Abstract
Tablet coating is a not unusual pharmaceutical technique of applying a thin polymer-based film to a tablet or a granule containing Active Pharmaceutical Ingredients (APIs). Stable dosage paperwork are lined for some of reasons, the maximum crucial of that’s controlling the discharge profiles. The amount of coating at the surface of a tablet is vital to the effectiveness of the oral dosage shape. Capsules are typically coated in horizontal rotating pans with the coating answer sprayed onto the unfastened surface of the tablet mattress. The blessings of tablet coating are flavor covering, smell overlaying, bodily and chemical protection, protects the drug from the gastric surroundings and so on. There are various strategies for tablet coating which include sugar coating, film coating, and enteric coating. Latest trends in pharmaceutical technology are the development of coating techniques which overcomes the various risks associated with solvent based coatings. In those ultra-modern technologies coating substances are at once lined onto the surface of stable dosage bureaucracy without the use of any solvent. Numerous solvent less coatings are available inclusive of electrostatic dry coating, magnetically assisted impaction coating, compression coating, warm soften coating, powder coating, and supercritical fluid coating. Supercell Coating generation is a innovative tablet coating that appropriately deposits managed quantities of coating substances on capsules despite the fact that they may be extraordinarily hygroscopic or friable. Magnetically assisted impaction coating, electrostatic dry coating in solvent less coatings, aqueous film coating and Supercell coating technology also are available latest technique of coating. An ideal tablet ought to be free from any visual illness or functional defect. The improvements and innovations in tablet manufacture have now not reduced the issues, often encountered in the production, instead have accelerated the troubles, in particular due to the complexities of tablet presses; and/or the extra needs of high-quality. This review deal in detail approximately records, recent tablet coating technique and remedies associated with the tablet coating.
Keywords: Coating; History of coating; Supercell Coating; Magnetically Assisted Impaction Coating
Introduction
Tablet is a pharmaceutical solid dosage form, comprising a mixture of active substances and excipients, commonly in powder form, pressed or compacted right into a stable. Capsules Dosage form is one of a most desired dosage form everywhere in the world. Almost all drug molecules may be formulated in a tablet and procedure of producing of drugs is very simple, and may be very bendy. Coating is a manner by using which a basically dry, outer layer of coating material is implemented to the surface of a dosage shape to achieve precise benefits. Coating may be implemented to a wide range of oral solid dosage shape, along with capsules, capsules, multi particulates and drug crystals [1-3]. While coating composition is carried out to a batch of capsules in a coating pan, the tablet surfaces grow to be protected with a tacky polymeric film. Before the tablet floor dries, the carried out coating adjustments from a sticky liquid to tacky semisolid and sooner or later to a non-sticky dry surface pans. Many stable pharmaceutical dosage forms are produced with coatings, both on the external surface of the tablet, or on substances dispensed inside gelatin tablets. The tablet ought to launch the medicament steadily and the drug should be to be had for digestion. The coating procedure can be in particular formulated to adjust how speedy the tablet dissolves and in which the lively tablets are to be absorbed into the body after ingestion. The complete coating system is conducted in a chain of routinely operated acorn-formed coating pans of galvanized iron stainless-steel or copper [4-6]. The smaller pans are used for experimental, developmental, and pilot plant operations, the larger pan for industrial production.
Basic percepts involved in tablet coating
Tablet coating is the application of coating composition to moving bed of tablets with concurrent use of heated air to facilitate evaporation of solvent.
a) Solution wherein affects the release pattern as little as feasible and does not markedly exchanges the advent.
b) Modified release with specific requirement and release mechanism tailored to frame characteristic within the digestive tract.
c) Modified release with particular requirement and release mechanism tailored to body characteristic in the digestive tract.
d) Insulation is being provided by the Color coating.
e) To include every other drug or components adjuvant in the coating to keep away from Chemical incompatibilities or to provide sequential drug release.
f) To enhance the pharmaceutical elegance by means of use of unique shades and contrasting printing.
Coating process, design & control
In maximum coating methods, the coating solutions are sprayed onto the capsules because the tablets are being agitated in a pan, fluid mattress, and so forth. As the solution is being sprayed, a thin film is formed that adheres directly to every tablet. The coating may be shaped with the aid of an unmatched application or can be constructed up in layers through the use of more than one spraying cycles. Rotating coating pans are regularly used within the pharmaceutical industry. Uncoated tablets are located inside the pan and the liquid coating answer is brought into the pan even as the tablets are tumbling. The liquid part of the coating answer is then evaporated by using passing air over the surface of the tumbling drugs. In assessment, a fluid bed coater operates via passing air through a bed of tablets at a velocity sufficient to assist and separate the drugs as person gadgets. As soon as separated, the drugs are sprayed with the coating composition. The coating method is normally such as the subsequent steps:
a. Batch identification and Recipe selection
b. Loading/Dispensing
c. Warming
d. Spraying
e. Drying
f. Cooling
g. Unloading
History of Coating Technique
“Panning” turned into the unique word for the manner of adding a coating to a tablet. The word panning remains a commonplace time period that’s used in the confectionary enterprise. In past years coating carries out essentially using a rotating drum on a stand. A coating answer turned into brought, while the rotation of the pan allotted the solution during the mattress of tablets. The primary drawback of this technology was gradual looking ahead to the coating technique to dry; and the trick turned into to get it to dry evenly. With the advent of movie coating a movie or skinny membrane, normally representing 1-3 % of the whole tablet weight, became sprayed on the use of a perforated pan. To lower the overall manner time, holes had been made through the pan in order that handled air warm or cold will be pulled via the pan, just like a garments dryer, permitting the tablets to dry speedier. With this advent of stepped forward drying came the potential to replace the movie coating solution from a solvent primarily based option to a water primarily based solution [7-10].
Traditional Coating Techniques
Generally three methods are used for tablet coating:
a) Sugar Coating
b) Film Coating
Sugar coating
Steps in sugar coating:
- Seal coating
- Sub coating
- Syrup coating/Smoothing
- Color coating
- Polishing
- This involved the application of one or more coats of a water proofing substance in the form of alcoholic spray, such as pharmaceutical Shellac or synthetic polymers, such as CAP.
- Sugar-coatings are aqueous formulations which allow water to penetrate directly into the tablet core and thus potentially affecting product stability and possibly causing premature tablet disintegration.
- Large quantities of sugar-coatings are usually applied to the tablet core, typically increasing the tablet weight by 50- 100%.
- To cover and fill in the imperfections in tablet surface caused by sub coating.
- To impart desired color
- The first syrup coat contains some suspended powders and are called “grossing syrups”
- Dilute colorants can be added to provide tinted base that facilitates uniform coating in later steps.
- Syrup solutions containing the dye are applied until final size and color are achieved.
- Final syrup coating step
- Few clear coats of syrup may be applied.
- Desired luster is obtained in this final step
- Clean standard coating pan, canvas-lined coating pans
- Application of powdered wax or warm solution of waxes in suitable volatile solvent.
- Same as that of pan-pour sugar coating
- Method is relatively slow and relies heavily on skill and technique of operator
- Aqueous based film coating is not suitable due to localized over-wetting.
- Use of automated spraying system
- Immediate release
- Modified release
- Continuous coating
- Short processing time
- Flexible modular design
- No scale-up to parameters
- Production capacity of 6 cells coats 200K tph of 120 mg tablets
- R&D batch size (Minimum batch size of 30 grams)
- Enhancing technology
- Multi-layer coating
- Difficult-to-coat shapes
- Friable tablets
- “Low humidity process” suitable for moisture sensitive materials
- Enabling technology
- Accuracy of coating
- Kamble N, Chaudhari SP, Oswal RJ, Kshirsagar SS, Antre RV. Innovations in tablet coating technology. A review. International Journal of Applied Biology and Pharmaceutical Technology. 2011; 2: 214-218.
- Lachman leon et al. The Theory and Practice of Industrial Pharmacy. Second edition. Fourth Indian Reprint, Bombay: Published by Varghese Publishing house. 1991.
- Remington’s The Science and Practice of Pharmacy. Volume-I. 21st ed. Indian Edition, Lippincot Williams and Wilkins. 2005.
- Cole G, Hogan J, Aulton M. Pharmaceutical Coating Technology. Taylor and Francis. London. 1995.
- Qiao M, Zhang L, Yingliang M, Zhu J, Xiao W. A novel electrostatic dry coating process for enteric coating of tablets with Eudragit L100-55. European J Pharm Biopharm. 2013; 83: 293-300.
- Qiao M, Zhang L, Ma Y, Zhu J, Chow K. A Novel Electrostatic Dry Powder Coating Process for Pharmaceutical Dosage Forms: Immediate Release Coatings for Tablets. European J Pharm Biopharm. 2010; 3: 304-310.
- Pawar A, Deepak VB, Vineeta VK, Vilasrao JK. Advances in Pharmaceutical Coatings. International Journal of Chem Tech Research. 2010; 2: 733-737.
- Mazumder M, Sims R, Biris A, Sriramaa PK, Sainia D, Yurteri CU. Twentyfirst century research needs in electrostatic processes applied to research industry and medicine. Chem Eng Sci. 2006; 61: 2192-2211.
- Ramlakhan M, Chang Yu Wu, Satoru Watano, Rajesh N. Dave, Robert Pfeffer. Dry particle coating using magnetically assisted impaction coating: modification of surface properties and optimization of system and operating parameters. Powder Technol. 2011; 112: 137-148.
- Singh P, Solankyb TKS, Mudryya R, Pfefferc R, Dave R. Estimation of Coating Time in the Magnetically Assisted Impaction Coating Process. Elsevier. 2001; 11: 159-167.
- Lachman L, Lieberman HA, Joseph LK. The Theory and Practice of Industrial Pharmacy. Varghese Publishing House; Mumbai; Third Edition: 297-321.
- Lachman L, Liberman H, Kanig J. The Theory and Practice of Industrial Pharmacy; Third Edition: 293-345: 346-373.
- Aulton M. Pharmaceutics: The Science of Dosage Form Design. International Student Edition: 304-321: 347-668.
- Vyas S, Khar R. Controlled Drug Delivery Concepts and Advances; First Edition: 219-256.
- Ansel H, Allen L, Popovich N. Ansel’s Pharmaceutical Dosage Forms and Drug Delivery Systems; Eighth Edition: 227-259.
- Remington J. Remington: The Science and Practice of Pharmacy; 2: 1615- 1641.
- American Pharmaceutical Reviews. 2001; 4: 28-35.
- Vinay V, Sivakumar T, Tamizhmani T. Colon targeting drug delivery system: A review on recent approaches. International Journal of Pharmaceutical and Biomedical Science. 2011; 2: 11-19.
- Anil K. Philip, Betty Philip. Colon Targeted Drug Delivery Systems: A Review on Primary and Novel Approaches. Oman Medical Journal. 2010; 25: 70-78.
- Raju D, Padmavathy J, Sai Saraswathi V, Saravanan D, Aparna Lakshmi I. Formulation and development of enteric coated tablets of prednisolone as a colon targeted drug delivery. IJPSR. 2011; 2: 685-690.
- Zaid AN, Qaddomi A. Development and stability evaluation of enteric coated diclofenac sodium tablets using sureteric. Pak. J. Pharm. Sci. 2012; 25: 59- 64.
Sealing (Waterproofing):
Sub coating:
Smoothing/syrup coating:
Finishing:
Polishing:
The above characteristics of Sugar Coating are depicted in Table 1.
Type
CHARACTERISTIC
SUGAR COATING
Tablet
Appearance
Rounded with high degree of polish
Weight increase because of coating
30-50%
Logo or ‘break lines’
Not possible
Process
Operator training required
Considerable
Adaptability to GMP
Difficulty may arise
Process stages
Multistage process
Table 1: Characteristic of Sugar Coating.
Film coating
Film coating and sugar coating shares the same equipments and process parameters.
Two methods,
Pan-Pour method:
Pan-Spray method:
Types of film coating:
Film coating formulation (Composition of the coating liquid)
Polymer/Film Former: A film former capable of producing smooth thin films reproducible under the prescribed coating conditions.
Classified as
1. Non enteric materials
E.g. HPMC, MHEC, EC, HPC, Povidone, SCMC, PEG, Acrylate Polymers
2. Enteric materials
E.g. CAP, Acrylate Polymers, HPMCP, PVAP.
The above characteristics of film Coating are depicted in Table 2 & Table 3.
Type
CHARACTERISTIC
FILM COATING
Tablet
Appearance
Retain contour of original core.
Weight increase because of coating material
Usually not as shiny as sugar coat type
2-3%
Process
Logo or ‘break lines’
Possible
Operator training required
Process tends itself to automation and easy training of operator
Adaptability to GMP
High
Process stages
Usually single stage
Functional coatings
Easily adaptable for controlled release
Table 2: Characteristic of Film Coating.
S.No
Material
Type
Uses
Examples
1
Film Former
Enteric
Non Enteric
To Control the release of drug
HPMC, MHEC
2
Solvents
------------
To dissolve the polymers
IPA and Methylene chloride
3
Plasticizer
Internal plasticizing
It Pertains to the chemical modification of the basic polymer that alters the physical properties
Glycerol, PEG 2000-6000
External plasticizing
It incorporated with the primary polymeric film former, changes the flexibility, tensile strength of the film
Diethyl Phthalate (DEP), Dibutyl Phthalate (DBP) and Tributyl citrate
4
Colorants
In organic materials
For light shade: concentration of less than 0.01% may be used
Iron Oxides
5
Opaquant extenders
-----------
Formulations to provide more pastel colours and increase film coverage
Titanium dioxide, silicate (talc & aluminium silicates),
Table 3: Materials used in Film coating.
Recent Trends in Tablet Coating Techniques
Different methods of coating
Compression coating
These sorts of tablet have elements like, inner core and surrounding coat. The core is small porous tablet and organized on one turret. For preparing final tablet, a bigger die hollow space in every other turret is used wherein first the coat cloth is crammed to half of after which center tablet is robotically transferred, again the last space is filled with coat fabric and ultimately compression force is applied. Often, the coat is water soluble and disintegrates without difficulty after swallowing.
Preparation of the compression-coated: A carefully weighed quantity of powder mixture was positioned inside the die and compressed on a Carver Press (Wabash, IN) at a acknowledged force with the tooling proven to produce a cup-fashioned tablet. The cup changed into left in the die, and a recognized quantity of both a model drug and a mix containing the drug changed into located inside the cup and tamped gently with the punch in a prolonged role. A weighed amount of the coating combo changed into positioned on top of the die contents, and the cup changed into compressed for a second time with the punch in a retracted position at a regarded pressure to provide the final compression-coated tablets.
Electro static coating
In electrostatic powder coating, we spray powder via a place of strong electric area and excessive unfastened-ion concentration. Passing through this location, the particles are charged as discussed earlier. The process of powder particles charging within the electric area of corona discharge is governed with the aid of Pauthenier’s equation. Charging is most strongly laid low with field power, powder particle size and form, and the length of time the particle spends within the charge area [11-13].
Electrostatic dry coating: An electrostatic dry powder coating method for tablets become evolved for the first time with the aid of electrostatic dry powder coating in a pan coater gadget. The optimized dry powder coating method produces capsules with smooth surface, correct coating uniformity and release profile that are corresponding to that of the tablet cores. This novel electrostatic dry powder coating technique is an opportunity to aqueous or solvent based coating technique for pharmaceutical products is represented in Figure 2.
Figure 1: Represents the compression coating.
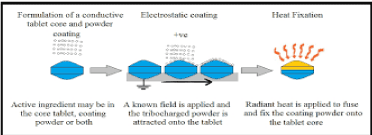
Figure 2: Represents the electrostatic coating.
The electrostatic coating method is extensively useful in paint technology, food technology, coating of residing cells, metal coatings and coating of capsules in addition to drugs. The precept of electrostatic powder coating states that spraying of a mixture of finely grounded particles and polymers onto a substrate floor without the usage of any solvent and then heating the substrate for curing on oven until the powder mixture is fused into film.
According to the charging mechanism, there are two types of spraying units:
a) Corona charging
b) Tribo charging.
a. Mechanism of corona charging: On this mechanism, the electrical breakdown and ionization of air with the aid of enforcing high voltage on a sharp pointed needle like electrode at the hole of the gun. The powder debris picks up the negative ions on their manner from the gun to the substrate. The movement of particles between the substrate and the charging gun is performed by way of the aggregate of electrical and mechanical forces. The mechanical forces generated by using the air blow the powder closer to the substrate from the spray gun. The electrical forces are derived from the electrical subject between the earthen substance and the charging tip of the spray gun, and from the repulsive forces between the charged particles. The electrical discipline may be adjusted to direct the control sample size, shape, powder’s drift, and powder density as it is released from the gun.
b. Mechanism of tribo charging: In the tribo charging, it makes using the principle of friction charging associated with the dielectric properties of solid substances and so that no loose ions and electric area might be present between the spray gun and levelheaded substance. For tribo charging guns, the electric forces are only regarded to the repulsive forces among the charged particles. After spraying, charged particles come into the distance adjoining to the substrate and the attractive forces among the grounded substrate and the charged particles makes the particle to deposit on the substrate.
Magnetically assisted impaction coating (MAIC)
A method is advanced for estimating the coating time in a magnetically assisted impaction coating tool. The combination of the host, guest and magnetic particles is assumed to stay in a fluidized state in which the distribution of velocities is Maxwell-Boltzmann kind. It is assumed that the collisions happens among the particles are critical for impinging the guest debris onto the host debris, and for that reason forming a semi-permanent coating on the surface of host debris. The coating time is depend upon several parameters, consisting of the quantity density of host substances, the diameter ratio of the host and visitor debris, the height of the fluidized particle bed and the properties of materials of the host and guest particles Figure 3 represents the MAIC [14-16].
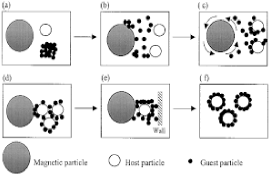
Figure 3: Represents the MAIC.
Mechanism of coating in the MAIC process:
Stage-I: Excitation of magnetic particles.
Stage-II: De-agglomeration of guest particles.
Stage-III: Shearing and spreading of guest particles on the surface of the host particles.
Stage-V: Magnetic-host-host particle interaction.
Stage-VI: Magnetic-host-wall interaction and
Stage-VI: Formation of coated products
Aqueous film coating technology
The sugar coating procedure could be very consuming and its miles depending at the abilities of coating operator, this approach has been replaced through advancement in film coating technology. This approach was begun with use of organic solvents like methylene.
Super cell coating technology
Supercell coating technology is an innovative tablet coating that accurately deposits controlled quantity of coating materials on drugs even supposing they’re extremely hygroscopic or friable. Due to the fact the tablets are loaded in huge rotating pans and vented for decent air drying, edges of tablets can get grounded off, intagliations can get filled in via coating material, and edges and corners may not be covered with identical thickness as the tablet faces in Figure 4 represents the super cell coating technology equipment.
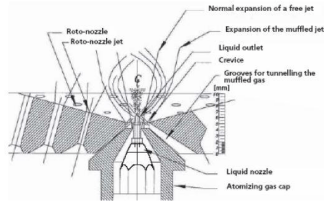
Figure 4: Represents the super cell coating technology equipment.
Unique functions of super cell coating technology
Dip coating
Dip coating is a famous way of creating thin films for research functions. Uniform films may be implemented onto flat or cylindrical substrates. For commercial strategies, spin coating is used greater often in Figure 5 represents the dip coating [17].
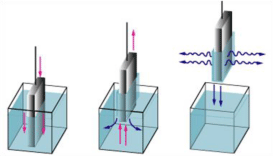
Figure 5: Represents the dip coating.
The dip coating process can be separated into five stages:
a) Immersion: The substrate is immersed inside the solution of the coating material at a constant pace.
b) Start-up: The substrate has remained inside the solution for a while and is starting to be pulled up.
c) Deposition: The thin layer deposits itself at the substrate even as it is pulled up. The retreating is carried out at a constant velocity to avoid any jitters. The speed determines the thickness of the coating.
d) Drainage: Extra liquid will drain from the surface.
e) Evaporation: The solvent evaporates from the liquid, forming the thin layer. For volatile solvents.
Polymer blends
Blends of GIT-insoluble polymers: Many GIT-insoluble polymers used for controlled drug delivery are derivatives of cellulose or acrylic acid, as an example ethyl cellulose and poly (ethyl acrylateco- methyl methacrylateco- trimethyl ammonioethyl methacrylate chloride) 1:2:0. (Eudragit® RS). Mixed aqueous dispersions of two acrylate: methacrylate copolymers (Eudragit® RL and Eudragit® RS) and studied the results of the blend ratio at the ensuing permeability of unfastened movies for Chlorpheniramine-maleate and phenylpropanolamine-HCl. As the two polymers incorporate unique quantities of quaternary ammonium corporations, they do not have interaction in the equal manner with water and drug molecules, resulting in extraordinary mobilities for water and drug inside the polymeric networks. As it is able to be seen in, the permeability of each tablets significantly increases with growing Eudragit® RL content material. As the latter carries greater quaternary ammonium agencies than Eudragit® RS, it’s miles extra hydrophilic and, thus, takes up extra water. Therefore, the polymer chain mobility will increase with increasing Eudragit® RL content, ensuing in increased drug mobilities inside the polymeric network. Within the case of phenylpropanolamine-HCl, the permeability in pure Eudragit® RL is 8-fold higher than in natural Eudragit® RS, whereas for chlorphenamine-maleate the mobility differs by means of a element of three. From a realistic point of view, the permeability of each tablet inside the macromolecular networks can successfully be adjusted over a huge range through honestly various the polymer blend ratio. For this reason, it can be predicted that the drug launch styles from dosage forms coated with this type of polymer blends can be effortlessly altered [18-21].
Blends of GIT-insoluble and GIT-soluble polymers: Ethyl cellulose is a frequently used GIT-insoluble polymer in controlled drug delivery systems. But, if carried out as a film coating mater flawlessly shaped membranes result in very low drug launch rates because ethyl cellulose is poorly permeable for maximum drugs. To triumph over this restriction, water-soluble polymers can be added to ethyl cellulose coatings.
For instance, Polyethylene Glycol (PEG), Polyvinyl Pyrrolidone (PVP) and hydrophilic cellulose ethers, which include Hydroxypropyl Methylcellulose (HPMC), were proposed. Upon touch with aqueous media, those components hydrate and probably leach out from the polymeric membranes, ensuing in more permeable films and increased drug release rates. In assessment to low molecular weight water watersoluble additives, those polymers are typically no longer considered as “true pore-formers”, because they do frequently no longer completely leach out from the coatings, and do not create properly-described porous structures. The up to now maximum frequently used watersoluble, macromolecular additive for ethyl cellulose-based totally film coatings is Hydroxypropyl Methylcellulose (HPMC).
Blends of GIT-insoluble and enteric polymers: Blends of GITinsoluble and enteric polymers are specifically exciting for the coating of solid dosage bureaucracy, because they could offer drug release profiles which might be triggered by using the pH of the surrounding surroundings alongside the GIT. Inside the stomach, each polymer is insoluble, while within the gut, the enteric polymer is soluble and can leach out from the coatings. This could result in large, dynamic changes in the physicochemical houses of the films at some point of the GIT transit and, as a result, to altered drug release kinetics.
Thrilling application of this kind of pH-sensitive coating substances is the possibility to render the discharge of weakly simple pills showing strongly pH-structured solubility pH-unbiased. The concept is on the way to compensate the decrease in drug solubility along the GIT by a simultaneous boom in drug permeability.
A novel method of manufacturing One-Step Dry-Coated (OSDrC) tablets
Diverse studies concerning controlled release shipping systems were performed for oral dosage forms. A dry-coated tablet is one of the most useful strong dosage paperwork that can be used for such managed launch transport systems with a sustained release pattern or a time programmed pulsatile release sample in OSDrC making manner.
Dry-coated tablets encompass a core and an outer layer. The outer layer absolutely surrounds the center tablet and, consequently, the choice of the materials for the outer layer significantly impacts the release pattern, since it relies upon on the discharge charge of the drug and the physical properties of the dry-coated tablets. Cellulose derivatives such as Hydroxypropyl Cellulose (HPC), Hydroxypropyl Methylcellulose (HPMC), Carboxymethyl Cellulose (CMC) or Hydroxyethyl Cellulose (HEC) are frequently used as outer layer materials because they’ve water soluble, gel forming and swelling traits for behind schedule drug launch pattern tablets.
This difficult to produce tablets that have a steady fixed outer layer thickness the use of industrial scale manufacturing. The conventional manufacturing approach is as follows
(1) Fill the inside of the die with the powder for the outer layer,
(2) Place the core tablet on the powder for the outer layer,
(3) Surround the core tablet with the powder for the outer layer
and
(4) Compress the powder which has the core tablet inside. This conventional manufacturing method, however, has a problem stemming from the setting of the core tablet supply system, i.e. the core tablet is not always located in the center of the tablet, to giving various thicknesses of the outer layer.
Vacuum film coating: (Fluid Bed Coating)
Spray coating can be used for all fluid bed systems, be it in batch or continuous operation or if the film is applied from a sprayed solution, suspension or hot melt. For this processing option the parameters have to be chosen to avoid agglomeration, i.e. liquid bridges between the air suspended particles. If spraying a solution or suspension the liquid only serves as a vehicle to deliver the coating material to the surface of the substrate. For hot melt coating the droplets must be small enough not to form solid bridges in Figure 7 represents vacuum film coating process.

Figure 6: Represents OSDrC making process.
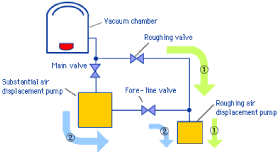
Figure 7: Represents vacuum film coating process.
Top spray coating process
Despite the fact that the top spray fluid mattress system is extensively used for granulation its use for coating is restrained. Top spraying is the most effective method and gives the best capability and lowest capital cost. It could be used for numerous manipulations of taste covering and many others. This method alternative is often used in the food, feed and chemical industries as the characteristic of the film mainly serves to enhance the general managing situations. An ideal film is typically no longer required for this function, however care should be taken that the droplets do not emerge as too viscous before touching the substrate, so that you can hold accurate spread ability.
Bottom spray coating process
The maximum normally recognized fluid-bed system for coating inside the pharmaceutical industry is bottom-spray technique. The precise functions of bottom-spraying are an air distributes plate and a partition that arrange fluidization of particles of debris via the partition. The nozzle is installed at the bottom of the product box and is focused in the coating zone. The quick distance between the coating materials and particles at some point of the coating technique minimizes spray drying and contributes to excessive uniformity and coating performance may be seen in Figure 9 represents bottom spray coating process.
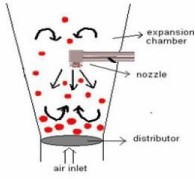
Figure 8: Represents top spray coating process.
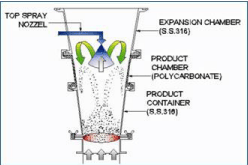
Figure 9: Represents bottom spray coating process.
Tagential spray coating process
This processing approach is with its physical concepts pretty much like bottom-spray coating handiest that the production motion is supplied by a motor driven rotor disc. Otherwise, the quality producing parameters are the same:
1. Uniform statistical residence time is warranted by defined rotor revolution speed.
2. The coating material is sprayed simultaneously within the rotating product.
3. The rolling movement of the particles affords an even higher separation force, as such preventing agglomeration.
The benefits of this processing option are mainly for the layering and subsequent film coating of pellets in Figure 10 represents spray coating process.
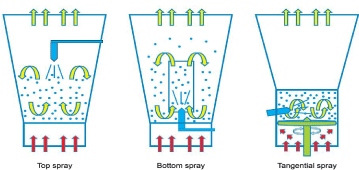
Figure 10: Represents spray coating process.
Time dependent and pH dependent polymer for colon targeted
The pH-dependent systems exploit the generally accepted view that pH of the human gastrointestinal increases progressively from the stomach pH 2-3, small intestine pH 6.5-7 to the colon 7.0-8.0. Taking the advantage of highest pH value of the colon content, the dosage form containing the active drug in a core is coated with pHdependent material which dissolves at the pH of the colon in Figure 11.
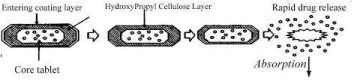
Figure 11: Represents Time Dependent and pH Dependent Polymer for
Colon.
Tablet Coating Defects
An ideal tablet should be free from any visual defect/ functional defect. The improvements and innovations in tablet manufacture have now not reduced the troubles, often encountered inside the production, rather have elevated the issues, in particular due to the complexities of tablet presses. During manufacturing an industrial pharmacist usually encounters number of problems. Majority of visual defects are due to inadequate fines, inadequate moisture in the granules ready for compression and due to faulty machine setting. Due to faulty formulation there are functional defects. Solving many of the manufacturing problems requires an in-depth knowledge of granulation processing and tablet presses, and is acquired only through an exhaustive study and a rich experience. Here, we will discuss the imperfections found in tablets along-with their causes and related remedies are depicted in table 4. The imperfections are known as: ‘VISUAL DEFECTS’ and they are either related to imperfections in anyone or more of the following factors.
S.NO
Tablet defects
Cause
Remedies
1
Blistering
Entrapment of gases in the film due to overheating during spraying.
Milder drying conditions are warranted in this case.
2
Chipping
Decrease in fluidizing air
Be careful not to over dry the tablets in the pre-heating stage.
3
Picking
It creates overly wet bed where adjacent tablets stick together and break apart
Increase in the drying air temperature and air volume
4
Twinning
Common problem
Increase the pan speed
5
Pitting
Temperature of the tablet core is greater than the melting point of the materials used in the formulation
Control of temperature of tablet core
6
Cratering
Coating solution penetrates the surface of the tablet
-------
7
Blooming
Mostly due to plasticizer
-----------------
8
Color variation
Alteration of the frequency and duration of the spray zone
A best way is to reformulate with different plasticizers
9
Cracking
Internal stress in the film exceeds tensile strength of the film
Use of high molecular weight polymer blends
10
Orange peeling
Inadequate spreading of the coating solution
Thinning the solution with additional solvent
11
Mottling
Degradation of the product
Prepare coating solution properly
Table 4: Tablet Coating defects cause and remedies.
Conclusion
In latest decades, coating of pharmaceutical dosage bureaucracy has been concern of superb developmental efforts aiming to make certain and enhance the nice of tablet dosage form. Magnetically assisted impaction coating and electrostatic dry coating avoids most important hazards of solvents primarily based coating. Techniques produce uniform coating however simplest with specialized instrumentation. Electrostatic dry coating calls for special type of powder coating composition. Electrostatic dry coating enables coating of tablet with different colors on either facet alongside-with printing on tablet on pharmaceutical dosage shape. Protection factors of those coatings in people remains to be unveiled for that reasons similarly studies in fitness and protection factors of those technologies will make sure the commercialization of those technologies in pharmaceutical enterprise. Enhancements regarding particle motion, warmness and power switch, film distribution, drying performance and non-stop processing have contributed to seriously develop this technology. However evaluation and fulfillment of in addition constructional enhancements in coating strategies appear to depend on correct analytical tools and advanced strategies for method modeling and control. In this regard, achieving most effective production efficiency and excessive product great nevertheless stays a major challenge for future research.
References
