
Rapid Communication
Austin J Urol. 2021; 7(1): 1066.
Augmentation Cystoplasty in Children: Institute of Kidney Diseases Peshawar Experience
Muhammad Naeem, Muhammad Kamran Khan*, Arshad, Majid Khan, Mehboob ul Wahab, Ihsanullah
Department of Urology, Institute of Kidney, Peshawar, Pakistan
*Corresponding author: Muhammad Kamran Khan, Assistant Professor of Pediatric Urology, Institute of Kidney Diseases, Peshawar, Pakistan
Received: May 11, 2021; Accepted: June 07, 2021; Published: June 14, 2021
Abstract
Objective: The aim of this study is to share our single-center experience of Augmentation Cystoplasty (AC) in children regarding indications, bowel segment used, associated procedures, and its complications.
Materials and Methods: We analyzed data of all pediatric patients who underwent AC at Institute of Kidney Diseases (IKD), Peshawar between July 2017 and March 2020.
Results: A total of 18 pediatric patients are included in the study who underwent Bladder augmentation (BA) either isolated in 1 (5.5%), or along with other associated procedures like Mitrofanoff 17 (94.4%), Antegrade Continent Enema (ACE) 6 (33.3%), Bladder Neck Reconstruction (BNR) 5 (27.7%) and Bilateral ureteric Re-implantation (B/L UR) in 3 (16.6%). Indication for Bladder augmentation were Neurogenic bladder (NGB) 13 (72.2%), Exstrophy Epispadias Complex (EEC) 3 (16.6%) and Posterior urethral value with small functional capacity bladder 2 (11.1%). Small gut used in 15 (83.3%) and large gut in 3 (16.6%) for BA. For Mitrofanff associated with bladder augmentation, Appendix was used in 14 (82.35%) and small gut (Monti) in 3 (17.6%) cases. For ACE associated with BA, in 4 (66.6%) Appendix used as right-sided ACE and in 2 (33.3%) small gut (Monti) as left-sided ACE. No intraoperative complications noted, while early post-op complications, 2 wound infection and 1 urinary leakage from wound, were noted in 3(16.6%) cases and late post-op complications (stomal stenosis) in 4(22.2%) patients.
Keywords: Augmentation cystoplasty; Mitrofanoff; Antegrade continent enema; Neurogenic bladder; Exstrophy epispadias complex; Mitrofanoff stenosis
Abbreviations and Acronyms
AC: Augmentation Cystoplastyl; Mit: Mitrofanoff; ACE: Antegrade Continent Enema; BNR: Bladder Neck Reconstruction; B/L UR: Bilateral Ureteric re-implantation; IKD: Institute of Kidney Disease; HMC: Hayatabad Medical Complex; NGB: Neurogenic Bladder; NGBB: Neurogenic Bladder and Bowel; EEC: Exstrophy Epispadias Complex.
Introduction
Fecal and urinary incontinence in spina bifida patient is one of the most devastating condition of the children. It has both social and psychological implications and decreased quality of life along with other comorbidities. Most of these patients are treated with various types of enemas to clean out the colon for bowel management and clean intermittent catheterization with or without anticholinergic medications for bladder management. When conservative measures of medication and clean intermittent catheterization fails in the management of Neurogenic bowel and bladder then surgical treatment is opted which includes botulinum toxin to the detrusor, continent catheterizable conduit with or without bladder augmentation [1-3].
AC is considered in the pediatric population for lower urinary tract reconstruction in cases of congenital urological anomalies like Neurogenic Bladder (NGB) associated with spina bifida as last resort when conservative measures and other minimally invasive measures like intradetrussor botulinum toxin have failed . In such cases, the bladder is augmented with viscoelastic tissue, commonly autogenous gastrointestinal segment. This increases bladder capacity and decreases the bladder pressures, thereby protecting the upper tract and also helps in improving the continence and symptoms [4]. AC is a good option with a reasonable satisfaction rate and good long term outcomes including improvement in symptoms and continence in both neurogenic and non-neurogenic bladder dysfunction [5-7]. It also improves urodynamic parameters and associated with variable rate of clean intermittent catheterizations [8].
International Consultation on Incontinence also recommend BA for reduced bladder capacity and compliance and detrusor over activity when other treatment options like medical treatment, Botulinum toxin injection, and/or neuromodulations have failed [7]. According to European Association of Urology guidelines, BA is recommended to decrease detrusor pressure and increase bladder capacity, in case of failure of more conservative options [9]. If patient are not able to perform the transurethral intermittent catheterization, Mitrofannof either using Appendix or small gut (Monti) channel is created to drain the bladder [10,11].
The aim of this study was to share our experience of AC for bladder dysfunction associated with neurogenic and non neurogenic bladders. We wanted to see the safety and efficacy of the technique, indications for the AC, bowel segment used and the complications associated with the procedure.
Materials and Methods
Between July 2017 and March 2020, after the institute ethical committee approval, all pediatric patients record who underwent BA were retrospectively identified. Total of 18 patients who underwent BA either alone or associated with other procedures including Mitrofanoff, Antegrade Continent Enema (ACE), Bladder Neck Reconstruction (BNR) at our institution were included in the study. Patient records were accessed through an institutional medical record system. Statistical analyses was performed with IBM® SPSS®, version 20.0.
Results
A total of 18 patients with mean age of 9.5 years identified, which included 10 (55.6%) female and 8 (44.4%) male patients. They underwent AC (Immediate Post Op Picture, Figure 1) either isolated or along with other procedures like Mitrofanoff, Antegrade continent enema, Bladder neck reconstruction, between July 2017 and March 2020. The details of the procedures performed in these patients are shown in Table 1. The indications of AC in these patients are given in Table 2.
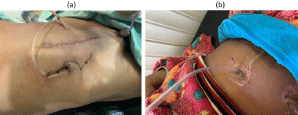
Figure 1: (a) Immediate Post Op Bladder Augmentation with VQZ Mitrofanoff.
(b) S/P Bladder Augmentation with Mitrofanoff and ACE (catheter can be
seen in both stomas).
Operation
Frequency
Percent
AC
AC + Mit
AC + Mit + ACE
AC + Mit + ACE + BNR
AC + Mit + BNR
AC+ Mit + BNR + Bil UR Re-implantation1
7
5
1
1
35.6
38.9
27.8
5.6
5.6
16.7Total
18
100
Table 1: Procedures.
Conditions
Frequency
Percent
NGB
13
72.2
EEC
3
16.7
PUV
2
11.1
Total
18
100
Table 2: Indications for Augmentation cystoplasty.
Bowel segments for AC were either taken from the small gut or large gut, Table 3. Out of 6 ACE associated with bladder augmentation, in 4 (66.6%) Appendix was used as right-sided ACE (split appendix Malone and Mitrofanoff) Figure 2 and in 2 (33.3%) small gut (Monti tube) as left-sided ACE where Monti tube is implanted to the descending colon.

Figure 2: Split (divided) appendix with catheters in both parts for Mitrofanoff
and ACE procedures.
Augmentation Cystoplasty
Frequency
Percent
Small Gut
15
83.3
Large Gut
3
16.7
Total
18
100
Table 3: Intestine Part Used for.
The mean hospital stay was 8 days and mean operative time for AC was 237+-11 Minutes. No intra-operative complications were found. Early postop complications were noted in 3 (16.6%) cases, which included 2 cases of wound infection and one case urinary leakage from wound and all these cases were managed conservatively with wound dressing and catheterization respectively and late post-op complications of stomal stenosis (one ACE stoma and 3 Mitrofanoff stoma) in 4 (22.2%) patients which required stomal revision.
Discussion
Bladder augmentation is already being practiced for other indications like interstitial cystitis and Tuberculous bladder [12] but later in the late seventies it became an important treatment option for bladder dysfunction. The most common indication for AC in our study was NGB (72.2 %). In the study by flood HD et al, they performed AC for NGB in 29.1%, interstitial cystitis in 29.1%, and post-inflammatory bladder retraction in 33.3% [13], while in the study by Mast P et al 89% of patient had AC for NGB bladder [14]. Fatih et al. noted 54.4% cases operated for AC were NGB [15]. This shows that NGB is one of the major indication which needs augmentation of the bladder at some stage of its management when the conservative management like clean intermittent catheterization with or without anticholinergics and minimally invasive procedures like intradetrussor botulinum toxin have failed.
The bladder can be augmented with different parts of the bowel like a small gut, large gut or stomach as well as with Dilated ureter of a non functioning kidney. We used either small gut (83.3%) or large gut (16.7%) in all of our cases. Gearhart et al used small gut in 60.86% and large gut in 23.7% [16] Fatih e al. found 44.82% cases of the small gut, 3.4% gastric, and 5.51 % ureteric use in AC [15]. This infers the wide use of small and large gut as the main sources of tissue to augment the bladder with good results. We did not use stomach or ureters for BA in any of cases.
Only one case was found to have isolated AC that was a 6 years old girl and parents opted for clean intermittent catheterization through urethra , remaining all included other procedures like Mitrofannof (94.4%), ACE (33.3%) for bowel management of neurogenic bowl, bladder neck reconstruction (27.7%), and Ureteric reimplantation (16.6%). In experience sharing by Fatih et al. they did Mitrofannof in (39.31%), BNR in (15.86%) [15]. In cases of NGB usually multiple associated procedures are required with AC either for bladder management only like Mitrofanoff or other problems like ACE for the bowel management etc as these patents have multiple co morbidities and this makes management of these patients very challenging both on part of surgeon and family. Therefore the preoperative counselling regarding lifelong follow up is necessary, as this require well motivated patients and family support.
For bowel dysfunction management in addition to AC, the associated procedure of ACE was performed on the right side using proximal part of appendix (split appendix Malone and Mitrofanoff) in 66% and on left-sided using Monti tube implanted in the descending colon in 33.3% of patients. Chang HK et al performed right-sided ACE in (50.98%) and left sided in (49.01%) [17]. Furtado Meyer et al, used 70% right and 30% left-sided antegrade continent enema [18].
Most of the long term complications associated with BA are those related to catheterizable channels. The main complications include stomal prolapse, stomal incontinence, and difficulty catheterizing, typically due to channel stricture/stenosis and false passage. The incidence of these complications varies widely based on the series, the type of channel used, and the length of follow-up. In general, the incidence of stomal prolapse ranges from 2–5 %, stomal incontinence 1–47 %, difficulty catheterizing 5–32 % and overall rates of surgical revision range from 18–58 % [19-22]. We did not notice any mucosal proplase and stomal incontinence in our series but three patients had mitrofanoff stomal stenosis and one patient has Malone stenosis (22%) at one year follow up. The patient with Malone stoma stenosis did not use the stoma after the nelaton tube removal for enema administration. we keep the Malone stomal catheter for a month initially and then administer enema through intermittent catheterization. One patient has difficulty in catheterizing Malone stoma, who required dilatation of stoma over guide wire in the clinic and since then patient is doing well and did not require any further procedures. Though in A.J.reseng series, 49% of the patients had difficulty in catheterizing the Malone stoma but had a long follow up of 4.7 years compared to the one year in our series. Obesity was seen as the only significant risk for stomal stenosis in there group of patients [23]. In our series, we did not notice any per urethral urinary leakage. The reason of 100 percent continence in these neurogenic patients might be the usage of fascial bladder neck slings in addition to bladder neck reconstruction to enhance chances of urinary continence.
No long term complications of BA like bladder stones, metabolic abnormalities, malignancies etc were noticed which is probably due to short follow up of these patients.
The limitations of this study are its retrospective nature, short follow up and small study sample. A larger cohort with long term follow up is needed to better identify the long term complications of the AC.
Conclusion
AC is a commonly performed procedure for bladder dysfunction with NGB and non neurogenic bladder when conservative and minimally invasive procedures are exhausted or failed. AC is associated with significant improvement in symptoms and urodynamic parameters with acceptable complications rate.
References
- Clark T, Pope JC, Adams MC, Wells N, Brock JW 3rd. Factors that influence outcomes of the Mitrofanoff and Malone antegrade continence enema reconstructive procedures in children. J Urol. 2002; 168: 1537–1540.
- Thomas JC, Dietrich MS, Trusler L, DeMarco RT, Pope JC, Brock JW, et al. Continent catheterizable channels and the timing of their complications. J Urol. 2006; 176: 1816–1820.
- Farrugia MK, Malone PS. Educational article: The Mitrofanoff procedure. J Pediatr Urol. 2010; 6: 330–337.
- Biers SM, Venn SN, Greenwell TJ. The past, present and future of augmentation cystoplasty. BJU Int. 2012; 109: 1280–1293.
- Bramble FJ. The treatment of adult enuresis and urge incontinence by enterocystoplasty. Br J Urol. 1982; 54: 693–696.
- Mundy AR, Stephenson TP. “Clam” ileocystoplasty for the treatment of refractory urge incontinence. Br J Urol. 1985; 57: 641–646.
- Drake MJ, Apostolidis A, Emmanuel A, Gajewski J, Harrison SC, Heesakkers J, et al. Neurologic urinary and faecal incontinence. Editors. In: Abrams P, Cardozo L, Khoury S, Wein A, editors. Incontinence. 5th Edition. Paris: ICUDEAU. 2013: 827-954.
- Veeratterapillay R, Thorpe AC, Harding C. Augmentation cystoplasty: Contemporary indications, techniques and complications. Indian journal of urology: IJU: journal of the Urological Society of India. 2013; 29: 322–327.
- Blok B, Pannek J, Castro Diaz D, del Popolo G, Groen J, Gross T, et al. Guidelines on neuro-urology. Arnhem: European Association of Urology. 2015.
- Cetinel B, Demirkesen O, Onder AU, Yaycioglu O, Ismailoglu V, Solok V. Reconstructive surgery in voiding dysfunction: experience with 69 patients. Urology. 2000; 56: 962–966.
- Cetinel B. Reconstructive surgery in neuropathic bladder. Adv Exp Med Biol. 2003; 539: 509–533.
- Kockelbergh RV, et al. Clam enterocystoplasty in general urological practice. Br JUral. 1991; 68: 38-41.
- Flood HD, Malhotra SJ, O’Connell HE, Ritchey MJ, Bloom DA, et al. Long-term results and complications using augmentation cystoplasty in reconstructive urology. Neurourol Urodyn. 1995; 14: 297–309.
- Mast P, Hoebeke P, Wyndaele JJ, Oosterlinck W, Everaert K. Experience with augmentation cystoplasty. A review. Paraplegia. 1995; 33: 560-564.
- Çanakli F, Yüksel S, Dogan HS, Aki FT, Tekgül S. Augmentation Cystoplasty: Long-Term follow up Of 112 Casesjpurol. 2008.
- Gearhart JP, Albertsen PC, Marshall FF, Jeffs RD. Pediatric Applications of Augmentation Cystoplasty: The Johns Hopkins Experience. The Journal of Urology. 1986; 136: 430-432.
- Chang HK, Chang EY, Han SJ, Choi SH, Oh JT. Long-term outcome of leftvs right-sided antegrade continence enema. J Pediatr Surg. 2012; 47: 1880- 1885.
- Meyer F, Macedo M, Filho H, Pinto T, Galvao L, Meneses C. The Malone Antegrade Continence Enema (MACE). Principle In Children: Is It Important If the Conduit Is Implanted In the Left or the Right Colon?. International braz j urol. 2008; 34: 206-212.
- Süzer O, Vates TS, Freedman AL, et al. Results of the Mitrofanoff procedure in urinary tract reconstruction in children. Br J Urol. 1997; 79: 279-282.
- Leslie B, Lorenzo AJ, Moore K, et al. Long-term followup and time to event outcome analysis of continent catheterizable channels. J Urol. 2011; 185: 2298-2302.
- Welk BK, Afshar K, Rapoport D, et al. Complications of the catheterizable channel following continent urinary diversion: their nature and timing. J Urol. 2008; 180: 1856-1860.
- Polm PD, de Kort LMO, de Jong TPVM, et al. Techniques used to create continent catheterizable channels: a comparison of long-term results in children. Urology. 2017; 110: 192-195.
- Rensing AJ, Koenig JF, Austin PF. Preoperative risk factors for stomal stenosis with Malone antegrade continence enema procedures. J Purol. 2017; 13: 631.e1.