
Case Report
Austin J Dermatolog. 2016; 3(5): 1063.
Remission of Pityriasis Rubra Pilaris with IL-17A Antagonist Secukinumab
Mariasy, Campanelli and Sauder DN*
Yardley Dermatology Associates, USA
*Corresponding author: Sauder DN, Yardley Dermatology Associates, 903 Floral Vale Blvd, Yardley, PA, 19067, USA
Received: October 12, 2016; Accepted: November 23, 2016; Published: November 25, 2016
Abstract
Pityriasis Rubra Pilaris (PRP) is a rare inflammatory disease of unknown etiology associated with follicular hyperkeratosis, scaly erythematous plaques, palmoplantar keratoderma and generalized erythroderma [1]. Treatment of PRP is difficult as there is no standard protocol. Traditional treatments used have included oral retinoids, methotrexate, TNF-a inhibitors and corticosteroids. After failing the above therapies, we then attempted to control this particular case with the oral phosphodiesterase-4 inhibitor (PDE4) Otezla without success. We report of a case of remission of Pityriasis Rubra Pilaris with the administration of the newly marketed interleukin-17A antagonist Secukinumab (Cosentyx, Novartis). These findings suggest a possible correlation between interleukin- 17a and this particular papulosquamous eruption.
Keywords: Pityriasis rubra pilaris; Erythroderma; Cytokines
Abbreviations
PRP: Pityriasis Rubra Pilaris; IL-17A: Interleukin 17A; Nb-UVB: Narrowband Ultraviolet B; MTX: Methotrexate
Introduction
Pityriasis Rubra Pilaris (PRP) is a rare skin disorder which can affect both adults and children. While current research is suggesting a genetic component in familial PRP with the mutation of CARD14 [2], acute adult onset PRP in patients over the age of fifty is the more common presentation of the disease. Treatment for PRP can prove difficult, and common first line therapies include oral retinoids, methotrexate, TNF-a inhibitors and UVB narrowband therapy [3]. More recently, cases have been reported achieving remission with non-traditional therapeutic methods such as the Phosphodiesterase 4 (PDE4) Inhibitor, apremilast (Otezla; Celgene) [4], though it yielded no improvement for our patient. We report our findings of remission with secukinumab to potentially suggest a connection amongst IL- 17A and the pathophysiological mechanism of PRP.
Case Presentation
A 68-year-old white male presented in office complaining of an itchy rash that had been present for several weeks. Clinically, he presented with erythematous scaling with patches on the neck, forehead and torso. Our initial clinical differential diagnosis included pityriasis lichenoides chronica, pityriasis rosea and nummular eczema. The pathology report was non diagnostic but suggestive of spongiotic dermatitis. Several weeks later, the rash progressed to grouped follicular keratotic papules with extensive erythema and marked palmarplantar hyperkeratosis. Additional punch biopsies were performed. The pathology showed regular acanthosis and psoriasiform hyperplasia and orthokeratosis consistent with PRP.
The patient then progressed to clinically resemble an acute adult onset PRP with erythroderma, (Figure 1) palmar plantar keratoderma and nails with thick, distal splinter hemorrhages. We considered traditional first-line systemic treatments and begin the patient on 25mg of acitrentin. In an attempt to offer some relief for his generalized erythroderma, the use of topical corticosteroids and emollients were implemented. At the one-month follow-up, the patient’s condition had worsened to the point where he was claiming that “plates of skin” were sloughing off of his body. We added UVB narrowband treatment tri-weekly at Fitzpatrick Skin Type 1 dosing. After no improvement in two months, we added Methotrexate at 15mg for a period of two months. His disease then progressed to include severe palm and sole pain along with extensive palmar plantar hyperkeratosis, with no improvement from the Nb-UVB or MTX. No response was seen after an additional month on methotrexate, which we then opted to discontinue and an oral Phosphodiesterase 4 (PDE4) Inhibitor, apremilast (Otezla; Celgene), was started. The patient remained on apremilast for approximately two months with no marked improvement. Due to the immuno-driven nature of PRP, we considered biologic therapy for the next course of action. After noting the efficacy of secukinumabin psoriasis patients (79% of patients achieved a PASI 90 at week sixteen.) [5], we began the patient on the loading dose of secukinumab: two 150mg syringes injected into the lateral thigh. Four weeks later we were able observe less erythema. He remained on a maintenance dose of 150mg and after eight weeks of continued secukinumab therapy the patient was significantly improved, with only mild erythema but absent of scaling or exfoliation. At his six-month follow-up there was no evidence of disease and the patient had achieved a complete remission.
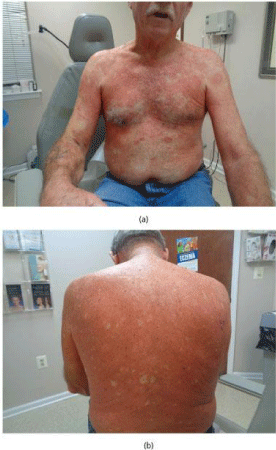
Figure 1: (a) Pityriasis rubra pilaris, generalized erythroderma, (b) PRP.
Discussion
The etiology of PRP remains uncertain and treatment can be challenging. Fundamentally, care for PRP patients must be tailored specifically to the patient’s needs, efficacy and comorbidities. Our patient saw no improvement with traditional first course therapeutic action of oral retinoids. UVB narrowband therapy failed to elicit a response, and at times even seemed to exacerbate symptoms. While there has been a recent study demonstrating remission of PRP with apremilast, it had no value for our patient [4]. Due to the aggressive nature of this particular case, biologic therapy was inevitable for the next course of action.
Secukinumab is the first human IL-17A antagonist first approved for the treatment of moderate to severe plaque psoriasis. Secukunimab is a human IgG1 monoclonal antibody that selectively binds to interleukin-17A cytokine: a naturally occurring cytokine that is involved in inflammatory and immune responses [6]. While TNF-a inhibition has been shown to be an effective therapy for refractory PRP, failures are well documented [7]. Therefore; we elected IL-17 inhibition for our first systemic biologic treatment for our patient (Figure 2).
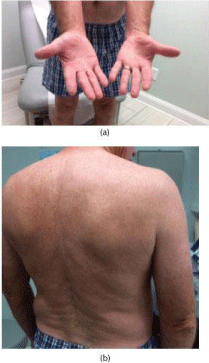
Figure 2: (a) PRP in remission, (b) Remission of PRP.
Our experience suggests that secukinumab may be an effective means to treat resistant PRP. Recently published data confirms our findings: that the IL-17A cytokine may be correlated with the mechanism of PRP [8]. Additional studies are imperative to establish any relationship between the etiology of PRP and interleukin 17.
References
- Albert MR, Mackool BT. Pityriasis rubra pilaris. Int J Dermatol. 1999; 38: 1-11.
- Fuchs-Telem D, Sarig O, van Steensel MA, Isakov O, Israeli S, Nousbeck J, et al. Familial Pityriasis Rubra Pilaris Is Caused by Mutations in CARD14. National Center for Biotechnology Information. U.S. National Library of Medicine. 2012; 91: 163-170.
- Kalner V, Mann DJ, Wasserman J, Petronic-Rosic V, Tsoukas MM. Pityriasis Rubra Pilaris Sensitive to Narrow Band-ultraviolet B Light Therapy. National Center for Biotechnology Information. U.S. National Library of Medicine. J Drugs Dermatol. 2009; 8: 270-273.
- Krase IZ, Cavanaugh K, Curiel-Lewandrowski C. Treatment of Refractory Pityriasis Rubra Pilaris with Novel Phosphodiesterase 4 (PDE4) Inhibitor Apremilast. JAMA Dermatology. 2016; 152: 348-350.
- Blauvelt A. Secukinumab vs. Ustekinumab. Dermatology Times. 2015.
- Novartis Pharmaceutical Corporation. Cosentyx [package insert]. Basel, Switzerland, 2016.
- Lu R, George SJ, Hsu S. Pityriasis Rubra Pilaris: Failure of Combination Treatment with Acitretin and Infliximab. Dermatology Online Journal. 2006; 12: 18.
- Haskin, Alessandra BA, Fisher A, Ginette A, Okoye MD. Successful Treatment of Refractory Pityriasis Rubra Pilaris with Secukinumab. JAMA Dermatology. 2016; 152: 1278-1280.