of solitary extramedullary plasmacytoma of the nasal tract. AJNR Am J Neuroradiol. 2002; 23: 1632-1636.
- Chakravarti A, Vishwakarma SK, Arora VK. Plasmacytoma of the maxilla. Indian J Otolaryngol Head Neck Surg. 1997; 49: 44-46.
- Alexiou C, Kau RJ, Dietzfelbinger H, Kremer M, Spiess JC, Schratzenstaller B, et al. Extramedullary plasmacytoma: tumor occurrence and therapeutic concepts. Cancer. 1999; 85: 2305-2314.