
Case Report
Austin J Surg. 2015; 2(6): 1074.
Air Embolism Due to Intrapulmonary Shunt Responsible of Multiorgan Ischemia in a HIV/HCV Co-Infected Liver Transplant Recipient
Cherchi V¹*, Baccarani U¹, Adani GL¹, Calandra S¹, Lorenzin D¹, Righi E², Bassetti M² and Risaliti A¹
¹Department of Medical and Biological Science, University of Medicine and Surgery of Udine, Italy
²Infectious Diseases Division, Santa Maria Misericordia Hospital, Italy
*Corresponding author: Cherchi V, Department of Medical and Biological Science, University of Medicine and Surgery of Udine, P.Le Santa Maria della Misericordia, 33100, Udine, Italy
Received: September 10, 2015; Accepted: October 16, 2015; Published: October 28, 2015
Abstract
Hepatopulmonary Syndrome (HPS) is characterized by an intrapulmonary vascular dilatation that occurs as a consequence of chronic liver disease in the absence of intrinsic cardiopulmonary disease. This produce arterial hypoxemia and the pulmonary symptoms of HPS are the result of oxygenation defects. We present a case of liver transplantation in patient affected by end-stage liver disease due to HCV co-infection, became hemiplegic on the left side on post operation day IV during the position of a central venous catheter in the right jugular vein. We investigate the presence of an Intrapulmonary Shunt (IPS) as a possible cause of peripheral air embolism according to several reports in the literature showing that IPS is a potential facilitator of Cerebrovascular Accident (CVA) or TIA.
We might conclude suggesting that the diagnosis of HPS should be considered and a contrast-enhanced echocardiography with bubble study should be performed in every patient during the pre-liver transplant workup especially in case of advanced end-stage liver disease (Child-Pugh C or elevated MELD score), when the prevalence of IPS is more frequent, moderate arterial hypoxemia also in the absence of significant dyspnea and evidence of severe left atrial dilatation.
Introduction
Hepatopulmonary Syndrome (HPS) is characterized by arterial hypoxemia due to intrapulmonary vascular dilatation that occurs as a consequence of chronic liver disease in the absence of intrinsic cardiopulmonary disease. HPS almost always occurs in association with advanced liver disease and it has been reported to be more frequently encountered in patients with Child-Pugh class C and high MELD values, achieving a prevalence of 10%-24% among patients with end-stage liver disease [1]. The prevalence of HPS among patients with HIV infection is unknown. The pulmonary symptoms of HPS are the result of oxygenation defects that occur as a result of acquired dilatations of the pulmonary blood vessels. HPS affects primarily the peripheral (precapillary and capillary) branches of the pulmonary vascular tree at the lung bases [2]. At these sites, the capillary diameter normally measures anywhere between 8 and 15 mm [3]. As a result of HPS, however, the capillary vessels may become dilated to 500 mm in diameter [4]. These pulmonary vascular dilatations cause apparent right-to-left intrapulmonary shunting (although there is no true anatomic shunt). Because the blood vessels are widely dilated, there is differential oxygenation of erythrocytes traversing the diameter of the abnormally dilated blood vessel. Erythrocytes flowing close to the vessel walls become appropriately oxygenated by the adjoining alveoli, whereas erythrocytes flowing midstream are not (i.e., there is a diffusion-perfusion defect). Midstream erythrocytes therefore fail to oxygenate, resulting in various degrees of hypoxemia. The diagnosis of HPS should be considered for patients with chronic liver disease who present with hypoxemia (especially if severe [5]) that is unexplained despite a thorough diagnostic work-up that essentially excludes intrinsic cardiac or pulmonary disease. Confirmation of the diagnosis of HPS rests on the findings of contrast-enhanced echocardiography, or bubble study [6]. Ten cubic centimeters of agitated saline are injected peripherally. Microscopic air bubbles (60– 90 mm in diameter) injected in the right atrium normally become lodged in the pulmonary capillaries (which are 8–15 mm in diameter) and are reabsorbed. The presence of microscopic air bubbles in the left heart chambers 3 or 4 beats after its appearance in the right atrium demonstrates the presence of the intrapulmonary vascular dilatations found in HPS. If bubbles are found in the left heart chambers, the differential diagnosis includes the possibility of an intracardiac shunt, such as a patent foramen ovale or atrial septal defect; however, with an intracardiac shunt, the microscopic air bubbles appear in the left heart chambers immediately after their appearance in the right heart chambers, with no delay. The presence of intrapulmonary shunts due to HPS has been reported as a potential facilitator of Cerebrovascular Accident (CVA) or TIA; intrapulmonary shunt occurred more frequently in the stroke group and was an independent predictor of CVA and/or TIA, also intrapulmonary shunt was an independent multivariate predictor of CVA or TIA in patients with crypto genetic CVA or TIA [7].
Case Description
We describe herein a case of a 43 y/o HIV positive Caucasian male affected by hemophilia and end-stage liver disease due to HCV co-infection. The patient was referred to our Center for liver transplantation with Child-Pugh class C and a MELD value of 30. Preoperative pulmonary work-up revealed a PaO2 of 76.2 mmHg in room air without significant dyspnea. The patients had also refractory as cites necessitating repeated large volume paracenthesis. A preoperative echocardiography was negative for intracardiac shunts, but revealed a severe left atrial dilatation, the pulmonary artery systolic pressure was normal (31 mmHg); the bubble study was not performed due to the absence of significant hypoxemia at arterial blood gas test in room air. Urgency liver transplantation (status 2A) was performed with a 70 y/o heart beating ABO compatible deceased female donor. The surgical procedure was uneventful and the liver recovered normal functions despite for progressive cholestasis that was attributed to early HCV recurrence. On post-transplant day VI the patient underwent emergency surgery for acute peritonitis due to complete ischemic necrosis and perforation of the sigmoid colon; a Hartman procedure was carried out successfully. Preoperative abdominal CT scan and intraoperative findings did not reveal any signs of thrombosis of the superior or inferior mesenteric vessels. On post transplant day IV during the position of a central venous catheter in the right jugular vein the patient became hemiplegic on the left side and a CT scan of the head revealed an ischemic lesion located on the right side at the front-parietal level, no sign of intracranial hemorrhage or thrombosis of the large cervical and intracranial vessels were found at CT scan; a carotid echo-Doppler was negative. Despite a negative pre-transplant echocardiography we decide to investigate the presence of an Intrapulmonary Shunt (IPS) as a possible cause of peripheral air embolism performing a contrast-enhanced echocardiography with bubble study that showed the presence of microscopic air bubbles in the left heart chambers 3 or 4 beats after its appearance in the right atrium suggestive of an IPS grade IV according to Vedrinne (Figure 1) [8]. According to several reports in the literature showing that IPS has been reported as a potential facilitator of Cerebrovascular Accident (CVA) or TIA we hypothesized that both ischemic insults, at least the cerebral one, occurred after transplantation in the absence of any evidence of vascular thrombosis, might be explained by peripheral air embolism due to intrapulmonary right to left shunt. In our case the presence of IPS was not investigated pre-transplant due to the absence of significant hypoxemia in room air and dyspnea, however the presence of a left atrial dilatation might have suggested us an increased risk of IPS as reported by Zamarian et al. [9] (Figure 2). Finally we might conclude suggesting that a contrast-enhanced echocardiography with bubble study should be performed in every patient during the pre-liver transplant workup especially in case of advanced end-stage liver disease (Child-Pugh C or elevated MELD score), when the prevalence of IPS is more frequent [10], moderate arterial hypoxemia also in the absence of significant dyspnea and evidence of severe left atrial dilatation.
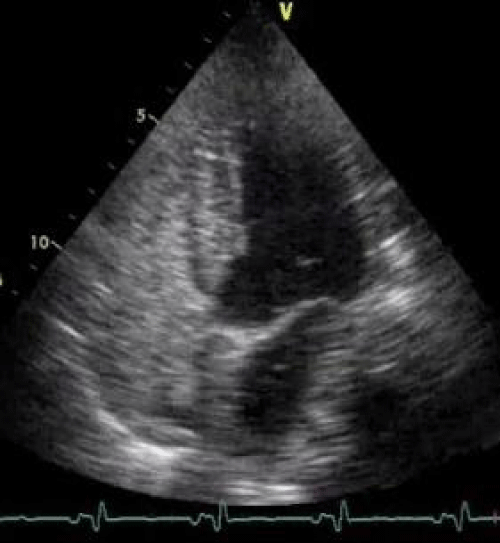
Figure 1: Bubble study: Pre contrast-injection echocardiograph image.
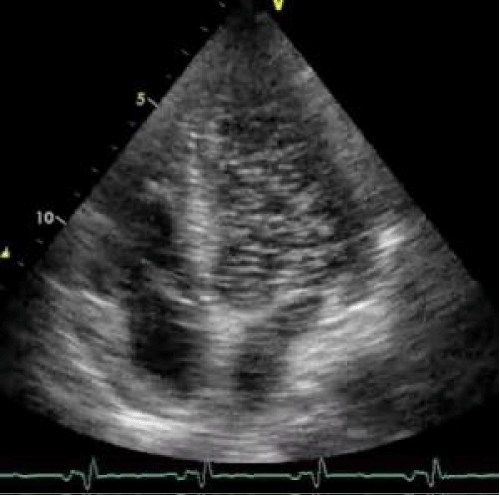
Figure 2: Bubble study: Findings of contrast-enhanced echocardiography.
References
- Torno MS, Witt MD, Sue DY. Hepatopulmonary syndrome in HIV-hepatitis C virus coinfection: a case report and review of the literature. Clin Infect Dis. 2004; 39: e25-29.
- Añel RM, Sheagren JN. Novel presentation and approach to management of hepatopulmonary syndrome with use of antimicrobial agents. Clin Infect Dis. 2001; 32: E131-136.
- Schraufnagel DE, Kay JM. Structural and pathologic changes in the lung vasculature in chronic liver disease. Clin Chest Med. 1996; 17: 1-15.
- Chiesa A, Ciappi G, Balbi L, Chiandussi L. Role of various causes of arterial desaturation in liver cirrhosis. Clin Sci. 1969; 37: 803-814.
- Krowka MJ. Clinical management of hepatopulmonary syndrome. Semin Liver Dis. 1993; 13: 414-422.
- Scott VL, Dodson SF, Kang Y. The hepatopulmonary syndrome. Surg Clin North Am. 1999; 79: 23-41.
- Abushora MY, Bhatia N, Alnabki Z, Shenoy M, Alshaher M, Stoddard MF. Intrapulmonary shunt is a potentially unrecognized cause of ischemic stroke and transient ischemic attack. J Am Soc Echocardiogr. 2013; 26: 683-690.
- Vedrinne JM, Duperret S, Bizollon T, Magnin C, Motin J, Trepo C, et al. Comparison of transesophageal and transthoracic contrast echocardiography for detection of an intrapulmonary shunt in liver disease. Chest. 1997; 111: 1236-1240.
- Kim BJ, Lee SC, Park SW, Choi MS, Koh KC, Paik SW, et al. Characteristics and prevalence of intrapulmonary shunt detected by contrast echocardiography with harmonic imaging in liver transplant candidates. Am J Cardiol. 2004; 94: 525-528.
- Zamirian M, Aslani A, Sharifkazemi MB. Prediction of intrapulmonary right to left shunt with left atrial size in patients with liver cirrhosis. Eur J Echocardiogr. 2008; 9: 1-4.