
Research Article
Austin J Pharmacol Ther. 2016; 4(2).1083.
Effect of Hydroalcoholic Extract of Centella Asiatica and its Synergy with N–Acetyl Cysteine on Marble– Burying Behavior in Mice: Implications for Obsessive– Compulsive Disorder
Shivani Sharma1 and Girdhari Lal Gupta2*
1Department of Pharmacy, Jaypee University of Information Technology, India
2Department of Pharmacology, School of Pharmacy and Technology Management, India
*Corresponding author: Girdhari Lal Gupta, Department of Pharmacology, School of Pharmacy and Technology Management, SVKM’s NMIMS University, V. L. Mehta Road, Vile Parle (W), Mumbai - 400056, Maharashtra, India
Received: May 25, 2016; Accepted: July 18, 2016; Published: July 28, 2016
Abstract
Traditionally, Centella asiatica Lin. (CA) whole plant is used as anti– compulsive, memory enhancing, anxiolytic and antidepressant effects. However, no scientific validation is available on the anti–compulsive potential of the plant. To reduce the doses of CA and N–acetyl cysteine combination study was also carried out to target two different neurotransmitters. This study was carried out to examine the effect of CA whole plant hydroalcoholic extract and its combination with N–acetyl cysteine on marble–burying behavior test. Mice from different groups were administered with vehicle, CA (50, 100, 200 or 500 mg/kg), N–Acetyl–L–Cysteine (NAC) (25, 50, 100 or 200 mg/kg) and fluoxetine (5, 10 or 20 mg/kg). One hour after CA and thirty minutes after NAC or fluoxetine administration, individual mice was subjected to marble burying test for thirty minutes and actophotometer for ten minutes. CA (200 and 500 mg/kg, p.o.), NAC (100 and 200 mg/kg, p.o.) and fluoxetine (10 and 20 mg/ kg, p.o.) significantly inhibited marble–burying behavior in mice. Moreover, in separate experiments, co–treatment with sub–effective dose of both CA (50 or 100 mg/kg, p.o.) and NAC (25 or 50 mg/kg, p.o.) significantly decreased marble–burying behavior at doses, which either of these alone could not protect the behavior. All dose levels of CA, NAC and fluoxetine showed insignificant changes in locomotor activity in actophotometer. Our results reveal that Centella asiatica possesses anti–compulsive activity and co–administration of CA with nutraceutical N–acetyl cysteine also show synergistic effects in reducing obsessive–compulsive behavior in mice.
Keywords: Centella asiatica; Fluoxetine; Obsessive–compulsive disorder
Introduction
Obsessive–compulsive disorder (OCD) is a mental disorder characterized by persistent intrusive thoughts (obsessions) and the expression of ritualistic repetitive behaviors (compulsion). The patient spends greater than one hour time each day in symptoms like doubting, checking, washing associated with OCD [1]. The World Health Organization has identified OCD as one of the top 10 disabling illnesses by lost income and decreased quality of life [2]. Financial cost associated with health care in the USA estimated to be around $10.6 billion per annum [3]. Lifetime prevalence in India and USA has been estimated 0.6% and 2–3% respectively [4,5]. The pathogenesis of OCD involved glutamatergic and serotonergic pathway dysregulation in orbito frontal cortex, anterior cingulate cortex and caudate nucleus [6]. Treatment of OCD with first–line selective serotonin reuptake inhibitors have also been reported with limited benefit and associated side effects [7]. Therefore, it is always desired worldwide to opt for safer and effective nutraceuticals [8] and plant–derived anti-compulsives mentioned in the traditional medical systems.
Centella asiatica Lin. (Umbelliferae/Apiaceae family) is a perennial creeping herb. Since ancient times, it has been used as anti–OCD, memory enhancing, anxiolytic, antidepressant, strength promoting, wound healing, immune booster, anti–epileptic and anti–stress herbal medicine. However, no scientific validation is available on the anti– compulsive potential of the plant. The main active principles of CA are the triterpenoids glycosides such as asiaticoside, madecassoside (asiaticoside A) and asiatic acid [9,10]. Moreover, total triterpenes of CA has already been reported to increase serotonin contents in rat brain [9], which was also taken as the strategy for selection of the plant first time to reverse the depletion of serotonin content in OCD.
N–Acetyl–L–Cysteine (NAC) has been evaluated in a diversity of neuropsychiatric disorders, including OCD, schizophrenia and bipolar disorder and appears to have evidence of both safety and tolerability. NAC has been reported for attenuating glutamatergic neurotransmission [11]. NAC augmentation in SSRI treatment– resistant obsessive–compulsive disorder has also been reported. Therefore, the present study was undertaken to investigate the effects of CA alone and its combinations with NAC to target two different neurotransmitter system in marble–burying behavior of mice– a well–accepted model of obsessive–compulsive disorder.
Materials and Methods
Plant material
Dried whole plant of Centella asiatica (CA) was purchased from Natural Remedies, Bangalore (Batch Number: ERD–40). The plant was coarsely powdered with a mechanical grinder and subjected to defatting with petroleum ether (60°–80 °C). The defatted air dried powder was extracted by Soxhlet apparatus using 70% v/v of ethanol at 50 °C for 48 hours. The solvent was evaporated under reduced pressure by rotary evaporator (Heidolph, Germany) to obtain a semisolid mass, which was further lyophilized (New Brunswick) for 24 hours. The obtained dry powder was stored at –20 °C till further use. The percentage extractive value was also calculated in terms of air–dried weight of plant material.
Chemicals
For standardization of CA extract, active marker compounds like asiatic acid, asiaticoside and madecassoside plus asiaticoside B were purchased from Natural Remedies, Bangalore. Parachlorophenylalanine, NAC, HPLC grade phosphoric acid and acetonitrile were purchased from Sigma Aldrich, USA. Fluoxetine was obtained from Cadila Pharmaceuticals Limited, India.
Standardization of CA extract by high performance liquid chromatography (HPLC)
Whole plant hydroalcoholic extract of CA was standardized for their triterpene composition like asiatic acid, asiaticoside and madecassoside plus asiaticoside B by high–performance liquid chromatography [10]. Each triterpene marker compound asiatic acid, madecassoside plus asiaticoside B and asiaticoside were diluted with HPLC grade methanol to obtain concentration of 200 μg/ml. HPLC system (Shimadzu, LC 2010 CHT), photodiode array detector (Shimadzu), Phenomenex– Luna 5μm (250 mm × 4.6 mm) C18 column and a personal computer with Empower software were used for analysis at detection wavelength 210 nm. The flow rate 1.5 ml/min of mobile phase having 0.05% phosphoric acid and 100% acetonitrile was maintained during separation. For sample preparation, hydroalcoholic CA extract 200 mg was dissolved in 50 ml of methanol by ultrasonic. Marker compounds and sample were filtered through a 0.45 μm nylon membrane filter paper thereafter three injections of 20 μl were performed. The standard curve was prepared by plotting peak area against concentration of marker compounds and concentrations of triterpenes in the sample were estimated from the standard curve.
Animal
Swiss male albino mice weighing 25 to 30 g were housed separately in groups of five per cage under controlled light (12:12 hrs light: dark cycle), relative humidity of 75 ± 5% and temperature (25 ± 2 °C) environment and behavioral assessment was conducted during the light cycle. Water and basal diet (Ashirwad, Chandigarh, India) were provided ad libitum. All procedures were carried out after due approval and under strict compliance with ethical principles and guidelines of the Institutional Animal Ethical Committee constituted as per CPCSEA (1716/PO/a/13/CPCSEA). All the experiments were carried out in a noise free room. Separate groups (n=6) were used for each set of experiment.
Preparation of test drugs
Parachlorophenylalanine (PCPA)–a serotonin depleting agent, was dissolved in 0.9% w/v sodium chloride. CA was suspended in 2% w/v of gum acacia in distilled water. The stock solution contained 100 mg/ml of CA. NAC and fluoxetine were dissolved in distilled water. PCPA was administered by the intraperitoneal route. All drugs except PCPA were administered via the oral route.
Selection of dose
The doses of CA extract, NAC were determined from previous studies and based on our another acute toxicity study, which were carried out according to Organisation for Economic Co–operation and Development (OECD) test guidelines 423 (Unpublished data). The hydroalcoholic extract of CA and NAC were found to be safe up to 2000 mg/kg. There was no mortality of animals in any of the groups used in different experimental design (data not shown).
Marble–burying behavior test
The anti–compulsive effect was assessed by the marble–burying behavior due to high predictive and good face validity. In brief, twenty small marbles of glass (diameter 10 mm), were arranged on the bedding evenly spaced in four rows of five. Individual mouse from different group was placed in separate plastic cages (21×38×14 cm) containing 5 cm thick sawdust bedding. After 30 min exposure to the marbles, mice were removed, and unburied marbles were counted. A marble was considered ‘buried’ if its two–third size was covered with sawdust. The total number of marbles buried was considered as an index of obsessive–compulsive behavior [12,13].
Locomotor activity tests
Motor activity was also assessed in all groups of mice using actophotometer (Techno, Lukhnow) for the duration of 10 min. Actophotometer operates on photoelectric cells that are connected in circuit with a digital counter. A count is recorded when the movement of animal cuts off the beam of light falling on the photocell. Every time the central arena was wiped out with the wet cotton and removed the fecal pellets [12].
Experimental design
Each experimental group had a separate set of mice being allotted randomly. All subjects were experimentally naïve at the beginning of each study. In all groups, mice were pretreated with PCPA– a serotonin depleting agent, once daily at a dose of 300 mg/kg for three consecutive days and 24h thereafter all drug treatment was given. Mice from different groups were administered with vehicle (2% w/v gum acacia in distilled water, p.o., n=6), CA (50, 100, 200 or 500 mg/ kg, p.o., n=6 per group), NAC (25, 50, 100 or 200 mg/kg, p.o., n=6 per group) and fluoxetine (5, 10, 20 mg/kg, p.o., n=6 per group). One hour after CA and thirty minutes after NAC or fluoxetine administration, individual mouse was subjected to marble burying test for thirty minutes and in actophotometer for ten minutes. In combination studies, CA (50 or 100 mg/kg, p.o., n=6 per group) was administered 30 min prior to NAC (25 or 50 mg/kg, p.o., n=6 per group) and 30 min thereafter individual mouse was subjected to marble burying test and actophotometer test. The doses of PCPA and fluoxetine were based on previous reports [14].
Statistical analysis
All data were expressed as mean ± SEM (n=6) and analyzed by one–way analysis of variance (ANOVA), followed by post hoc Student Newman–Keuls test. The groups treated with extract, NAC and fluoxetine were compared with vehicle group. The groups treated with combination of CA and NAC were compared with the respective CA group. Differences were considered to be significant at P<0.05.
Results
The percentage yield of the hydroalcoholic CA extract was 24% w/w. Figure 1 shows the HPLC chromatograms of CA extract (A) and marker compounds (B). On HPLC analysis for quality assurance, whole plant hydroalcoholic extract of CA showed the presence of asiaticoside (6.3% w/w), madecassoside plus asiaticoside B (6.1% w/w) and asiatic acid (2.2 % w/w).
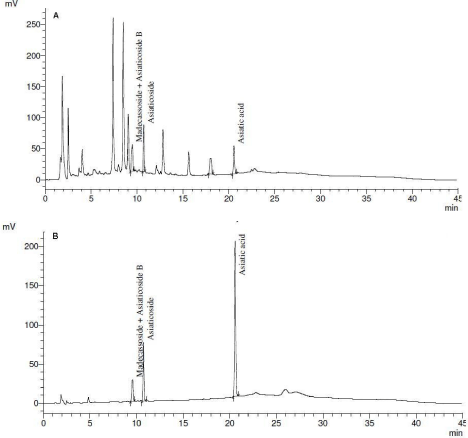
Figure 1: HPLC chromatograms of Centella asiatica extract (A) and marker
compounds (B).
Effects of NAC, fluoxetine on marble–burying behavior and locomotor activity
NAC (100 and 200 mg/kg, p.o.) [F (4, 29) =9.276, P<0.0001] and fluoxetine (10 and 20 mg/kg, p.o.)[F (3, 23) =50.447, P<0.0001] – a drug used in the treatment of obsessive–compulsive disorder, significantly inhibited marble–burying behavior in mice, without any effect on motor activity [F (4, 29) =2.428, P=0.0743 and F (3, 23) =0.8108, P=0.5208] respectively. The lower dose of NAC (25 or 50 mg/kg, p.o.) and fluoxetine (5 mg/kg, p.o.) were found to be insignificant (P>0.05) anticompulsive effects (Table 1).
Treatment (mg/kg, p.o.)
Number of marble buried
Number of locomotion count
Vehicle
15.00 ± 0.8165
462.66 ± 54.618
NAC (25)
12.167±1.352
386.50±13.691
NAC (50)
11.50 ± 0.9916
322.00±35.415
NAC (100)
9.167±0.5426*
363.16±31.916
NAC (200)
6.667±1.256**
341.66±25.892
Fluoxetine (5)
12.833±0.7491
390.50±16.988
Fluoxetine (10)
5.167±1.078**
385.50±42.503
Fluoxetine (20)
2.333±0.7419**
441.66±45.559
All values are Mean ± SEM; n= 6 in each group; *P<0.01**P<0.001as compared with vehicle control, Student–Newman–Keuls test. NAC –N–acetyl cysteine
Table 1: Effects of NAC, fluoxetine on marble burying behavior and locomotor activity in mice.
Effects of CA and combination of CA with NAC on marble– burying behavior and locomotor activity
Centella asiatica (200 and 500 mg/kg, p.o.) [F (4, 29) =14.080, P<0.0001] significantly inhibited marble–burying behavior in mice, without any effect on motor activity [F (4, 29) =0.4085, P=0.8008]. The lower dose of CA (50 or 100 mg/kg, p.o.) was found to be insignificant (P>0.05) (Table 2). Moreover, in separate experiments, co–administration of CA (50 or 100 mg/kg, p.o.), 30 min prior to NAC (25 mg/kg, p.o.), significantly reduced marble–burying behavior at doses, which either of these alone could not protect the behavior [F (3, 23) =34.918, P<0.001, F (3, 23) =33.488, P<0.0001] respectively. Insignificant change in the locomotor activity were observed irrespective of the treatment CA or NAC [F (3, 23) =1.493, P=0.246, F (3, 23) =2.656, P=0.0763] as compared to the vehicle treated group (Table 2).
Treatment (mg/kg, p.o.)
Number of marble buried
Number of locomotion count
Vehicle
15.00 ± 0.8165
462.66 ± 54.618
CA (50)
13.33 ± 1.160
398.83±30.198
CA (100)
12.167 ± 1.138
460.50±57.692
CA (200)
8.667 ± 0.8208**
441.50 ± 54.432
CA (500)
5.833 ± 1.014**
407.16 ± 25.347
CA (50) + NAC (25)
2.167±0.3073#
358.16±33.822
CA (100) + NAC (25)
1.500 ±0.6708#
313.50±33.117
CA (50) + NAC (50)
1.333±0.4944#
375.50±26.678
CA (100) + NAC (50)
3.333±0.4216#
438.83±41.234
All values are Mean ± SEM; n= 6 in each group; **P<0.001as compared with vehicle, #P<0.001 as compared with respective CA control, Student–Newman–Keuls test. CA–Centella asiatica; NAC –N–acetyl cysteine
Table 2: Effects of CA and combination of CA with NAC on marble burying behavior and locomotor activity in mice.
In another group treated with CA (50 or 100 mg/kg, p.o.), 30 min prior to NAC (50 mg/kg, p.o.), also significantly decreased marble–burying behavior at doses, which either of these alone could not protect the behavior [F (3, 23) =48.056, P<0.0001, F (3, 23) =32.195, P<0.0001] respectively. The locomotor activity did not change significantly as compared to corresponding CA given alone in actophotometer [F (3, 23) =2.319, P=0.1063, F (3, 23) =1.932, P=0.1570] (Table 2).
Discussion
This is the first report to reveal that CA exhibit anti–compulsive effect by inhibiting marble–burying behavior and it was comparable to standard drug fluoxetine. Co–administration of CA with NAC also showed synergistic effects in reducing compulsive disorder in mice.
The plant CA was quantitatively standardized for known active components like asiaticoside, madecassoside plus asiaticoside B and asiatic acid by HPLC because triterpene components in CA are not always same due to diverse environmental conditions, location and accessions.
In this study, fluoxetine (as a positive control) inhibited marble– burying behavior without affecting locomotor activity. The observed anti–compulsive effect of fluoxetine is also well in accordance with earlier reports [14]. Pharmacological treatment of OCD is targeted primarily at monoaminergic neurotransmission, particularly at the serotonin and glutamate systems. The selective serotonin reuptake inhibitors (SSRIs) are the mainstay of pharmacotherapy and are of benefit in 50–60% of patients [15]. The older tricyclic antidepressant clomipramine is commonly prescribed and maybe slightly more effective, though its higher side effect burden can limit its use. In disease refractory to these agents, needs additionally antiglutamatergic agents [16], suggesting that multiple neuronal systems involve in the pathophysiology of OCD, which complicates its therapy. Pharmacological augmentation with nutraceuticals like NAC, which antagonize the glutaminergic receptor may be efficacious.
Marble–burying behavior is a well–accepted pharmacological tool for evaluation of anti–compulsive activity in mice. Indeed, careful analysis of marble burying behavior has later led to the conclusion that it does not model anxiety, but may rather relate to compulsive behavior. Thus, mice did not avoid the marbles when given the opportunity to do so, suggesting that the marbles have no aversive or fear provoking properties. Repeated exposure to marbles did not lead to habituation to marble burying suggesting that this behavior is not related to novelty or fear. Marble burying is related to digging behavior and may in fact be more appropriately considered as an indicative measure of repetitive digging [17].
In our study CA significantly inhibited marble–burying behavior in mice, without any effect on motor activity. Moreover, total triterpenes of CA has already been reported to increase serotonin contents in rat brain [9]. In our study CA also inhibited marble burying behavior. The modification of serotonin neurotransmitter by CA may be responsible due to triterpenes like asiatic acid, madecassic acid and asiaticoside for the observed anti–compulsive effect. The presence of triterpenes in extract was also confirmed by HPLC analysis.
Moreover, N–acetylcysteine significantly inhibited marble– burying behavior in mice, without any effect on motor activity. The observed anti–compulsive effect of NAC is also well in accordance with earlier reports [11]. The anticompulsive effect of NAC may be due to attenuation of glutamatergic neurotransmission. NAC is converted to cysteine and up taken by glial cells results in stimulation of inhibitory metabotropic glutamate receptors on glutamatergic nerve terminals and thereby reduce the synaptic release of glutamate [11]. Thus NAC may be useful in treating psychiatric disorders involving glutamatergic dysfunction such as OCD. NAC also may have an advantage over other pharmacological agents due to lack of significant side effects.
Moreover, co–administration of Centella asiatica and N–acetyl cysteine, significantly reduced marble–burying at doses, on which either of these alone could not protect the behavior. Taken together with these findings, the present results suggest that the glutamatergic system and serotonergic system play an important role in the marble– burying behavior.
In the treatment of CA, NAC and fluoxetine locomotor activity were not changed significantly (P>0.05) and hence the involvement of the locomotor component maybe ruled out. Therefore, these effects were not attributable to changes of motor function.
Conclusion
First time our finding suggests that CA shows remarkable anti– OCD potential and confirming the traditional claims. Synergistic effects of CA with NAC could also be used at lower doses in the treatment of OCD. Moreover, in future identification of major active components and precise anti–compulsive mechanisms need to be focused.
References
- 1. Rasmussen SA, Eisen JL. The epidemiology and clinical features of obsessive–compulsive disorder. Psychiatr Clin North Am. 1992; 15: 743–758.
- 2. Bobes J, González MP, Bascarán MT, Arango C, Sáiz PA, Bousoo M. Quality of life and disability in patients with obsessive–compulsive disorder. Eur Pschiatry. 2001; 16: 239–245.
- 3. Eaton WW, Martins SS, Nestadt G, Bienvenu OJ, Clarke D, Alexandre P. The burden of mental disorder. Epidemol Rev. 2008; 30: 1–14.
- 4. Reddy YC, Rao NP, Khanna S. An overview of Indian research in obsessive compulsive disorder. Indian J Psychiatry. 2010; 52: 200–209.
- 5. Ruscio AM, Stein DJ, Chiu WT, Kessler RC. The epidemiology of obsessive–compulsive disorder in the National Comorbidity Survey Replication Mol Pychiatry. 2010; 15: 53–63.
- 6. Graybiel AM, Rauch SL. Toward a neurobiology of obsessive–compulsive disorder. Neuron. 2000; 28: 343–347.
- 7. Pallanti S, Hollander E, Bienstock C, Koran L, Leckman J, Marazziti D, et al. Treatment non–response in OCD: methodological issues and operational definitions. Int J Neuropsychopharmacol. 2002; 5: 181–191.
- 8. Camfield DA, Sarris J, Berk M. Nutraceuticals in the treatment of obsessive compulsive disorder (OCD): a review of mechanistic and clinical evidence. Prog Neuropsychopharmacol Biol Psychiatry. 2011; 35: 887–895.
- 9. Chen Y, Han T, Rui Y, Yin M, Qin L, Zheng H. Effects of total triterpenes of Centella asiatica on the corticosterone levels in serum and contents of monoamine in depression rat brain. Zhong Yao Cai. 2005; 28: 492–496.
- 10. Hashim P, Sidek H, Helan MH, Sabery A, Palanisamy UD, Ilham M. Triterpene composition and bioactivities of Centella asiatica. Molecules. 2011; 16: 1310–1322.
- 11. Egashira N, Shirakawa A, Abe M, Niki T, Mishima K, Iwasaki K, et al. N–acetyl–L–cysteine inhibits marble–burying behavior in mice. J Pharmacol Sci. 2012; 119: 97–101.
- 12. Umathe SN, Vaghasiya JM, Jain NS, Dixit PV. Neurosteroids modulate compulsive and persistent behavior in rodents: implications for obsessive–compulsive disorder. Prog Neuropsychopharmacol Biol Psychiatry. 2009; 33: 1161–1166.
- 13. Greene-Schloesser DM, Van der Zee EA, Sheppard DK, Castillo MR, Gregg KA, Burrow T, et al. Predictive validity of a non–induced mouse model of compulsive–like behavior. Behav Brain Res. 2011; 221: 55–62.
- 14. Kaurav BP, Wanjari MM, Chandekar A, Chauhan NS, Upmanyu N. Influence of Withania somnifera on obsessive compulsive disorder in mice. Asian Pac J Trop Med. 2012; 5: 380–384.
- 15. Koran LM, Hanna GL, Hollander E, Nestadt G, Simpson HB, American Psychiatric Association. Practice guideline for the treatment of patients with obsessive–compulsive disorder. Am J Psychiatry. 2007; 164: 5–53.
- 16. Decloedt EH, Stein DJ. Current trends in drug treatment of obsessive–compulsive disorder. Neuropsychiatr. Dis Treat. 2010; 6: 233–242.
- 17. Thomas A, Burant A, Bui N, Graham D, Yuva-Paylor LA, Paylor R. Marble burying reflects a repetitive and perseverative behavior more than novelty–induced anxiety. Psychopharmacology (Berl). 2009; 204: 361–373.