
Original Article
J Pediatr & Child Health Care. 2023; 8(1): 1060.
Diagnostic Value of PTX-3 Combined with PCT and CRP for Neonatal Sepsis
Yan Jin1,2; Meng Nan Lu2; Yanfeng Xiao2; Chunyan Yin2*
¹Department of Pediatrics, Shanxi Sengong Hospital, PR China
²Department of Pediatrics, Second Affiliated Hospital of Xi’an Jiaotong University, PR China
*Corresponding author: Chunyan Yin Department of Pediatrics, Second Affiliated Hospital of Xi’an Jiaotong University, Xi’an, ShanXi 710049, PR China. Tel: +86-029-87679543; Fax: 860298767542 Email: [email protected]
Received: June 24, 2023 Accepted: July 27, 2023 Published: August 03, 2023
Abstract
Aim: Neonatal sepsis is a critical illness among neonates and is the leading cause of neonatal death. In this study, we compared the changes in serum PTX-3 levels in neonates with sepsis and non-septicemia, as well as the diagnostic advantages and value of serum PTX-3 compared to traditional indicators such as WBC, CRP, and PCT.
Methods: A total of 109 neonates were included in this study. Neonates were divided into three groups: 35 neonates were assigned to the sepsis group; 36 to the local infection group; and 38 assigned to the control group. Blood was collected to measure complete blood counts, CRP, PCT, liver and kidney function, lactate, and PTX-3 levels.
Results: CRP, PCT, and PTX-3 in the sepsis group were significantly higher compared to the local infection group and non-infection group (P<0.01 or P>0.05). PCT and PTX-3 levels in the local infection group were significantly higher compared to the non-infection group (P>0.05). The combination of WBC+CRP+PCT+PTX-3 was more sensitive for the diagnosis of neonatal infection. For the diagnosis of neonatal sepsis, CRP was the most sensitive index, with PTX-3 having the best specificity. The combination of WBC+CRP+PTX-3 was found to significantly improve the sensitivity and specificity of diagnosis.
Conclusion: PTX-3 could be used as a new biomarker for neonatal sepsis, while the combination with WBC, CRP, and PCT could significantly improve the sensitivity and specificity for sepsis diagnosis.
Keywords: Neonatal sepsis; Neonatal infection; Early diagnosis; Pentraxin3; Receiver operating characteristic analysis
What is already Known on this Topic
1. The clinical signs of neonatal sepsis are nonspecific, as well as blood culture that is considered as a gold standard have a low positive rate in sepsis due to the widespread use of antibiotics
2. The serum PTX-3 plays an important role in the occurrence and progression of various infectious diseases
3. The specificity and sensitivity of serum PTX-3 was excellent in adult sepsis patients.
What this Paper Adds
1. The diagnostic value of PTX-3 combined with WBC, CRP, and PCT was firstly evaluated through Receiver Operating Characteristic (ROC) analysis for the early diagnosis of neonatal infection and sepsis.
2. It is the first study to report the sensitivity, specificity, and cut-off points of the combined indicators relevant to PTX-3 for neonatal infection and sepsis.
For the diagnosis of neonatal sepsis, CRP was the most sensitive single indicator, and PTX-3 had the strongest specificity. The combination of WBC+CRP+PTX-3 could significantly improve the sensitivity and specificity of diagnosis.
Background
Procianoy RS, Silveira RC. The challenges of neonatal sepsis management. J Pediatr (Rio J). 2020 Mar-Apr; 96 Suppl 1(Suppl 1):80-86. is a systemic inflammatory response caused by pathogenic microorganisms entering the blood of neonates in a variety of ways [1], and is a critical illness observed in neonates after infections [2]. Although neonatal sepsis has a relatively low incidence, it has unfortunately a high mortality rate [3]. Early diagnosis and prompt treatment are necessary to improve survival and reduce mortality.
The clinical diagnosis of neonatal sepsis includes clinical signs and specific laboratory indicators, such as C-Reactive Protein (CRP), Procalcitonin (PCT), and White Blood Cell count (WBC) [4,5]. However, the clinical signs and symptoms of neonatal sepsis are nonspecific and could be easily confused with other noninfectious causes although laboratory indicators could be used as a complementary diagnosis. Blood culture remains the gold standard for neonatal sepsis, with a positive rate of only 3% in sepsis due to the widespread use of antibiotics [6], which limits the use of the culture method for diagnosis. In addition, traditional sepsis markers, such as white blood cell counts, tend to stabilize only 12 hours after birth [7]. There is a physiological increase in PCT within 24 hours of birth, and it begins to decrease to normal at 96 hours. This gives rise to a lag in the diagnosis of neonatal sepsis [8]. Therefore, finding novel biomarkers for the early diagnosis of neonatal sepsis is urgently needed to guide treatment.
Pentraxin3, located in the q25 region of chromosome 3, consists of 2 introns and 3 exons [9]. PTX-3 is secreted by endothelial cells, neutrophils, mononuclear phagocytes, chondrocytes, renal innate cells, and dendritic cells after exposure to inflammatory factors such as interleukin-1Β and tumor necrosis factor-a. It plays an important role in the occurrence and progression of infectious diseases, kidney diseases, and cardiovascular and cerebrovascular diseases [10,11]. Previous studies have demonstrated the specificity and sensitivity of serum PTX-3 in adult sepsis patients could reach 0.77 and 0.68 [12]. Furthermore, PTX-3 was found to be more reliable compared to PCT, CRP, and other indicators for prognosis. However, studies on PTX-3 in neonatal sepsis are limited.
The purpose of this study was to compare the changes in serum PTX-3 levels in neonatal sepsis and non-septicemia, as well as the diagnostic advantages and value of serum PTX-3 with WBC, CRP, and PCT in neonatal patients with sepsis.
Method
Study Enrollment
From June 2020 to December 2020, 109 neonates who were admitted to the Second Affiliated Hospital of Xi'an Jiaotong University and the Department of Neonatology of Shaanxi Sengong Hospital were enrolled in the study. Patient characteristics included age of 1 day to 21 days, gestational age of 34 weeks to 41 weeks, and birth weight of 1795g to 4200g. Patients with congenital malformations, intrauterine viral infections, previous antibiotic treatment, or lack of parental consent were excluded from the study. Our study was approved by the Ethics Committee of the Second Affiliated Hospital of Xi'an Jiaotong University, and informed consent was obtained from the parents or guardians.
Study Groups
The 109 neonates enrolled in the study were divided into three groups: the sepsis group had 35 children (23 males and 12 females) who were diagnosed with neonatal sepsis, including 33 cases of clinical sepsis, 2 cases of confirmed sepsis (1 case was Escherichia coli, 1 case was Staphylococcus epidermidis). In the local infection group, there were 36 neonates with mild infections but were below the standard required for the diagnosis of sepsis, of which, 34 cases had infectious pneumonia and 2 cases with omphalitis. The control group consisted of 38 neonates without infections (18 males and 20 females) and included 5 from diabetic mothers, 10 from pregnancy-induced hypertension mothers, 7 from intrauterine distress, and 5 with macrosomia. There were 2 infants with hypothyroidism during pregnancy, 5 premature infants, and 4 infants with oligohydramnios during delivery.
Diagnostic Criteria
The expert consensus on the diagnosis and treatment of neonatal sepsis prepared by the Neonatal Group of the Chinese Pediatric Society, Chinese Medical Association in 2019 was used as the diagnostic criteria for neonatal sepsis [13].
Specimen Collection and Testing
After admission, venous blood was collected for routine blood tests, CRP, PCT, liver and kidney function, lactic acid, and other tests. Blood cultures were performed at the same time. In addition, 1ml of venous blood was centrifuged at 3000g for 6 min, and then the resulting serum was placed in a sterile tube and frozen, store at -80? until required. Serum PTX-3 levels were measured using the double antibody sandwich method ABC-ELISA (R&D systems, Minneapolis, MN, USA). PCT levels were determined using the electrochemiluminescence immunoassay on a Roche Cobas 8000 e602 automatic analyzer (Roche Diagnostics, Mannheim, Germany). CRP was measured using the QuikRead Go CRP System (Orion Diagnostica, Finland).
Statistical Analysis
All measurement data were evaluated for normality using the Kolmogorov-Smirnov test, and measurement data conforming to a normal distribution were expressed as mean ± SD. Comparison between the two groups was performed using the T-test. ANOVA test was performed for measurement data between multiple groups with homogenous variance. Comparison between the two groups was performed using the Bonferroni post-test. Enumeration data were expressed as rates, and the correlation between two groups of data was analyzed using Pearson correlation. Binary logistic regression was used to identify useful biological indicators for neonatal infection and neonatal sepsis. Receiver Operating Characteristic (ROC) curves were used to determine the optimal diagnostic cut-offs for WBC, CRP, PCT, and PTX-3 in neonatal infection and sepsis. P<0.05 was considered statistically significant.
Results
Characteristics of the study groups
A total of 109 neonates were enrolled in this study and included 38 neonates in the non-infection group, 36 in the local infection group, and 35 in the sepsis group. The incidence rates of maternal premature rupture of membranes in the sepsis group, local infection group, and non-infection group were 29.6%, 12.5%, and 2.7%, respectively, and the differences were statistically significant, P<0.05. There were no statistical differences for gender, mode of delivery, gestational age, birth weight, and the presence of amniotic fluid contamination at birth among the three groups, P>0.05 (Table 1).
General Information
Non-infection group (38)
Local infection group (36)
Sepsis group (35)
x2/F
P
Gender: (Male/Female)
18/20
25/11
23/12
4.347
0.114
Mode of delivery: (Vaginal delivery /caesarean section)
13/25
20/16
18/17
3.829
0.148
Gestational age at birth (weeks)
38.3±1.7
39.1±1.2
38.9±1.4
19.129
0.160
Birth weight (g)
3140±671
3358±386
3322±477
1.819
0.167
Premature rupture of membranes (yes/no)
1/37
4/32
8/27
7.129
0.028
Amniotic fluid contamination (yes/no)
7/31
6/30
4/31
0.723
0.696
Table 1: Characteristics of the study groups.
Comparison of PTX3 Levels and Infection Indicators in the Sepsis, Local Infection, and Non-Infection Groups
PTX3 levels and infection indicators in the sepsis, local infection, and non-infection groups were analyzed. WBC levels in the sepsis group and the local infection group were higher compared to the non-infection group, and the difference was statistically significant (P<0.05). Platelet counts (PLT) and distribution width (PDW) were not significantly different among the three groups (P>0.05). There were significant differences in Lactate Dehydrogenase (LDH) and Ca among the three groups (P<0.05). There were no significant differences in Albumin (ALB), Serum Creatinine (Scr), Lactic Acid (Lac), and Creatine Kinase MB Form (CK-MB) among the three groups (P>0.05). Serum CRP, PCT, and PTX-3 levels in the sepsis group were significantly higher compared to the local infection group and non-infection group (P<0.01 or P<0.05); PCT and PTX-3 levels in the local infection group were significantly higher compared to the non-infection group (P>0.05), while there was no statistical difference in CRP between the two groups (Table 2).
Detection Indicator
Sepsis group
(n=35)Local infection group
(n=36)Non-infection group
(n=38)F
P
WBC (×109/L)
18.19±6.13 **
15.93±5.12*
12.92±4.31
6.703
0.002
PLT (×109/L)
277.29±91.54
255.56±53.87
249.74±58.97
1.558
0.215
PDW (fL)
10.73±0.85
11.21±1.88
10.84±0.95
1.399
0.251
Lac (mmol/L)
2.72±0.90
2.60±0.85
2.28±0.72
2.038
0.135
Ca (mmol/L)
2.22±0.22 **#
2.31±0.10
2.35±0.12
6.945
0.001
LDH (U/L)
542.03±179.87**
493.86±157.04
427.08±134.22
3.980
0.022
ALB (g/L)
38.21±6.16
36.79±4.05
36.1±2.73
2.001
0.140
Scr (umol/L)
54.17±14.04
49.26±15.71
52.00±13.42
1.037
0.358
CRP (mg/L)
17.94±5.83 **##
6.26±2.02
2.95±0.92
11.203
0.000
PCT (ng/ml)
3.50±1.11 **#
2.02±0.68**
0.23±0.07
13.002
0.000
PTX-3 (ng/ml)
25.89±4.58 **##
22.11±4.28**
17.00±3.89
40.407
0.000
Table 2: Comparison of PTX-3 and infection indicators in the sepsis, local infection, and non-infection groups.
Correlation Analysis of PTX-3 with WBC, CRP, and PCT for the Three Groups of Neonates
Pearson correlation analysis of serum PTX-3 levels with WBC, CRP, and PCT in neonates of the sepsis, infection, and non-infection groups was performed. The results showed that PTX-3 levels were correlated with CRP and PCT (P<0.01), with correlation coefficients r of 0.275 and 0.318, respectively. PTX-3 did not correlate with WBC (P>0.05) (Figure 1).
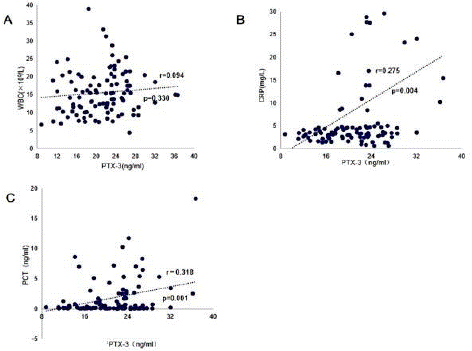
Figure 1: Correlation analysis of PTX-3 levels with WBC, CRP, and PCT for the three groups. (A) Correlation between serum PTX-3 and WBC. (B) Correlation between serum PTX-3 and CRP. (C) Correlation between serum PTX-3 and PCT.
Multivariate Logistic Regression Analysis of Neonatal Infection-Related Inflammatory Indicators
Using the infection group (including sepsis group and local infection group) and the non-infection group as dependent variables, and WBC, PLT, CRP, PCT, PTX-3, PDW, Lac, Ca, LDH, ALB, Scr, and CK-MB as independent variables, multivariate logistic regression analysis was performed. The results showed that PCT, PTX-3, and PDW had significant changes and were closely related to neonatal infection (P<0.05) (Table 3).
Table 3: Multivariate Logistic Regression Analysis of Inflammatory Indicators Related to Neonatal Infection.
Multivariate Logistic Regression Analysis of Neonatal Sepsis-Related Indicators
Using WBC, PLT, CRP, PCT, PTX-3, PDW, Lac, Ca, LDH, ALB, Cr, and CK-MB as independent variables, multivariate logistic regression analysis was performed with the sepsis group and local infection group as dependent variables. The results showed that WBC, PLT, CRP, PTX-3, PDW, and pH showed significant changes in neonatal sepsis, with PTX-3 and CRP having the most significant changes (P<0.01) (Table 4).
Variables
B
S.E.
Wald
df
P
OR
95% CI
WBC
0.136
0.045
9.045
1
0.003
1.146
1.049~1.252
PLT
0.013
0.004
9.418
1
0.002
1.013
1.005~1.021
CRP
0.104
0.023
19.953
1
0.000
1.11
1.060~1.162
PTX-3
0.28
0.068
16.923
1
0.000
1.323
1.158~1.512
PDW
-0.602
0.248
5.89
1
0.015
0.548
0.337~0.891
pH
-8.85
4.419
4.011
1
0.045
0.000
0.000~0.828
Table 4: Multivariate Logistic Regression Analysis of Inflammatory Indicators Related to Neonatal Sepsis.
The Diagnostic Value of the ROC Curve in Predicting WBC, CRP, PCT, and PTX-3 In Neonatal Infection
ROC curve analysis was performed on WBC, CRP, PCT, and PTX-3 in the infection group (i.e., sepsis group and local infection group) and non-infection group. The results showed that the combined indicators PTX-3+CRP+PCT+WBC had the largest area under the curve of 0.954, with a sensitivity of 97.4%, specificity of 83.1%, and the best cut-off value of 0.714 (P<0.01). Among the single indicators, the area under the curve of 0.878 for PTX-3 was the largest, with a sensitivity of 84.2%, specificity of 84.5%, and the best cut-off value of 20.36 (P<0.01). This was followed by PCT, with Area Under the ROC curve (AUC) value of 0.776, a sensitivity of 92.1%, specificity of 63.4%, and the best cut-off value of 0.52 (P<0.01). Next was CRP, with AUC value of 0.727, a sensitivity of 100%, specificity of 42.3%, and the best cut-off value of 4.85 (P<0.01) (Table 5 & Figure 2A).
Predictive value
AUC
S.E.
P
95% CI
Best cut-off value
Sensitivity %
Specificity %
WBC+CRP+PCT+PTX-3
0.954
0.018
0
0.918~0.990
0.714
97.4
83.1
WBC
0.678
0.052
0.002
0.577~0.780
16.87
84.2
46.5
CRP
0.727
0.047
0
0.634~0.819
4.85
100
42.3
PCT
0.776
0.043
0
0.691~0.861
0.52
92.1
63.4
PTX-3
0.878
0.035
0
0.810~0.946
20.36
84.2
84.5
Table 5: The diagnostic value of WBC, CRP, PCT, and PTX-3 combined indicators for neonatal infection.
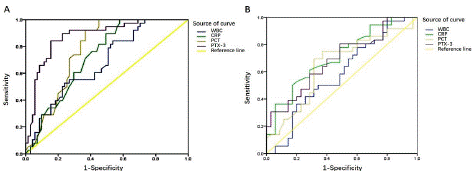
Figure 2: ROC curve predicts the diagnostic value of WBC, CRP, PCT, and PTX-3 in neonatal infection and sepsis. (A) ROC curve to determine the best cut-off value of WBC, CRP, PCT, and PTX-3 for neonatal infection. (B) ROC curve to determine the best cut-off value of WBC, CRP, PCT, and PTX-3 for neonatal sepsis.
The Diagnostic Value of the ROC Curve in Predicting WBC, CRP, PCT, And PTX-3 For Neonatal Sepsis
ROC curve analysis was performed for WBC, CRP, PCT, and PTX-3 in the sepsis and local infection groups. The results showed that the area under the curve of the combined indicators WBC+CRP+PTX-3 was the largest, which was 0.855, with a sensitivity of 83.3%, specificity of 80%, and the best cut-off values of 0.614 (P<0.01). Among the single indicators, the area under the CRP curve was the largest, which was 0.710, and had a sensitivity of 82.9%, specificity of 50%, and the best cut-off value of 5.35 (P<0.01), followed by PTX-3, with an AUC value of 0.689, a sensitivity of 51.4%, specificity of 80.6%, and the best cut-off value of 25.39 (P<0.01), (Table 6 & Figure 2B).
Predictive value
AUC
S.E.
P
95% CI
Best cut-off value
Sensitivity %
Specificity %
WBC+CRP+PTX-3
0.855
0.028
0
0.800~0.909
0.614
83.3
80
WBC
0.582
0.043
0.065
0.497~0.666
20.21
40
77.8
CRP
0.71
0.039
0
0.634~0.787
5.35
82.9
50
PCT
0.65
0.067
0.03
0.566~0.733
1.55
68.6
69.4
PTX-3
0.689
0.04
0
0.610~0.768
25.39
51.4
80.6
Table 6: Diagnostic value of the combined indicators WBC, CRP, PCT, and PTX-3 for neonatal sepsis.
Discussion and Conclusions
To the best of our knowledge, this study is the first study to use the ROC curve to analyze the value of PTX-3 combined with WBC, CRP, and PCT for the early diagnosis of neonatal infection and sepsis. It is also the first study to report the sensitivity, specificity, and cut-off points of the above-mentioned combined indicators for neonatal infection and sepsis. We found that PTX-3 levels were the most valuable single indicator for the early diagnosis of neonatal sepsis. However, the combination of WBC+CRP+PCT+PTX-3 was more effective for the diagnosis of neonatal infection. For the diagnosis of neonatal sepsis, CRP was found to be the most sensitive single indicator; however, PTX-3 had the strongest specificity. The combination of WBC+CRP+PTX-3 could significantly improve the sensitivity and specificity of diagnosis.
We performed blood routine examinations of the sepsis, the local infection, and the non-infection groups. The results showed that there was a statistical difference in WBC counts among the three groups (P<0.05). WBC in the sepsis group was significantly higher compared to the local infection and non-infection groups (P<0.05). However, correlation analysis demonstrated no correlation between WBC and PCT, CRP, and PTX-3 levels. The reasons could be as follows: First, WBC is the most commonly used laboratory indicator for determining the presence or absence of infection, however, several factors affect WBC clinically. In addition to infection and inflammation, breastfeeding, strenuous exercise, and emotional changes could lead to elevated WBC counts. In addition, normal newborns have higher WBC levels compared to children and infants. Second, the white blood cell counts in a healthy individual are stable. During an acute infection, especially with bacterial infections, the white blood cell count increases significantly, and these levels significantly correlate with the severity of bacterial infection [14]. However, in severe infections, toxins produced by disease-causing viruses or bacteria could destroy tissues and white blood cells in circulation. Severe infection could lead to bone marrow suppression or immune dysfunction, which in turn affects the production of granulocytes, resulting in reduced white blood cell counts [15]. Hence the above factors could affect the correlation between WBC and PCT, CRP, and PTX-3 levels.
It is further observed that there were significant differences in CRP and PTX-3 levels among the three groups (P<0.05). The results of multivariate logistic regression analysis exhibited that WBC, PLT, CRP, PCT, PTX-3, and PDW were found to closely relate to neonatal infection, with PTX-3 being the most closely related to neonatal sepsis and neonatal infection. To date, numerous biomarkers are able to objectively evaluate the pathophysiological process of sepsis, such as inflammatory response, immune response, and coagulation function. These play a significant role in early diagnosis, monitoring of curative effects, and prognosis. PTX-3 is a new type of inflammatory indicator. It belongs to the same superfamily as the classic inflammatory index CRP. However, the cell origin, regulatory mechanisms, and ligands of the two are different [16]. C-reactive protein is secreted by the liver and is induced by pro-inflammatory factors such as interleukin-6. Because Neonates do not have fully developed and functional organs and hence are less sensitive to bacterial infections and thereby secrete lower levels of C-reactive protein [17]. PTX-3 is mainly induced by tissue and immune cells at the site of inflammation through tumor necrosis factor-a and interleukin-1Β (rather than IL-6) [18]. Compared to CRP, PTX-3 could reflect local tissue inflammation and injury much faster. In addition, PTX-3 levels were found to positively correlate with the severity of the inflammatory response, with PTX-3 levels gradually increasing in patients with sepsis, severe sepsis, and septic shock [19]. Hence, compared to CRP, changes in PTX-3 levels are much faster during neonatal infection and could detect neonatal infections much earlier.
ROC curve for PTX-3, PCT, CRP, and WBC showed that the combined diagnosis value of which was higher compared to any single indicator, surpporting excellent significance in clinically determine neonatal infection. Besides, PTX-3 had the highest diagnostic efficiency rather than other single indicators. When PTX-3 levels exceeded 20.36 ng/ml, it strongly suggested the presence of neonatal infection. The reflection of PTX-3 to rapid inflammatory process makes it had a better diagnostic value [20-21]. In addition, the diagnostic value of the combined indicators WBC+CRP+PTX-3 in the sepsis and local infection groups were greatest, followed by CRP among the single indicators [23]. CRP is a very stable protein both in vitro and in vivo. Only during severe or systemic infections, especially during sepsis and continuous inflammation, tissues and various cell types can produce CRP and release it into the blood, resulting in an increase in CRP levels [24]. Hence CRP is more effective for the diagnosis of neonatal sepsis due to the indicative role of elevated CRP in severe extensive infection.
Author Statements
Ethics Statement
The study was approved by the Ethics Committee of the Second Affiliated Hospital of Xi'an Jiaotong University.
Patient Consent for Publication Statement
Informed consent was obtained from the parents or guardians.
Funding
Basic research of natural science in Shaanxi Province: 2020JQ-549.
References
- Bang AT, Bang RA, Baitule SB, Reddy MH, Deshmukh MD. Effect of home-based neonatal care and management of sepsis on neonatal mortality: field trial in rural India. Lancet. 1999; 354: 1955-61.
- Procianoy RS, Silveira RC. The challenges of neonatal sepsis management. J Pediatr (Rio J). 2020; 96: 80-6.
- Weston EJ, Pondo T, Lewis MM, Martell-Cleary P, Morin C, et al. The burden of invasive early-onset neonatal sepsis in the United States, 2005-2008. Pediatr Infect Dis J. 2011; 30: 937-41.
- Kumar V, Shearer JC, Kumar A, Darmstadt GL. Neonatal hypothermia in low resource settings: a review. J Perinatol. 2009; 29: 401-12.
- Simonsen KA, Anderson-Berry AL, Delair SF, Davies HD. Early-onset neonatal sepsis. Clin Microbiol Rev. 2014; 27: 21-47.
- Guerti K, Devos H, Ieven MM, Mahieu LM. Time to positivity of neonatal blood cultures: fast and furious? J Med Microbiol. 2011; 60: 446-53.
- Liu C, Zhang Y, Shang Y, Fang C, He Q, et al. Clinical values of common biomarkers for efficacy monitoring of antibiotics in early-onset neonatal sepsis. Transl Pediatr. 2020; 9: 669-76.
- Wang SY, Yu JL. Diagnostic value of procalcitonin in neonatal early-onset sepsis. Zhongguo Dang Dai Er Ke Za Zhi. 2020; 22: 316-22.
- Cao RY, Yang J, Zheng Y, Li H, Zhao Q, Ding Y et al. The potential value of copeptin and PENTRAXIN3 for evaluating the severity of coronary stenosis in patients with coronary artery disease. Clin Biochem. 2021; 87: 32-8.
- Cui X, Zhang H, Cao A, Cao L, Hu X. Cytokine TNF-a promotes invasion and metastasis of gastric cancer by down-regulating PENTRAXIN3. J Cancer. 2020; 11: 1800-7.
- Zhang J, Koussih L, Shan L, Halayko AJ, Chen BK, et al. TNF up-regulates PENTRAXIN3 expression in human airway smooth muscle cells via JNK and ERK1/2 MAPK pathways. Allergy Asthma Clin Immunol. 2015; 11: 37.
- Hamed S, Behnes M, Pauly D, Lepiorz D, Barre M, et al. Diagnostic value of Pentraxin-3 in patients with sepsis and septic shock in accordance with latest sepsis-3 definitions. BMC Infect Dis. 2017; 17: 554.
- Professional committee on hospital infection management and control of pediatric branch of Chinese Medical Association. Expert consensus on clinical application of serum procalcitonin detection in children with infectious diseases. Zhong Hua Er Ke Za Zhi. 2019; 1: 9-15.
- Stocker M, van Herk W, El Helou S, Dutta S, Schuerman FABA, et al. C-reactive protein, procalcitonin, and white blood count to rule out neonatal early-onset sepsis within 36 hours: A secondary analysis of the neonatal procalcitonin intervention study. Clin Infect Dis. 2021; 73: e383-90.
- Crouser ED, Parrillo JE, Seymour C, Angus DC, Bicking K, et al. Improved early detection of sepsis in the ED with a novel monocyte distribution width biomarker. Chest. 2017; 152: 518-26.
- Dongel I, Gokmen AA, Gonen I, Kaya S. Pentraxin-3 and inflammatory biomarkers related to posterolateral thoracotomy in Thoracic Surgery. Pak J Med Sci. 2019; 35: 464-9.
- Quadir AF, Britton PN. Procalcitonin and C-reactive protein as biomarkers for neonatal bacterial infection. J Paediatr Child Health. 2018; 54: 695-9.
- Sjöberg B, Snaedal S, Stenvinkel P, Qureshi AR, Heimbürger O, et al. Three-month variation of plasma PENTRAXIN 3 compared with C-reactive protein, albumin and homocysteine levels in haemodialysis patients. Clin Kidney J. 2014; 7: 373-9.
- Lee YT, Gong M, Chau A, Wong WT, Bazoukis G, et al. Pentraxin-3 as a marker of sepsis severity and predictor of mortality outcomes: A systematic review and meta-analysis. J Infect. 2018; 76: 1-10.
- Fahmey SS, Pentraxin MN. 3 as a novel diagnostic marker in neonatal sepsis. J Neonatal Perinat Med. 2019; 12: 437-42.
- Hamed S, Behnes M, Pauly D, Lepiorz D, Barre M, et al. Pentraxin-3 predicts short- and mid-term mortality in patients with sepsis and septic shock during intensive care treatment. Clin Lab. 2018; 64: 999-1011.
- Macallister K, Smith-Collins A, Gillet H, Hamilton L, Davis J. Serial C-reactive protein measurements in newborn infants without evidence of early-onset infection. Neonatology. 2019; 116: 85-91.
- Ruan L, Chen GY, Liu Z, Zhao Y, Xu GY, et al. The combination of procalcitonin and C-reactive protein or presepsin alone improves the accuracy of diagnosis of neonatal sepsis: a meta-analysis and systematic review. Crit Care. 2018; 22: 316.