Abstract
Background: Oxidative stress has been described as a well-established causative factor to deteriorate neurons and is associated with ageing and age related neurodegenerative disorders. Vitamin E is a lipophilic natural antioxidant and the generic term for tocopherols and tocotrienols. Vitamin E acts as an antioxidant which play a very important role for normal neurological function as well as in neurodegenerative disease that it is being treated. It is most effective in Alzheimer’s Disease (AD) and has been shown to play a central role in neuroprotection and found to be in effective for neuroprotection in Parkinson’s Disease (PD). Vitamin E is a chain breaking antioxidant which acts as a non-specific protective chemical shield for neurons and protects neurons through antioxidant-independent pathways; they are highly effective and further investigations on their activity might lead to an even more effective application of antioxidants. Reactive oxygen species and free radical generation can lead to neuronal damage associated with AD, PD, amyotrophic lateral sclerosis, Huntington Disease (HD) and cerebral ischemic stroke many signs of oxidative damage. From this review, we would like to highlight the importance of vitamin E in neuroprotection. Conclusion: Since, Vitamin E therapy has not proven promising results in clinical trial; it could suggest that if a person uses vitamin irregularly, as a nutritional supplement, they may have less progression of neurodegenerative disease.
Keywords: Vitamin E; Free radicals; Neurodegenerative disease
Abbreviations
AD: Alzheimer’s Disease; PD: Parkinson’s Disease; HD: Huntington Disease; ALS: Amyotrophic Lateral Sclerosis; ROS: Reactive Oxygen Species; RNS: Reactive Nitrogen Species; SOD1: Superoxide Dismutase 1
Introduction
A number of evidence suggests that generation of free radicals, such as Reactive Oxygen Species (ROS) and Reactive Nitrogen Species (RNS), in the human body causes oxidative stress. Oxidative stress has been shown to play a critical role in development of neurodegenerative disease by enhancing the apoptotic cell death, neuroinflammation, mitochondrial dysfunction, synapse damage, neuronal demyelination, neurofibrillary pathology, tau hyperphosphorylation, aggregation of beta-amyloid Superoxide Dismutase 1 (SOD1) and, alpha-synuclein. All these contributory factors can induce oxidative damage and age related neurodegenerative problem and thus induces pathological condition like Alzheimer’s Disease (AD), Parkinson Disease (PD) and, Amyotrophic Lateral Sclerosis (ALS).
Vitamin E was identified as an essential nutrient [1,2]. The term vitamin E refers to a family of tocochromanols, neutral plant lipids that include tocopherols and tocotrienols. Vitamin E is a fat soluble component which is also known as tocopherol. Vitamin E is highly enriched in human body in which α-tocopherol is most abundant from vitamin E family and highly essential component for human health. The major function of α-tocopherol is believed to be an antioxidant that prevents biological and body physiology from oxidative damage. The most important part of this vitamin is that its antioxidant activity is due to its free radical quenching activity. These free radicals mainly damage the lipid bilayer of cellular components which is made up of unsaturated lipids. Free radical causes the damage of lipid bilayer which is known as lipid peroxidation. Vitamin E prevents the lipid peroxidation of biological membrane by inhibiting or by the interfering the chain reaction of oxidative damage.
Chemistry of Vitamin E
Vitamin E is a group of potent, lipid-soluble, chain-breaking antioxidants, consists of a chroman head with two rings (one phenolic acid ring and one heterocyclic ring) linked to an isoprenoid-derived hydrophobic tail. The vitamin can exist as two types of homologous series: the tocopherol and tocotrienol structures. Both structures are similar except the tocotrienol structure has farnesyl rather than a saturated isoprenoid C16 side chain. Tocopherols (Vitamin E) are equipped to perform a unique function; they can interrupt free radical chain reactions by capturing the free radical; this imparts to them their antioxidant properties. The free hydroxyl group on the aromatic ring is responsible for the antioxidant properties. The hydrogen from this group is donated to the free radical, resulting in a relatively stable free radical form of the vitamin.
Antioxidants mechanism of Vitamin E
Vitamin E members possess a common hydroxylated chromanol ring and a hydrophobic 13-carbon phytyl side chain. Tocopherols have a saturatedphytylchain; whereas the side chain of tocotrienols contains three double bonds. The extent and location of chromanol ring methylation distinguish the various vitamin families. One of the striking features of vitamin E biology is that regardless of the tocopherol composition of dietary intake, the α-tocopherol form is selectively enriched in plasma and tissues of most organisms. Hence, it has been recently proposed that other members of the vitamin E family display biological activities. Accordingly, several studies and observations suggested that α-tocopherol as the major lipid soluble antioxidant in humans [3]. Reports also suggested that that α-tocopherol is an essential nutrient whose absence in human or in mammals severely compromises the health. It has also been noted that the α-tocopherol should be maintained and the recommended daily intake is15mg per day for each healthy adult.
Role of Vitamin E in brain and neuronal injury
Vitamin E (α-tocopherol) is expressed in the brain [4]. Additionally, expression levels of α-tocopherol were shown to increase in patients afflicted with oxidative stress–related diseases [5]. Findings specify that vitamin E is maintained in the CNS and that α-tocopherol may serve to maintain and regulate the brain function (Figure 1). Vitamin E deficiency manifests primarily as neurological and neuromuscular disorders [6-9]. Vitamin E deficiency induced axonalpathy affects sensory neurons. Vitamin E deficiency is a likely contributing factor to the associated neurological deficits. An important pathological hallmark of vitamin E deficiency is the presence of axonal swellings, intracellular aggregates of proliferated endoplasmic reticulum, mitochondria and neurofilaments [10]. These physical barriers for axonal transport may involve in the disruption of neuron function. The evidence regarding this loss is also found with attenuated rates of axonal retrograde and anterograde transport and altered respiratory control in mitochondria isolated from brains of vitamin E deficient rats [11]. In axonal dystrophy, deposition of lipid peroxidation products in lipofuscin aggregates is a hallmark of vitamin E which causes severe oxidative stress.
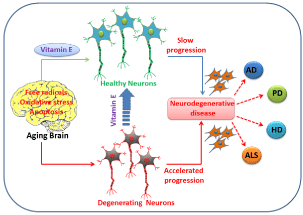
Figure 1: This picture depicts the two different pathways for an aging brain
that is undergoing oxidative stress or apoptosis. Supplement of Vitamin E can
turn degenerating neurons into healthy neurons and slow the progression of
neurodegenerative disease.
Vitamin E and their action on free radicals
Vitamin E chain-breaking natural antioxidant delays or inhibits the oxidation of free radicals and helps to prevent the cellular damage cause by free radicals [12]. Studies show two major function of vitamin E, as a natural antioxidant and some specialized function involving enzyme activities and signaling pathways. On the other hand studies have been also carried out so far and proved the role of vitamin E in protection against oxidative stress, against cellular damage, amyloid beta deposition [13]. Thus, Vitamin E plays an important role in neuroprotection against oxidative stress in neurodegenerative disease and maintains brain health.
Mechanism of neuroprotection by Vitamin E
In the brain, under normal physiological conditions, astrocytes store Vitamin E (α-tocopherol) in perinuclear, a vesicle which regulates the health of the neighboring neuron [14]. During the initiation of oxidative stress, expression of α-tocopherol gene in the astrocytes increases to facilitate delivery of α-tocopherol to the neurons and thus protects neurons from oxidative stress damage (Figure 2). It is unclear if plasma levels of vitamin E actually reflect brain and neural tissue concentrations of a-tocopherol or whether these individuals had adequate cellular uptake mechanisms and/ or efficient Vitamin E transport mechanisms. α-tocopherol travels through the CNS and the mechanisms that regulate this transport process. α-tocopherol transport across the blood-brain barrier was suggested to occur through a scavenger receptor class B1-mediated process [15-17]. In the murine, brain apolipoprotein E particles are thought to play a role in transport α-tocopherol through the cerebrospinal fluid [18,19]. Therefore, it seems that vitamin E utilizes transport routes in the CNS that are shared with other lipid transport mechanisms.
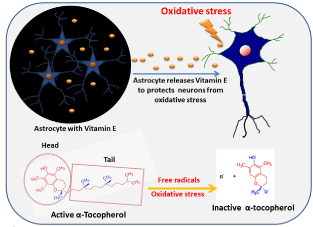
Figure 2: This diagram depicts the role of astrocytes in protection from
oxidative stress. In brain oxidative stress causes active alpha-Tocopherol
denaturation which in turn into inactive alpha-tocopherol.
Vitamin E and Alzheimer’s Disease (AD)
Alzheimer’s Disease (AD) which mainly occurs in old age population after 65 years of age occurs as a result of protein oxidation and lipid peroxidation via a free radical mechanism. It was observed that humans with AD had reduced concentrations of plasma antioxidant micronutrients which suggest that inadequate antioxidant activity is a risk factor in this disease. In another study, it was proven that high plasma levels of vitamin E are associated with a reduced risk of AD in patients. In this neurodegenerative disease beta amyloid (Aβ) protein a pathological hallmark of AD; inducescytotoxicity through an oxidative stress mechanism due to high hydrogen peroxide production leading to neuronal cell death and, finally, AD. Vitamin E blocks the production of hydrogen peroxide by inhibiting chain reaction of free radical formation which finally resulted in cytotoxicity. It has been reported that Vitamin E reduces Aβ-induced cell death in rat brain rat hippocampal cell cultures [20] and PC12 cells [21-23] and mitigates the excitatory amino acid-induced toxicity in neuroblastoma cells [24]. In 1997 the cooperative study on AD suggested that vitamin E treatment may slow down the disease progression in patients either with moderate or severe form of AD condition. High doses of vitamin E delayed the loss of the patient’s ability to carry out daily activities and their consequent placement in residential care for several months. Mangialasche, et al. [22,23], suggested that plasma levels of tocopherols and tocotrienols together with automated Magnetic Resonance Imaging (MRI) measures can help to differentiate patients with AD and Mild Cognitive Impairment (MCI) from the control subjects and prospectively predict the MCI conversion into AD. Therefore, it could be suggests that plasmatocopherols and tocotrienols as indirect indicators of AD pathology.
Oxidative stress, Vitamin E and AD
Accumulating evidence suggests the primary exposure of brain to oxidative stress. Oxidative stress is generally characterized by an imbalance in production of Reactive Oxygen Species (ROS) and antioxidative defense system which are responsible for the removal of ROS [25], that causes protein oxidation, lipid oxidation, DNA oxidation and glycoxidation [26,27] which are closely associated with the development of Alzheimer’s Disease (AD). AD is characterized by neuronal and synaptic loss [28] which is associated with accumulation of extracellular Aβ plaques produced from APP processing and intracellular NFTs composed of aggregated hyper phosphorylated tau proteins in brain, proliferation of astrocytes and activation of microglial. α-tocopherol a powerful, lipid-soluble chain-breaking antioxidant is found in the lipid membranes [29]. Evidence show vitamin E has attenuate toxic effects of β-amyloid and improves cognitive performance in rodents [30-32]. Sano et al. [33], reported treatment with α-tocopherol (2000 IU a day) in patients with moderately severe impairment from AD, contributed to reduced neuronal damage and slowed the progression of AD, which indicates that the α-tocopherol may delay clinically important function deterioration in AD patients. A report by Sung, et al. [34], suggested that vitamin E administered early prior to the appearance of the pathology during the evolution of AD, suppresses brain lipid peroxidation, mitigates brain oxidative stress and significantly reduces Aβ levels and senile plaque deposition in transgenic mice model of AD. However, no significant effect is observed on the amyloidotic phenotype of these animals if vitamin E supplementation after amyloid plaques were already deposited despite a reduction in brain oxidative stress [34]. This could suggest that regular intake of rice bran oil can protect the brain form oxidative injury as well as inhibit the progression of AD pathology.
Vitamin E and Parkinson disease
Parkinson’s Disease (PD) is a neurodegenerative movement disorder of central nervous system characterized by the degeneration and progressive loss of dopaminergic neurons in the substantia nigra pars compacta. Oxidative stress causes low levels of endogenous antioxidants, increased reactive species, augmented dopamine oxidation and, plays an important role in the etiology and progression of PD [35]. It has been suggested that Vitamins E has protective effects against neuronal death in PD. Formation of free radicals and reactive quinone species due to the oxidative chemistry of dopamine. Such dopamine-derived quinones may react with several cellular targets which results in deleterious processes; studies have shown a strong correlation between quinone species and in the pathogenesis of PD and progression of the disorder. Vitamin E is an example of alternative treatment for PD that was found to be ineffective or showed harmful side effects. Studies show a strong correlation between that free radical damage and PD. A large study in the 1980s was done with early PD by using Vitamin E, a moderately potent antioxidant. The study of Vitamin E did not demonstrate a slowing effect or neuroprotection and in fact, showed that it could potentially be harmful to PD patients; because of lack of sufficient data there is a need to study more about this. Other studies have shown beneficial effects with high dose of vitamin E dietary supplementation or parental vitamin E administration (e.g. vitamin E succinate) that can be a successful therapeutic strategy for the prevention or treatment of PD [36]. The study with vitamin E supplementation shows higher vitamin E consumption, in the top 1/5 to be precise, showed a 36% reduction of the severity of their symptoms compared to those in the lowest quintile (1/5) of subjects. A different study showed that vitamin E as a treatment of PD. Overall, researchers found that diets with even a moderate amount of vitamin E led to a 19% reduction in risk of developing PD.
Vitamin E and Huntington disease
Huntington’s Disease (HD) is a genetically autosomal dominant neurodegenerative disease also characterized by psychiatric symptoms, movement disorder and, progressive dementia, noticeable with people between the 40 to 50 years of age. It leads to death 15 to 20 years after the onset of symptoms [37]. HD is a familial disease, passed from parent to child through a mutation of the HD gene. The mechanism(s) of mutation is Htt-induced degeneration involving loss of neurotrophic functions which results from excessive activation of glutamate-gated ion channels; causes oxidative stress that kills neurons [38]. The gene mutation may cause a subtle impairment of energy metabolism leading to oxidative stress that ultimately leads to neuronal degeneration, initially in the striatum and later in other brain regions [39]. Reports also suggest increase in level of oxidative stress and neuroinflammation in human and animal models of HD. Fewer antioxidant trails have been studied in HD preclinical models relative to the other diseases. Flint Beal’s group, who pioneered the quinolinic acid model of HD, reported that priorad ministration of the antioxidants vitamin E, β-carotene, or ascorbic acid do not provided protection against quinolinate-induced striatal neurotoxicity to toxin challenge. Human clinical trials in HD are ongoing. Double-blind, placebo-controlled study of high-dose α-tocopherol on a HD patients has been carried out by Peyser, et al [40]. Previous evidence shows that Vitamin E had no effect on neurologic or psychiatric symptoms but post hoc analysis reveals a significant effect of intervention on neurological symptoms in early stage patient [40].
Vitamin E and Amyotrophic lateral sclerosis
Amyotrophic Lateral Sclerosis (ALS) disorder involves the death of neurons due to oxidative stress. The pathogenesis of amyotrophic lateral sclerosis is supported by the presence of biomarkers of oxidative damage in sporadic ALS patients [41] and by the occurrence of familial ALS among carriers of several distinct mutations in the copper/zinc Superoxide Dismutase gene (SOD1), a critical component involve in cellular antioxidant defense mechanisms [42]. Studies show the role of Vitamin E to delay the onset of clinical disease expressing mutant copies of the gene coding for SOD1, in transgenic mice animal model of ALS [43]. Although, some other reports suggest that vitamin Esupplementation was found to be ineffective in randomized trials of ALS patients [44]. However, it remains possible that high intake of vitamin E in a healthy persons diet could reduce disease risk or delay its onset. Studies also carried to find the association of vitamin E intake with ALS by Wang, et al. [45], showed evidence that longterm vitamin E supplementation can lower ALS rates and therefore, a possible protective effect of vitamin E deserves further consideration. More evidence is needed to prove that antioxidants, such as vitamin E, provide a clinical therapeutic effect against oxidative stress-related diseases [46]. Since oxidative stress may contribute to the pathogenesis of ALS, Ascherio, et al. [47], examined whether individuals who regularly use supplements of the antioxidant vitamins E and C have a lower risk of ALS than nonusers and suggest that vitamin E supplementation could have a role in ALS prevention. Veldink, et al. [48], suggested that intake of polyunsaturated fatty acids and vitamin E reduces the risk of developing ALS. ALS is associated with oxidative stress, mitochondrial dysfunction, protein misfolding, axonal strangulation, apoptosis, inflammation, glutamate excitotoxicity and defects in neurotrophic biology.
Conclusion
Cumulative studies and clinical reports suggest that vitamin E has antioxidative effect and prevents neuronal degeneration from oxidative damage. We recommend that regular supplementation of vitamin E or foods enriched with vitamin E will slowdown the progression of neurodegeneration and thus maintain brain health.
References
- Wolf G. The discovery of the antioxidant function of vitamin E: The contribution of Henry A. Mattill J Nutr. 2005; 135: 363-366.
- Patel J, Matnor NA, Iyer A, Brown L. A Regenerative Antioxidant Protocol of Vitamin E and alpha-Lipoic Acid Ameliorates Cardiovascular and Metabolic Changes in Fructose-Fed Rats. Evid Based Complement Alternat Med. 2011; 2011: 120801.
- Yau TM, Weisel RD, Mickle DA, Burton GW, Ingold KU, Ivanov J, et al. Vitamin E for coronary bypass operations. A prospective, double-blind, randomized trial. J Thorac Cardiovasc Surg. 1994; 108: 302-310.
- Hosomi A, Goto K, Kondo H, Iwatsubo T, Yokota T, Ogawa M, et al. Localization of alpha-tocopherol transfer protein in rat brain. Neurosci Lett. 1988; 256: 159-162.
- Copp RP, Wisniewski T, Hentati F, Larnaout A, Ben Hamida M, Kayden HJ. Localization of alpha-tocopherol transfer protein in the brains of patients with ataxia with vitamin E deficiency and other oxidative stress related neurodegenerative disorders. Brain Res. 1999; 822: 80-87.
- Germano M, Shkuropatov AY, Permentier H, de Wijn R, Hoff AJ, Shuvalov V, et al. Pigment organization and their interactions in reaction centers of photosystem II: optical spectroscopy at 6 K of reaction centers with modified pheophytin composition. Biochemistry. 2001; 40: 11472-11482.
- Gohil K, Vasu VT, Cross CE. Dietary alpha-tocopherol and neuromuscular health: search for optimal dose and molecular mechanisms continues. Mol Nutr Food Res. 2010; 54: 693-709.
- Muller DP. Vitamin E and neurological function. Mol Nutr Food Res. 2010; 54:710-718.
- Muller L, Theile K, Bohm V. In vitro antioxidant activity of tocopherols and tocotrienols and comparison of vitamin E concentration and lipophilic antioxidant capacity in human plasma. Mol Nutr Food Res. 2010; 54: 731-42.
- Southam E, Thomas PK, King RH, Goss-Sampson MA, Muller DP. Experimental vitamin E deficiency in rats. Morphological and functional evidence of abnormal axonal transport secondary to free radical damage. Brain. 1991; 114: 915-936.
- Cuddihy SL, Ali SS, Musiek ES, Lucero J, Kopp SJ, Morrow JD, et al. Prolonged alpha-tocopherol deficiency decreases oxidative stress and unmasks alpha-tocopherol-dependent regulation of mitochondrial function in the brain. J Biol Chem. 2008; 283: 6915-6924.
- Luque-Contreras D, Carvajal K, Toral-Rios D, Franco-Bocanegra D, Campos-Pena V. Oxidative stress and metabolic syndrome: cause or consequence of Alzheimer's disease? Oxid Med Cell Longev. 2014; 497802.
- Grundman M. Vitamin E and Alzheimer disease: the basis for additional clinical trials. Am J ClinNutr. 2000; 71: 630-636.
- Lamarche F, Signorini-Allibe N, Gonthier B, Barret L. Influence of vitamin E, sodium selenite, and astrocyte-conditioned medium on neuronal survival after chronic exposure to ethanol. Alcohol. 2004; 33: 127-138.
- Balazs Z, Panzenboeck U, Hammer A, Sovic A, Quehenberger O, Malle E, et al. Uptake and transport of high-density lipoprotein (HDL) and HDL-associated alpha-tocopherol by an in vitro blood-brain barrier model. J Neurochem. 2004; 89: 939-950.
- Mardones P, Strobel P, Miranda S, Leighton F, Quinones V, Amigo L, et al. Alpha-tocopherol metabolism is abnormal in scavenger receptor class B type I (SR-BI)-deficient mice. J Nutr. 2002; 132: 443-449.
- Castillo O, Rozowski J, Cuevas A, Maiz A, Soto M, Mardones F, et al. Nutrients intake in elderly people living in Providence, Santiago de Chile. Rev Med Chil. 2002; 130: 1335-1342.
- Vatassery GT, Lam C, Smith WE, Quach HT. Apolipoprotein E exerts selective and differential control over vitamin E concentrations in different areas of mammalian brain. J Neurosci Res. 2006; 84: 1335-1342.
- Vatassery GT, Quach HT, Smith WE, Santacruz KS, Roy S. Apolipoprotein e deficiency leads to altered brain uptake of alpha tocopherol injected into lateral cerebral ventricles. BiochimBiophys Acta. 2007; 1772: 797-803.
- Sano M, Ernesto C, Thomas RG, Klauber MR, Schafer K, Grundman M, et al. A controlled trial of selegiline, alpha-tocopherol, or both as treatment for Alzheimer's disease. The Alzheimer's Disease Cooperative Study. N Engl J Med. 1997; 336: 1216-1222.
- Mangialasche F, Kivipelto M, Mecocci P, Rizzuto D, Palmer K, Winblad B, et al. High plasma levels of vitamin E forms and reduced Alzheimer's disease risk in advanced age. J Alzheimers Dis. 2010; 20: 1029-1037.
- Mangialasche F, Solomon A, Kareholt I, Hooshmand B, Cecchetti R, Fratiglioni L, et al. Serum levels of vitamin E forms and risk of cognitive impairment in a Finnish cohort of older adults. Exp Gerontol. 2013; 48: 1428-1435.
- Mangialasche F, Westman E, Kivipelto M, Muehlboeck JS, Cecchetti R, Baglioni M, et al. Classification and prediction of clinical diagnosis of Alzheimer's disease based on MRI and plasma measures of alpha-/gamma-tocotrienols and gamma-tocopherol. J Intern Med. 2013; 273: 602-621.
- Pavlik VN, Doody RS, Rountree SD, Darby EJ. Vitamin E use is associated with improved survival in an Alzheimer's disease cohort. Dement Geriatr Cogn Disord. 2009; 28: 536-540.
- Harman D. The aging process. Proc Natl Acad Sci. 1981; 78: 7124-7128.
- Moreira PI, Zhu X, Nunomura A, Smith MA, Perry G. Therapeutic options in Alzheimer's disease. Expert Rev Neurother. 2006; 6: 897-910.
- Nunomura A, Castellani RJ, Zhu X, Moreira PI, Perry G, Smith MA. Involvement of oxidative stress in Alzheimer disease. J Neuropathol Exp Neurol. 2006; 65: 631-641.
- Kamat PK, Kalani A, Rai S, Swarnkar S, Tota S, Nath C, et al. Mechanism of Oxidative Stress and Synapse Dysfunction in the Pathogenesis of Alzheimer's Disease: Understanding the Therapeutics Strategies. Mol Neurobiol. 2016; 53: 648-661.
- Foy CJ, Passmore AP, Vahidassr MD, Young IS, Lawson JT. Plasma chain-breaking antioxidants in Alzheimer's disease, vascular dementia and Parkinson's disease. QJM. 1999; 92: 39-45.
- Devore EE, Grodstein F, van Rooij FJ, Hofman A, Stampfer MJ, Witteman JC, et al. Dietary antioxidants and long-term risk of dementia. Arch Neurol. 2010; 67: 819-825.
- Devore EE, Kang JH, Stampfer MJ, Grodstein F. Total antioxidant capacity of diet in relation to cognitive function and decline. Am J Clin Nutr. 2010; 92: 1157-1164.
- Montiel T, Quiroz-Baez R, Massieu L, Arias C. Role of oxidative stress on beta-amyloid neurotoxicity elicited during impairment of energy metabolism in the hippocampus: protection by antioxidants. Exp Neurol. 2006; 200: 496-508.
- Sano M, Ernesto C, Thomas RG, Klauber MR, Schafer K, Grundman M, et al. A controlled trial of selegiline, alpha-tocopherol, or both as treatment for Alzheimer's disease. N Engl J Med. 1997; 336: 1216-1222.
- Sung S, Yao Y, Uryu K, Yang H, Lee VM, Trojanowski, et al. Early vitamin E supplementation in young but not aged mice reduces Abeta levels and amyloid deposition in a transgenic model of Alzheimer's disease. FASEB J. 2004; 18: 323-325.
- Sutachan JJ, Casas Z, Albarracin SL, Stab BR, Samudio I, Gonzalez J, et al. Cellular and molecular mechanisms of antioxidants in Parkinson's disease. Nutr Neurosci. 2012; 15: 120-126.
- Fariss MW, Zhang JG. Vitamin E therapy in Parkinson's disease. Toxicology. 2003; 89: 129-146.
- Browne SE, Ferrante RJ, Beal MF. Oxidative stress in Huntington's disease. Brain Pathol. 1999; 9: 147-163.
- Leavitt BR, Hayden MR. Is tetrabenazine safe and effective for suppressing chorea in Huntington's disease. Nat Clin Pract Neurol. 2006; 2: 536-537.
- Jenkins BG, Rosas HD, Chen YC, Makabe T, Myers R, MacDonald M, et al. 1H NMR spectroscopy studies of Huntington's disease: correlations with CAG repeat numbers. Neurology. 1998; 50: 1357-1365.
- Peyser CE, Folstein M, Chase GA, Starkstein S, Brandt J, Cockrell JR, et al. Trial of d-alpha-tocopherol in Huntington's disease. Am J Psychiatry. 1995; 152: 1771-1775.
- Barber SC, Shaw PJ. Oxidative stress in ALS: key role in motor neuron injury and therapeutic target. Free RadicBiol Med. 2010; 48: 629-641.
- Siddique T, Nijhawan D, Hentati A. Familial amyotrophic lateral sclerosis. J Neural TransmSuppl. 1997; 49: 219-233.
- Gurney ME, Cutting FB, Zhai P, Doble A, Taylor CP, Andrus PK, et al. Benefit of vitamin E, riluzole, and gabapentin in a transgenic model of familial amyotrophic lateral sclerosis. Ann Neurol. 1996; 39: 147-157.
- Graf M, Ecker D, Horowski R, Kramer B, Riederer P, Gerlach M, et al. High dose vitamin E therapy in amyotrophic lateral sclerosis as add-on therapy to riluzole: results of a placebo-controlled double-blind study. J Neural Transm (Vienna). 2005; 112: 649-660.
- Wang H, O'Reilly EJ, Weisskopf MG, Logroscino G, McCullough ML, Schatzkin A, et al. Vitamin E intake and risk of amyotrophic lateral sclerosis: a pooled analysis of data from 5 prospective cohort studies. Am J Epidemiol. 2011; 173: 595-602.
- Majima HJ, Indo HP, Suenaga S, Matsui H, Yen HC, Ozawa T. Mitochondria as possible pharmaceutical targets for the effects of vitamin E and its homologues in oxidative stress-related diseases. Curr Pharm Des. 2011; 17: 2190-2195.
- Ascherio A, Weisskopf MG, O'Reilly E J, Jacobs EJ, McCullough ML, Calle EE, et al. Vitamin E intake and risk of amyotrophic lateral sclerosis. Ann Neurol. 2005; 57: 104-110.
- Veldink JH, Kalmijn S, Groeneveld GJ, Wunderink W, Koster A, de Vries JH, et al. Intake of polyunsaturated fatty acids and vitamin E reduces the risk of developing amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry. 2007; 78: 367-371.
