Editorial
Austin J Otolaryngol. 2014;1(1): 3.
Pleomorphic Adenoma of the Accessory Parotid Gland
Hiroyoshi Iguchi* and Hideo Yamane
Department of Otolaryngology and Head & Neck Surgery, Osaka City University Graduate School of Medicine, Japan
*Corresponding author: Hiroyoshi Iguchi, Department of Otolaryngology and Head & Neck Surgery, Osaka City University Graduate School of Medicine: 1-4-3, Asahi-machi, Abeno-ku, Osaka, 545-8585, Japan
Received: May 18, 2014; Accepted: May 20, 2014; Published: May 22, 2014
Keywords
Salivary gland; Accessory parotid gland; Pleomorphic adenoma; MRI; Surgery
Abbreviations
APG: Accessory Parotid Gland; PA: Pleomorphic Adenoma; CT: Computed Tomography; MRI: Magnetic Resonance Imaging; FNAC: Fine–Needle Aspiration Cytology
Editorial
The accessory parotid gland (APG) is commonly located anterior to the main parotid gland, between the zygomatic arch and the Stensen duct, and attached to the masseter muscle. The APG is connected to the Stensen duct by one or more tributary ducts [1]. The incidence of APGs is 21–61% [2]. However, APG tumors are rare with an incidence of only 1–7.7 % of all parotid gland tumors [1,3]. All tumor pathologies that occur in the main parotid gland may also occur in the APG. According to previous reports, the most common subtype of benign APG tumors is pleomorphic adenoma (PA) [4,5]. Newberry et al. reported that 58.6% of benign APG tumors were PA [4], and in our previous study on 65 APG tumors in Japan, PA accounted for 80.6% of benign APG tumors [6].
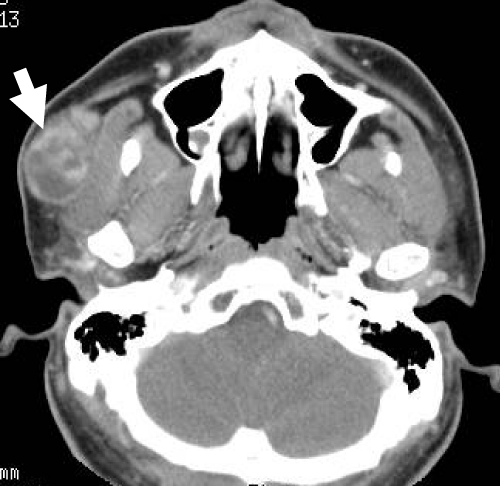
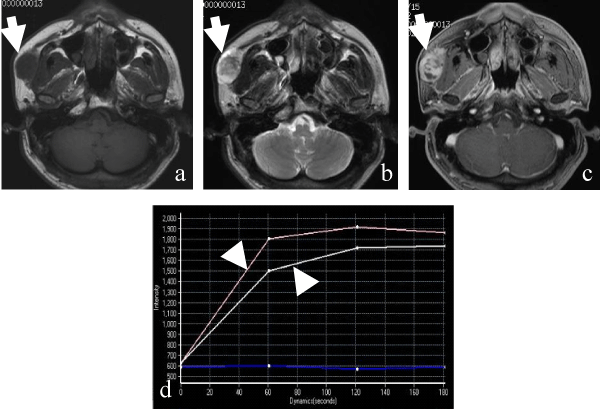
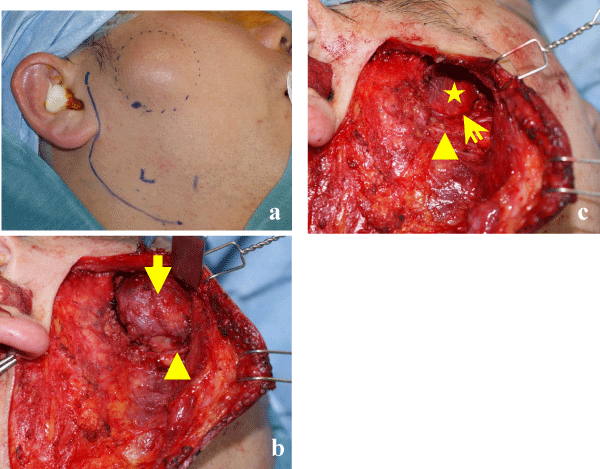
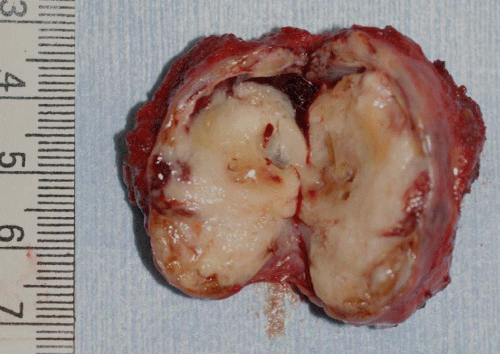
Here, we describe a typical case of a large PA of the APG treated in our hospital. A 35–year–old man presented with a 5–year history of a slow–growing, right mid–cheek mass in the pre–parotid region. The mass was mobile with a smooth surface. He denied tenderness on palpation and facial weakness. Computed tomography (CT) of the neck confirmed a well–circumscribed and heterogeneously enhanced, 36 × 23–mm–sized mass situated anterior to the right main parotid gland and on the surface of the masseter muscle (Figure 1).The ovoid mass demonstrated low and heterogeneously highsignal intensity on T1– and T2–weighted magnetic resonance imaging (MRI), respectively, and heterogeneous enhancement on contrast–enhanced T1–weighted MR images. A time–signal intensity curve on dynamic MRI revealed a gradual enhancement pattern (Figure 2). Ultrasonography–guided fine–needle aspiration cytology (FNAC) indicated PA. Standard parotidectomy incision with anterior extension was chosen for complete and safe exposure of the mass, facial nerve branches, and Stensen duct. The mass with the surrounding salivary tissue was completely separate from the main parotid gland and connected to the Stensen duct via a tributary duct.The mass was completely resected without damaging the tumor capsule, facial nerve branches, or Stensen duct (Figure 3). The intraoperative pathological diagnosis using a frozen specimen was PA. The tumor was well encapsulated and solid, and the cut surface was yellowish white (Figure 4). Postoperative complications such as facial weakness and salivary fistula were not observed.
Figure 1: An axial contrast-enhanced computed tomography image showing an ovoid lesion (arrow) with heterogeneous enhancement located on the outer layer of the masseter muscle.
Figure 2: Axial magnetic resonance images.a.T1-weighted image, b. T2- weighted image, c. contrast-enhanced T1-weighted image, d. time-signal intensity curve on dynamic magnetic resonance imaging (MRI). The wellcircumscribed tumor (arrows) is located anterior to the main parotid gland. The tumor demonstrates low and heterogeneously high signal intensity on T1-and T2-weighted images, respectively, and heterogeneous enhancement on contrast-enhanced T1-weighted images. The time-signal intensity curve on dynamic MRI indicates a gradual enhancement pattern (arrow heads).
Figure 3: Intraoperative findings. a. skin incision (standard parotidectomy incision),b. exposure of the tumor and buccal branch of the facial nerve, c. after tumor resection. Dotted circle indicates the location of the tumor. Arrow: tumor, arrow head: buccal branch of the facial nerve, double arrow: Stensen duct, asterisk: masseter muscle.
Figure 4: Cut surface of the resected tumor. The tumor is completely covered with a fibrous capsule.
PA of the APG usually presents as an asymptomatic and painless mid–cheek mass growing slowly over years [5]. Malignant transformation should be suspected if the tumor presents with clinical symptoms such as rapid enlargement, tenderness, and facial weakness.
CT and MRI are very useful tools in the evaluation of tumor localization and in the differentiation between benign and malignant tumors. CT findings of PA show a well–circumscribed ovoid or lobulated mass with homogeneous or heterogeneous enhancement, and intratumoral calcification may occasionally be seen. MRI findings of PA are characterized by an ovoid or lobulated, well–defined mass with hypointensity on T1–weighted MR images, homogeneous or heterogeneous hyperintensity on T2–weighted MR images, and well enhancement on contrast–enhanced T1–weighted MR images [7]. In addition, a gradual enhancement pattern of the time–signal intensity curve on dynamic MRI may aid the imaging–based diagnosis of PA [8].
Ultrasonography–guided FNAC is a minimally invasive procedure that provides valuable information for the preoperative histopathological evaluation of APG tumors [5,9]. However, care should be taken to rule out the possibility of false negative FNAC results, because it is often difficult to distinguish between PA and a low–grade malignancy such as low–grade mucoepidermoid carcinoma, which is the most common malignancy occurring in the APG, using FNAC [5]. Carcinoma ex PA should also be considered [10]. Therefore, the surgical strategy for an APG tumor should be determined based on the results of intraoperative pathological examination using frozen sections [5,11].
In the management of PA of the APG, complete resection of the tumor with a margin of normal salivary tissue without damaging the fibrous capsule is recommended to avoid tumor recurrence. Since an APG tumor is located near the facial nerve branches (especially the zygomatic and buccal branches) and the Stensen duct, care should be taken to avoid damage to these structures. Four surgical approaches have established thus far: 1) a standard parotidectomy incision or a facelift incision; 2) an intraoral approach; 3) a direct skin incision; and 4) an endoscopic approach [4]. Of these, a standard parotidectomy incision has been the most commonly used approach for complete tumor excision and prevention of facial nerve and Stensen duct injury, regardless of whether the tumor is benign or malignant. Furthermore, the facelift approach provides more favorable cosmetic results [2,12]. The only disadvantage of this approach may be a long skin incision. The intraoral approach has the advantage of no skin incision on the face, but it cannot be used when the tumor is large. Although there is risk of facial nerve injury, Schmutzhard et al. reported successful preservation of facial nerve function during intraoral APG tumor resection by facial nerve monitoring [13]. Direct skin incision over the tumor is not recommended due to the risk of facial nerve and Stensen duct injury and incomplete tumor resection [2]. The short skin incision may be the best advantage of this approach, but a wound in the mid–cheek region is cosmetically undesirable. A recently introduced endoscopic approach for benign APG tumors yields satisfactory results both from the viewpoint of complete tumor resection and a smaller wound [14,15]. This endoscopic technique could also be applicable for low–grade and small malignancies. The decision on the surgical approach should be made after understanding the benefits as well as risks.
In conclusion, an APG tumor should be considered a differential diagnosis in a patient with a mid–cheek mass. An APG tumor may be suspected to be PA when the above–described typical clinical manifestations and imaging findings are present. However, as the incidence of malignant APG tumorsis reportedly as high as 26–50% [2,4], malignancy cannot be ruled out even when preoperative diagnosis suggests a benign pathology.
References
- Johnson FE, Spiro RH. Tumors arising in accessory parotid tissue. Am J Surg. 1979; 138: 576-578.
- Ramachar SM, Huliyappa HA. Accessory parotid gland tumors. Ann Maxillofac Surg. 2012; 2: 90-93.
- Perzik SL, White IL. Surgical management of preauricular tumors of the accessory parotid apparatus. Am J Surg. 1966; 112: 498-503.
- Newberry TR, Kaufmann CR, Miller FR. Review of accessory parotid gland tumors: pathologic incidence and surgical management. Am J Otolaryngol. 2014; 35: 48-52.
- Yang X, Ji T, Wang LZ, Yang WJ, Hu YJ, Zhong LP, et al. Clinical management of masses arising from the accessory parotid gland. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011; 112: 290-297.
- Iguchi H, Wada T, Yamamoto H, Yamada K, Matsushita N, Okamoto S, et al. [Clinical features of accessory parotid gland tumors]. Nihon Jibiinkoka Gakkai Kaiho. 2013; 116: 1300-1307.
- Ikeda K, Katoh T, Ha-Kawa SK, Iwai H, Yamashita T, Tanaka Y. The usefulness of MR in establishing the diagnosis of parotid pleomorphic adenoma. AJNR Am J Neuroradiol. 1996; 17: 555-559.
- Yabuuchi H, Matsuo Y, Kamitani T, Setoguchi T, Okafuji T, Soeda H, et al. Parotid gland tumors: can addition of diffusion-weighted MR imaging to dynamic contrast-enhanced MR imaging improve diagnostic accuracy in characterization? Radiology. 2008; 249: 909-916.
- Klotz DA, Coniglio JU. Prudent management of the mid-cheek mass: revisiting the accessory parotid gland tumor. Laryngoscope. 2000; 110: 1627-1632.
- Tamiolakis D, Chimona TS, Georgiou G, Proimos E, Nikolaidou S, Perogamvrakis G, et al. Accessory parotid gland carcinoma ex pleomorphic adenoma. Case study diagnosed by fine needle aspiration. Stomatologija. 2009; 11: 37-40.
- Iguchi H, Wada T, Matsushita N, Oishi M, Teranishi Y, Yamane H, et al. Adenocarcinoma, NOS of the Accessory Parotid Gland. Dentistry, Oral Disorders & Therapy. 2013; 1: 02.
- Lin DT, Coppit GL, Burkey BB, Netterville JL. Tumors of the accessory lobe of the parotid gland: a 10-year experience. Laryngoscope. 2004; 114: 1652-1655.
- Schmutzhard J, Schwentner IM, Andrle J, Gunkel AR, Sprinzl GM. Resection of accessory parotid gland tumors through a peroral approach with facial nerve monitoring. J Craniofac Surg. 2007; 18: 1419-1421.
- Li B, Zhang L, Zhao Z, Shen G, Wang X. Minimally invasive endoscopic resection of benign tumours of the accessory parotid gland: an updated approach. Br J Oral Maxillofac Surg. 2013; 51: 342-346.
- Xie L, Zhang D, Lu MM, Gao BM. Minimally invasive endoscopic-assisted resection of benign tumors in the accessory parotid gland: 5 case studies. Head Neck. 2012; 34: 1194-1197.