Abstract
The Hereditary Leiomyomatosis and Renal Cell Cancer (HLRCC) is a rare genodermatosis caused by heterozygous mutations in the fumarate hydratase gene (FH, Chromosomal Locus 1q42.1). This tumor predisposition syndrome is characterized by young onset of cutaneous leiomyomata (that arise from the erector pilorum muscle), uterine leiomyomas and/or renal cell carcinoma (‘type 2’ papillary renal cancer or collecting duct carcinoma). Other tumors such as uterine and cutaneous leiomyosarcoma, ovarian mucinous cystadenoma, adrenal cortical adenomas, Leydig cell tumors of the testis and benign renal cysts have been described in FH mutation carriers.
Here we report the case of a 29 year old Caucasian girl carrier of HLRCC phenotype confirmed by histological and genetic analysis.
Keywords: Hereditary leiomyomatosis and renal cell cancer; Cutaneous leiomyomatosis; Fumarate hydratase; Familial cancer syndrome
Case Presentation
We describe the case of a 29 years old Caucasian girl who came to our attention for several nodular lesions involving extremities and trunk. Physical dermatological examination showed about 40-50 skin-colored to light brown firm nodular elements distributed mainly over the upper and lower limbs without tendency to confluence; few elements were also visible on the trunk. The patient reported that the lesions are painful especially during winter and in response to pressure. Skin lesions arose during adolescence and gradually increased in number and size.
Clinical features of skin lesions led us to the hypothesis of multiple cutaneous leiomyomas. Therefore, after proper informed consent, we performed an excisional biopsy for diagnostic confirmation. Histology features allowed a diagnosis of piloleiomyoma, an uncommon benign skin neoplasm arising from the smooth muscles of erector pilorum muscle; it presents as skin-colored to reddish-brown dermal papules or nodules, which may range from one to hundreds elements (Figure 1a and 1b). When a solitary single lesion is present, it might represents a sporadic, non familial condition; conversely, in presence of multiple piloleiomyomas the hypothesis of inherited cutaneous leiomyomatosis, (specifically Hereditary Leiomyomatosis and Renal Cell Cancer - HLRCC) must be taken into account [1]. HLRCC is an autosomal-dominant hereditary syndrome caused by heterozygous mutations in the fumarate hydratase gene (FH, Chromosomal Locus 1q42.1). This genodermatosis predisposes to develop multiple, symptomatic piloleiomyomas, uterine fibroids in young women and early onset renal tumors which can progress and metastasize, even if they are small- sized [1-3].
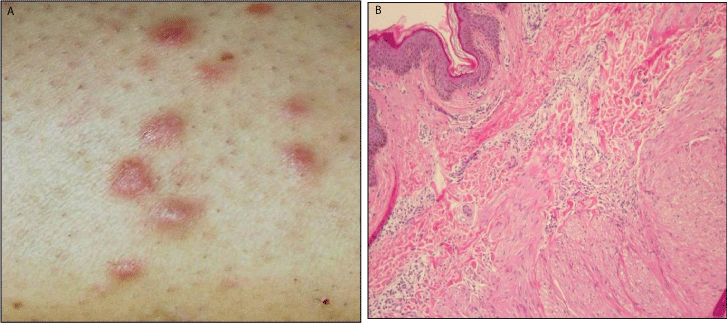
Figure 1: Multiple cutaneous piloleiomyomas of our patient: clinical
appearance. b. Histological examination of piloleiomyoma: a poorly
circumscribed proliferation of haphazardly arranged smooth muscle fibers
located in the dermis. They are usually separated from the epidermis by a
thin grenz zone.
Accordingly, we investigated the personal and family oncologic history of our patient. She also referred a personal history of numerous symptomatic uterine leiomyomas 20 aged onset; due to menorrhagia, hypermenorrhoea and pelvic pain she undergo myomectomy at 25 years old. Moreover, in her family also the father, two brothers and some relatives of both sexes of paternal origin had the same skin lesions (Figure 2). Furthermore, a paternal aunt and her daughter underwent to hysterectomy at 40 and myomectomy at 35 years old, respectively, for symptomatic uterine fibroids (Figure 2).
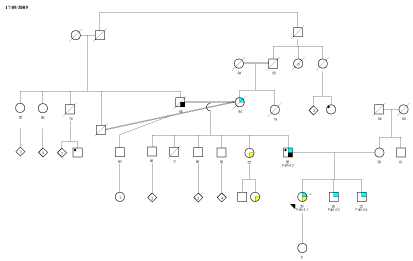
Figure 2: The pedigree of our patient. In light blue are indicate piloleiomyomas, in yellow uterine myomas, in black other malignancies. The double line between
parents indicates consanguineous marriages (between blood relatives), The proband (Fam 4.1) is indicate by an arrow. Her father (Fam 4.2) received a diagnosis
of chronic lymphocytic leukemia at age 52. Her paternal grandfather died for colorectal cancer.
Because of her personal and family history, after genetic counseling, the patient was tested for germline mutation in the FH gene. Direct sequencing on DNA extracted from peripheral blood demonstrated the heterozygous missense mutation c.875_876insAA (p.Val293Argfs*37). The nomenclature describes an insertion of two Adenosine between nucleotides 875 and 876 of the cDNA. This leads a frame shifting change with Valine-293 as the first affected amino acid and the creation of a new reading frame with a premature stop codon 36 amino acids downstream (Figure 3). Afterwards, we demostred the same germline FH mutations in her father and in boths brothers.
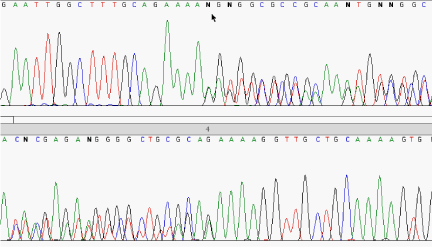
Figure 3: Electropherogram of FH mutation c.875_876insAA (p.Val293Argfs*37).
Discussion
We report a case of an uncommon and frequently misdiagnosed genodermatosis, HLRCC, a Familiar Tumor Syndrome associated with inherited cutaneous leiomyomas [1-3].
Cutaneous leiomyomas are rare benign tumors that arise from smooth muscle of the skin accounting for 75% of extra uterine leiomyomas. On the basis of their histological origin, they are classified as piloleiomyomas, vascular leiomyomas and genital leiomyomas, emerging from erector pilorum muscle, dermal blood vessels and genitals, respectively.
HLRCC is a rare autosomal dominant hereditary syndrome, caused by heterozygous mutations in the fumarate hydratase gene (FH gene, Chromosomal Locus 1q42.1) that encodes an enzyme involved in the tricarboxylic acid cycle which catalyses the conversion of fumarate to malate.
FH is an unusual example of a gene in which germline mutations in one or both copies lead to distinct pathologic phenotypes. In fact, homozygous or compound heterozygous germline mutations in FH cause Fumarate hydratase deficiency (FHD, OMIM 606812), a recessive, inborn error of metabolism characterized by rapidly progressive neurologic impairment but no tumors. Most individuals with FHD survive only a few months; a very few survive to early adulthood [4].
Whereas HLRCC is a Familial Cancer Syndrome that predisposes heterozygous carriers to develop several benign and malignant skin and visceral tumors which often occur as multiple primary cancers and typically onset more frequently and at an earlier age respect the general population. Specifically, HLRCC patients develop piloleiomyomas, early onset multiple uterine leiomyomas and Renal Cell Carcinoma (RCC) [4-7].
The majority of HLRCC individuals (100% in men and 85% in women) are affected by piloleiomyomas. These cutaneous lesions generally present in the second, third or fourth decades and tend to increase in size and number with age. Clinically, they appear as skin-colored to light brown dermal papules or nodules distributed mainly over the trunk and extremities, occasionally on the face. They may occur in different patterns: single, grouped/clustered, segmental, and disseminated. Approximately 40% of FH mutation carriers have mild cutaneous phenotype with five or fewer lesions, although the cutaneous leiomyomas may reach a considerable number, up to hundreds of elements. Individuals note that the skin leiomyomas are sensitive to light touch and/or cold temperature and, less commonly, painful. They are characterized histologically by proliferation of interlacing bundles of smooth muscle fibers with centrally located, long blunt-edged nuclei. They can resemble dermatofibromas, neurofibromas, Schwannomas, cutaneous metastases, angiolipomas and angioleiomyoma [1,4,5,7].
Almost 85% of the affected women develop uterine leiomyomas which onset at a younger age than women in the general population and tend to be numerous and large. Age at diagnosis ranges from 18 to 52 years (median 30 years old). Almost 70% of HLRCC carriers underwent to myomectomy or hysterectomy at or before age 40 years. They experiencing irregular menstruation, pelvic pain, hypermennhoea, menorrhagia and sideropenic anemia.
RCC occur in about 10%-16% of individuals with HLRCC, typically at young age; median age of detection is 44 years, but they have been described also in childhood. These malignancies show an aggressive clinical course, even at a size less than 1 cm and display a characteristic histopathology; they are more frequently classified as ‘type 2’ papillary renal cancer or collecting duct carcinoma. Type 2 papillary renal cell carcinoma consists of large tumor cells with eosinophilic cytoplasm and pseudostratified nuclei. Other types of renal cancers reported in HLRCC patients range from tubulopapillary RCC to collecting-duct RCC [5,7].
FH mutation carriers have also an increased risk of uterine and cutaneous leiomyosarcoma, ovarian mucinous cyst adenoma, adrenal cortical adenomas, Leydig cell tumors of the testis and benign renal cysts [8-10].
Since the high malignancy of certain cancers associated with mutation of FH gene (in particular RCC, uterine and cutaneous leiomyosarcomas) and their tendency to arise into young age (earlier then the general population), early identification of this genetic syndrome through accurate skin examination, is important to lay out adequate cancer screening.
When the FH variant in the family is detected, molecular genetic testing should proposed to at-risk family members in order to allow early surveillance and treatment of the tumors. FH mutation carriers should follow a specific surveillance program which consists of annual dermatologic and gynecologic consultation and early abdominal MRI (starting at 8 years of age). Renal ultrasound is not recommended for routine screening for renal cell carcinoma but it is useful for detection renal cystic lesion [1-5].
References
- Tomlinson IPM, Afrina Alam N, Rowan AJ, Barclay E, Jaeger EEM, Kelsell D, et al. Germline mutations in FH predispose to dominantly inherited uterine fibroids, skin leiomyomata and papillary renal cell cancer. Nature Genetics. 2002; 30: 406-410.
- Alam NA, Olpin S, Leigh IM. Fumarate hydratase mutations and predisposition to cutaneous leiomyomas, uterine leiomyomas and renal cancer. Br J Dermatol. 2005; 153: 11-17.
- Lehtonen HJ, Kiuru M, Ylisaukko-Oja SK, Salovaara R, Herva R, Koivisto PA, et al. Increased risk of cancer in patients with fumarate hydratase germline mutation. J Med Genet. 2006; 43: 523-526.
- Smit DL, Mensenkamp AR, Badeloe S, Breuning MH, Simon ME, van Spaendonck KY, et al. Hereditary leiomyomatosis and renal cell cancer in families referred for fumarate hydratase germline mutation analysis. Clin Genet. 2011; 79: 49-59.
- Gardie B, Remenieras A, Kattygnarath D, Bombled J, Lefèvre S, Perrier- Trudova V, et al. Novel FH mutations in families with hereditary leiomyomatosis and renal cell cancer (HLRCC) and patients with isolated type 2 papillary renal cell carcinoma. J Med Genet. 2011; 48: 226-234.
- Kerrigan JF, Aleck KA, Tarby TJ, Bird CR, Heidenreich RA. Fumaric aciduria: clinical and imaging features. Ann Neurol. 2000; 47: 583-588.
- Menko FH, Maher ER, Schmidt LS, Middelton LA, Aittomäki K, Tomlinson I, et al. Hereditary leiomyomatosis and renal cell cancer (HLRCC): renal cancer risk, surveillance and treatment. Fam Cancer. 2014; 13: 637-644.
- Weiler L, Poulalhon N, Slama A, Guillaud-Bataille M, Thomas L. Isolated cutaneous leiomyosarcoma revealing a novel germline mutation of fumarate hydratase gene. Br J Dermatol. 2015.
- Ristau BT, Kamat SN, Tarin TV. Abnormal Cystic Tumor in a Patient with Hereditary Leiomyomatosis and Renal Cell Cancer Syndrome: Evidence of a Precursor Lesion? Case Rep Urol. 2015; 303872.
- Ylisaukko-oja SK, Cybulski C, Lehtonen R, Kiuru M, Matyjasik J, Szymañska A, et al. Germline fumarate hydratase mutations in patients with ovarian mucinous cystadenoma. Eur J Hum Genet. 2006; 14: 880-883.
