1Department of Anesthesia Critical Care and Pain Management, Harvard Medical School, USA
2Department of Anesthesia Critical Care and Pain Management, Beth Israel Deaconess Medical Center, USA
3Department of Surgery, Beth Israel Deaconess Medical Center, USA
*Corresponding author: McGinty-Froncek C, Department of Anesthesia, Critical Care and Pain Management, Harvard Medical School, Beth Israel Deaconess Medical Center, Boston, MA, USA
Received: September 30, 2014; Accepted: October 28, 2014; Published: October 28, 2014
Citation: McGinty-Froncek C, Geerling J, Hess P and Mallur PS. The Successful Management of Severe Subglottic Stenosis Under Jet Ventilation. Austin J Surg. 2014;1(8): 1036. ISSN: 2381-9030.
A previously healthy thirty year-old woman presented with respiratory distress secondary to severe subglottic stenosis at twenty-seven weeks gestation. We report her presentation and diagnosis. Next, we describe a means of intra-operative management with balloon dilation and lasering using a total intravenous anesthetic and two methods of sub-glottic jet ventilation. Critical for the success of this operation and all shared airway cases is thoughtful pre-operative planning and intra-operative communication between the surgical and anesthesia teams. Lastly, we address barotraumas, one of the complications of using jet ventilation and recommend considering chest ultrasounds for diagnosis of pneumothorax.
Keywords: Subglottic stenosis; Jet ventilation; Thoracic ultrasound; Pneumothorax
PFTs: Pulmonary Function Tests; FEV1: Forced Expiratory Volume in one second; FEV1/FVC: Forced Expiratory Volume in one second divided by Forced Vital Capacity; ENT: Ear Nose and Throat Surgeon; CO2: Carbon Dioxide; TIVA: Total Intravenous Anesthesia; ANSI: American National Standards Institute
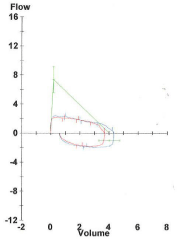
The patient, a 30 year old woman, presented at twenty seven weeks gestation with progressive shortness of breath and stridor. Until recently she was healthy and lived a very active lifestyle. She first noticed noisy breathing with exercise two years prior for which she saw a pulmonologist. At that time, her pulmonary function tests were normal. She was presumed to have asthma and prescribed inhalers. Her only prior surgical history was a reportedly uneventful shoulder surgery as a teenager for which she was incubated with an endotracheal tube. Her symptoms never limited her functional capacity until pregnancy. However, through the first two trimesters her symptoms worsened exponentially and it became evident that this was not the normal physiologic changes of pregnancy. New PFTs obtained by the pulmonologist showed a moderately reduced FEV1 and reduced FEV1/FVC ratio. The flow volume loop suggested a fixed intrathoracic obstruction with flattening of both inspiratory and expiratory loops (Figure 1). She was referred to an otolaryngologist for further evaluation.
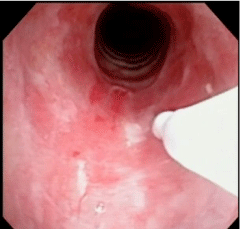
Based on the new pulmonary functions tests and a nasal endoscopy the ENT diagnosed her with laryngospasm, recommended voice rest and then referred her to a speech pathologist. Over a few weeks her condition rapidly deteriorated leaving her unable to speak in full sentences or ambulate more than a few feet. She sought a second opinion with an ENT whose repeat nasal endoscopy revealed significant subglottic stenosis (Figure 2). By his exam, airway diameter was estimated to be four to six millimeters with circumferential scar tissue just below the vocal cords. Despite the absolute preference to avoid radiation to the developing fetus, it was felt a cat scan was necessary in order to fully characterize the lesion to devise a plan for surgical correction. The imaging confirmed a fixed circumferential focal narrowing just below the vocal cords. The trachea was widely patent distal to the stenosis.
The patient was assessed by our thoracic surgeon, interventional pulmonologist, and otolaryngologist who all concurred that the lesion was most amenable to direct laryngoscopy, balloon dilation, and CO2 lasering by our ENT that specializes in laryngology. In the pre-operative holding area, she received pre-medication with midazolam, glycopyrolate, and dexamethasone. In the operating room after standard American Society of Anesthesiology monitors were placed the patient was thoroughly pre-oxygenated reaching an end tidal oxygen level of greater than 90% prior to induction of general anesthesia using fentanyl, propofol, and succinylcholine. The plan to use jet ventilation necessitated a Total Intravenous Anesthetic (TIVA) for maintenance anesthesia. The TIVA consisted of propofol and remifentanyl infusions, as well as intermitted rocuronium boluses.
After induction, the surgeon attempted to place number four universal modular glottiscope, however due to an anterior larynx, there was difficulty obtaining exposure. Next a Dedo laryngoscope was attempted without success. Finally, visualization was obtained using an Ossof laser laryngoscope. During this period the patient was apneic and the saturation remained in the upper nineties. The subglottis was approximately ninety percent stenosed with a three-millimeter aperture. Due to the severity of the stenosis, it was determined that the Mons- Hunsaker tube could not be passed. Instead, silastic tubing attached to the venturi adaptor on the laryngoscope for subglottic jet was used. First, the Acutronic Monsoon automatic high frequency jet ventilator was connected however the high-pressure alarm triggered and we quickly switched to the Manujet jet ventilator for low frequency jet ventilation. Chest expansion was noted with minimal chest recoil. In hopes of preventing barotraumas brief periods of jet ventilation was alternated with periods in which the patient was apneic and the catheter removed from the stenosis to give room air excursion. Next the stenosis was balloon dilated under intermittent apnea up to twelve millimeters in order to facilitate placement of the Hunsaker tube for subglottic jet ventilation.
Visualization and palpation revealed there was predominantly firm scar, though some soft granulation tissue on the posterior aspect. Under ANSI guidelines, the CO2 laser was used with a depth of one and power of eight to make radial incisions. In order to avoid an airway fire the laser was used under intermittent apnea and the area distal to the stenosis was protected with a moist pledget. A steroid injection was done at the site of the stenosis prior to the final balloon dilation to eighteen millimeters. After this, there was markedly improved airway diameter at the subglottic level.
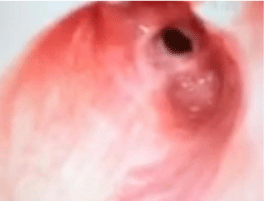
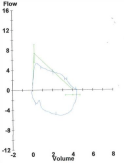
A bronchoscopy and bronchoalveolar lavage was performed to clear the trachea of secretions and blood. The patient was taken out of suspension and a laryngeal mask airway was placed to ventilate the patient during emergence from anesthesia. Prior to emergence a chest ultrasound exam was done bilaterally to asses for pneumothorax. She was extubated, taken to the post-operative care unit, and later transferred to the floor. On post-operative day one her breathing was dramatically improved and she was ready for discharge home. The flow volume loop from the PFTs done on post-op day six show a dramatic improvement (Figure 3). The patient’s nasal endoscopy done eighteen days post-operatively showed a widely patent trachea with some erythema at the site of prior stenosis (Figure 4). She continued to do well throughout her pregnancy and went on to deliver twelve weeks post-op. Five months post-op the laryngologist performed an in office bronchoscopy under local anesthesia. He noted a scar band along the medial aspect of the right main stem only minimally projecting into the lumen. She was referred to our interventional pulmonologist who performed a bronchoscopy under general anesthesia confirming the webbing of the right main stem bronchus, as well as minimal webbing in at the right upper lobe take off. Balloon dilation of the right main stem bronchus was performed with immediate improvement. At the time of the bronchscopy the gastroenterologists performed an endoscopy and Bravo pH study to determine if gastroesophagel reflux was potentially the cause of her subglottic stenosis. The results indicated a normal amount of acid reflux in the distal esophagus. Additionally, laboratory test for lupus and Wegener’s granulomatosis were negative. The only identified risk factor for subglottic stenosis was her prior intubation although thought to be unlikely given the brevity of the operation. The subglottic stenosis was labeled as idiopathic. She will continue to have follow-up visits with the laryngologist until one year post-op and thereafter return only if symptoms of dyspnea or stridor reoccur.
Subglottic stenosis, especially a case as severe as our patient’s requires careful planning and closes communication between the surgical and anesthesia teams. Pre-operative planning included consultations with an otolaryngologist, thoracic surgeon, and interventional pulmonologists to determine the best surgical approach to management. Just as there is not one surgeon for the job there is also not just one tool or one method of jet ventilation to use. A of the key elements to the success of our management was establishing primary, secondary, and tertiary plans. All tools were readily available in the operating room and tested before induction to ensure proper functioning. As you see with our case several laryngoscopes had to be tried before finding the right fit. Additionally our first choice of subglottic jet ventilation via the Hunsaker tube was deemed impossible once the airway was visualized. We were able to avoid an emergency tracheostomy or worse a failed airway by having a secondary method of jet ventilation immediately available.
As in our case, jet ventilation has been successfully used in the management of subglottic stenosis. However, it may not be appropriate for all patients with subglottic stenosis. The ease of performing the tracheostomy expeditiously, as well as the patient’s athleticism allowing her to tolerate periods of intermittent apnea were deciding factors in proceeding with general anesthesia instead of performing an awake tracheostomy. If one were managing a patient with challenging anatomy for a tracheostomy or a patient with a limited pulmonary reserve then an awake tracheostomy under local anesthesia should potentially be considered first line management.
There are three main categories of jet ventilation based on the catheter location; supraglottic jet ventilation, subglottic jet ventilation, and percutaneous-transtracheal jet ventilation. Our initial plan was to perform sub-glottic jet ventilation via a Hunsaker-Mon tube. This catheter measures 4.3mm in external diameter, has a central lumen for gas delivery and a side port for pressure monitoring, as well as end-tidal CO2 measurements [1]. Unfortunately, the external diameter was too large to fit through the narrow aperture of our patient’s stenotic airway. Therefore we used silastic tubing connected to the venturi attachment on the laryngoscope to perform subglottic jet ventilation. Alternatively, for subglottic jet ventilation one can use an Acutronic Laser Jet 40 double lumen catheter [1]. On the other hand, supraglottic jet ventilation can be employed by directly attaching the jet tubing to the suspension laryngoscope. This leaves a tubeless surgical field free from visual obstruction. The disadvantages of supraglottic jet is that it is more prone to developing increases in airway pressure, there is greater vocal cord movement, and it does not allow for pressure monitoring or measurement of end tidal CO2 [2].
That using jet ventilation during subglottic stenosis cases should consider incorporating thoracic ultrasound into their practice in order to rule out pneumothorax given the risk of barotraumas. The sensitivity of ultrasound is higher than standard upright anteroposterior chest radiography for the diagnosis of a pneumothorax [3]. Although CT remains the gold standard for diagnosis of pneumothorax it is less unappealing for several reasons. A CT scan would require patient transport out to the operating room, a time delay before diagnosis is established and particularly important in this case it would expose our patient, as well as her developing fetus to additional radiation. Although a CT was not done post-operatively to rule out pneumothorax, one was deemed necessary as part of the pre-operative evaluation of the patient’s stenosis. The imaging confirmed the stenosis was limited to the subglottic area, therefore amenable to balloon dilation and lasering. Alternatively, had the CT shown stenosis extending into the distal trachea she may have required an alternative surgical approach such as a tracheal resection. The CT scan clarified both the type of surgeon to manage the patient and the surgical approach.
Pre-operative pulmonary function test, Flow Volume Loop indicating a fix intrathoracic obstruction.
Pre-Operative nasal endoscopic view of the patient’s subglottic stenosis.
Post-operative day 6 Pulmonary Function Tests, Flow Volume Loop.
Eighteen days post-operative nasal endoscopic image of the patient’s trachea.