Research Article
Austin J Nutri Food Sci. 2014;2(3): 1018.
The Wolfberry Bud Tea has Enhanced Antioxidant Activities
Xueyan Fu1, Yu Huang1, Xiaolan Kang1, Tingting Li1, Xin Jia1, Lin Dong1, Lei Wu2, Hui He2 and Dingbo Lin2*
1Ningxia Engineering and Technology Research Center for Modernization of Hui Medicine,, Ningxia Medical University, China
2Department of Nutritional Sciences, Oklahoma State University, USA
*Corresponding author: :Dingbo Lin, Department of Nutritional Sciences, Oklahoma State University, 301 HSCI, Stillwater, OK 74078, USA
Received: February 19, 2014; Accepted: March 05, 2014; Published: March 13, 2014
Abstract
Wolfberry bud tea and leaf tea have been consumed as anti–aging drinks in China and Far East Asia for years. However, the capacity against oxidative stress is not well documented. In this study, the total phenolics and flavonoids were extracted with 55 % ethanol and the contents were measured in samples of three wolfberry bud tea and three wolfberry leaf tea, respectively. The antioxidant activity were characterized by the FRAP and DPPH assays. The results showed that contents of total phenolics and flavonoids and the antioxidant activity were higher in the wolfberry bud tea than those in the wolfberry leaf tea. The antioxidant activity in wolfberry bud tea was significantly correlated with the contents of total phenolics and flavonoids. Data suggested that the wolfberry bud tea may serve as a significant source of natural flavonoids and ⁄or phenolics against oxidative stress.
Keywords: Antioxidant activity; Flavonoids; Phenolics; Wolfberry bud tea.
Introduction
Wolfberry, Chinese name goji berry, is a common name of two closely related species, Lycium barbarum Mill and L. chinense Mill., both in the family Solanaceae. Wolfberry is native to Asia and southeastern Europe. It has also been consumed as a fruit type of food in Indian tribes in the States of New Mexico and Arizona in the United States [1,2]. Wolfberry fruits and leaves are rich in phenolics, carotenoids, and other nutrients [3–7]. According to traditional Chinese medicine literature, wolfberry fruits can nourish liver and kidney, help re–balance of energy homeostasis, boost immune system, and improve vision through regulation of AMP–activated protein kinase and mitochondrial biogenesis [3,8–10].
The wolfberry bud and⁄or tea leaf has been consumed in China for over 2000 years. They have been proposed to be beneficial to stamina improvement, tranquillizing, thirst–quenching and anti–aging. However, how the bioactive constituents of the wolfberry bud tea exert their influence on oxidative stress is not well understood [11]. Recent in vitro studies show that wolfberry leaf hydrolysates promote growth of probiotic cells [12]. The leaf powder coincidently lowers body weight and the levels of serum triglyceride and LDL–cholesterol in obese rats [13]. Addition of methanol extracts of wolfberry leaves enhances the overall antioxidant activities in yogurt [14]. Yet, there is no report on characterization of the wolfberry bud tea. Here we determined contents of total phenolics and flavonoids, and in vitro antioxidant properties of selected wolfberry bud and⁄or leaf tea. The result showed that the wolfberry bud tea had a higher antioxidant activity than that of the wolfberry leaf tea, which positively correlatedwith the contents of phenolics and flavonoids.
Materials and Methods
The sources of wolfberry tea
Lycium chinense Mill. (wolfberry) bud tea and leaf tea were purchased from local grocery stores in Yinchuan, China. Three bud tea samples are from the Ninganbao Local Specialty Company (sample # 1), the Ningxia Lycium Bud Food Industry (sample #2), and the Ningxia Yongfuyuan Industrial and Trading Company (sample # 3), respectively. Wolfberry leaf tea samples were from the Yinchuan Activity Subsidiary Agricultural Products Company (sample # 4), the Ningxia Yongfuyuan Industrial and Trading Company (sample #5), and Ningxia Huixiangbao Local Specialty Company (sample #6). Tea samples were grounded. Fine powders were collected through a 0.38mm screen and then stored for further experiments at + 4°C.
Chemicals and reagents
All chemicals and reagents used were purchased from the Ningxia Huixiangbao Local Specialty Company, Yinchuan, Ningxia, China.
Ethanol extraction of total phenolics and flavonoids
One gram of tea powder was mixed with 40 mL 55 % ethanol (v⁄v) and then subjected to ultrasonic extraction twice at 45oC for 30 min of each. The mixture was filtered, and aqueous phase, e.g., the fraction of total phenolics and flavonoids, was combined, and the final volume was brought up to 100 mL with 55 % ethanol for further analysis.
Determination of total phenolic content
Total phenolic content was determined according to the Folin– Ciocalteu method (FCM) [3,15]. Note, the FCM is an electron transfer based assay and measures a sample's reducing capacity, which is expressed as phonelic contents [16,17]. One hundred µl wolfberry tea extracts were mixed with 500 µl Folin–Ciocalteu's reagent and 2 mL distilled water, and incubated at room temperature for 5min. After that, 1.5 ml 20 % Na2CO3 [v⁄v] and 900 µl distilled water were added. The mixture was incubated for 30 min at 40°C. Then the absorbance was measured at 760 nm using a spectrophotometer. Results were expressed as mg gallic acid equivalent per gram wolfberry tea.
Measurement of flavonoid content
Total flavonoid content was determined as described previously with minor modification [18]. Briefly, 200 µl wolfberry tea extracts were mixed with 1 mL 55 % ethanol [v⁄v] and 200 µl 5 % Na2NO2 for 6 min. After addition of 300 µl 10 % AlCl3, the mixture was incubated at room temperature with constant shaking for 6 min. The reaction was stopped by adding 1 mL 1 M NaOH. OD494 was detected using a spectrophotometer. Results were expressed as mg rutin equivalent per gram wolfberry tea.
Free radical scavenging activity of wolfberry bud and⁄or leaf tea
Free radical scavenging activity was measured by the 1,1–diphenyl– 2–picrylhydrazyl [DPPH] assay as described [19]. In brief, 20 µl wolfberry tea extracts [referred as a sample] were mixed with 5 mL DPPH solution [referred as a control] in methanol and incubated in dark at room temperature for 1 hr. Then OD515 was measured. The percentage of free radical scavenging activity was calculated according to equation:
DPPH radical scavenging rate (%) = [Acontrol–Asample] ⁄Acontrol] x100%
Ferric reducing activity of plasma (FRAP) assay
The antioxidant power of wolfberry tea extracts was determined by a FRAP assay [20]. The working FRAP reagent was prepared by mixing 10 volumes of 300 mM acetate buffer [pH 3.6], with 1 volume of 10 mM TPTZ [2,4,6–tri(2–pyridyl)–s–triazine] in 40 mM hydrochloric acid and with 1 volume of 20 mM ferric chloride, and was pre–warmed at 37 °C. Twenty µL wolfberry tea extracts was mixed with 4980 µL new prepared FRAP reagent, and incubated at 37 °C for 10 min. In the end, the absorbance of the samples was measured at 593 nm. Trolox was used as a standard. The FRAP value was calculated against a Trolox standard calibration curve.
Statistics analysis
The results were analyzed by one–way ANOVA. Regression analysis was done using SAS 9.1. Experiments were at least triplicate. The level of significance was considered at p ≤ 0.05, and marked by different letters. All data are mean ± S.D.
Results and Discussion
The wolfberry bud tea has higher contents of total phenolics and flavonoids than those in the wolfberry leaf tea
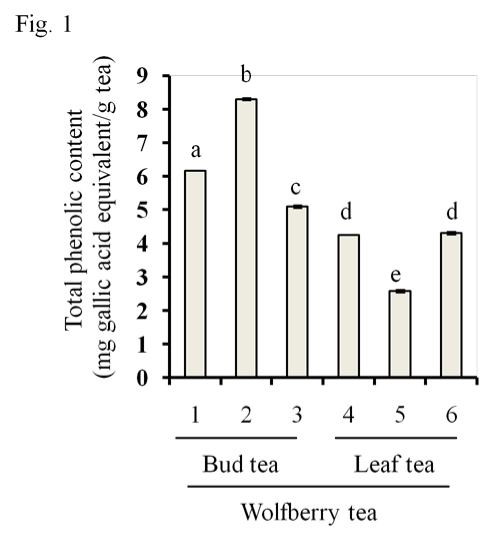
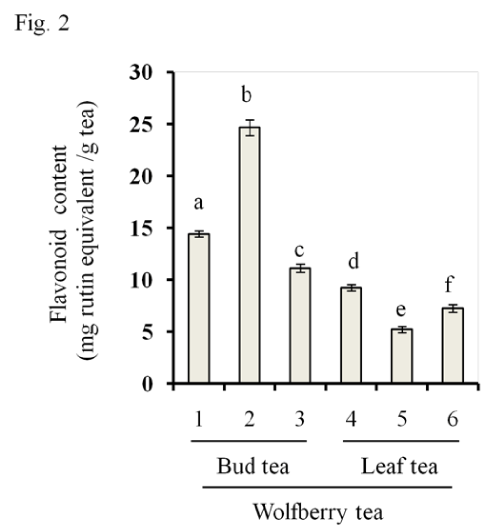
As shown in Figure 1 and 2, all three bud tea samples contained significantly greater amounts of total phenolics and flavonoids than three leaf tea samples [Figure. 1, total phenolics, Figure. 2, total flavonoids]. Wolfberry bud tea sample #2 had highest contents of both phenolics and flavonoids than other 2 bud tea samples (#1 and #3). No significant difference was found between leaf tea samples #4 and #6. The amounts of total phenolics and⁄or flavonoids in wolfberry leaf tea were comparable to other tea products as reported [21–23]. We profiled the compositions of the extracts by HPLC (data not shown), the results showed that quercetin and the glycosides such as rutin are predominant in the wolfberry tea extract, which were in consistent with previous reports [24,25]. We reported that “Zhongning” wolfberry fruits contained total phenolics at 7.8+0.4 mg quercetin equivalent⁄g fruits [3], indicating that the wolfberry bud tea had as a high content of total phenolics as that of the wolfberry fruits.
Figure 1: Contents of total phenolics in wolfberry tea. Total phenolics were extracted with 55 % ethanol from the wolfberry bud tea (Samples # 1–3) and leaf tea (Samples #4–6), respectively. Total phenolic content was expressed as mg gallic acid equivalent per gram tea. The statistic significance at p≤0.05 was marked by different letters.
Figure 2: Flavonoids in wolfberry tea. Flavonoids were extracted with 55 % ethanol from wolfberry bud tea (Samples # 1–3) and leaf tea (Samples #4–6). Flavonoid content was expressed as mg rutin equivalent per gram tea. The statistic significance at p≤0.05 was marked by different letters.
Antioxidant activity
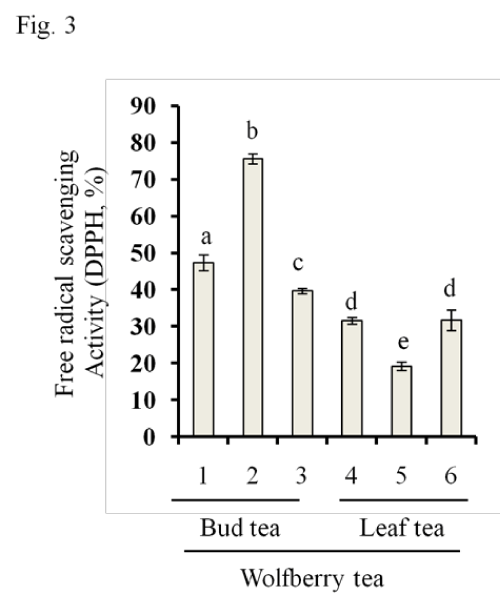
Two methods were applied to determine antioxidant activity: the DPPH assay to determine a free radical scavenging activity and the FRAP assay to determine a ferric reducing power of wolfberry tea extracts. As shown in Figure 3, the wolfberry bud tea sample #2 had highest radical scavenging activity, followed by samples # 1, and #3. Lowest activity was observed in leaf tea sample #5. No significant difference was found between samples #4 and #6.
Figure 3: Free radical scavenging activity in wolfberry tea. Free radical scavenging activity was determined by DPPH assay as described in the section of Materials and Methods . The results from wolfberry bud tea (Samples # 1–3) and leaf tea (Samples #4–6) were shown. The statistic significance at p≤0.05 was marked by different letters.
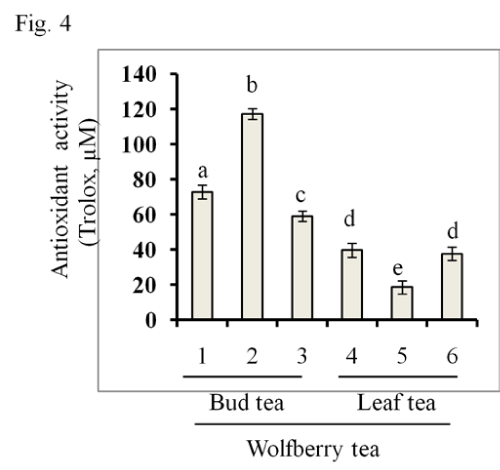
The wolfberry tea antioxidant activity was also investigated by FRAP assay as shown in Figure 4. Similar to the results from the DPPH assay, bud tea samples had higher antioxidant activities than leaf tea samples. The highest antioxidant activity was also found in sample #2, and the lowest one was sample #5.
Figure 4: Antioxidant activity in wolfberry tea. Antioxidant activity was determined by FRAP assay as described in the section of Materials and Methods. The results from wolfberry bud tea (Samples # 1–3) and leaf tea (Samples #4–6) were shown. The statistic significance at p≤0.05 was markedby different letters.
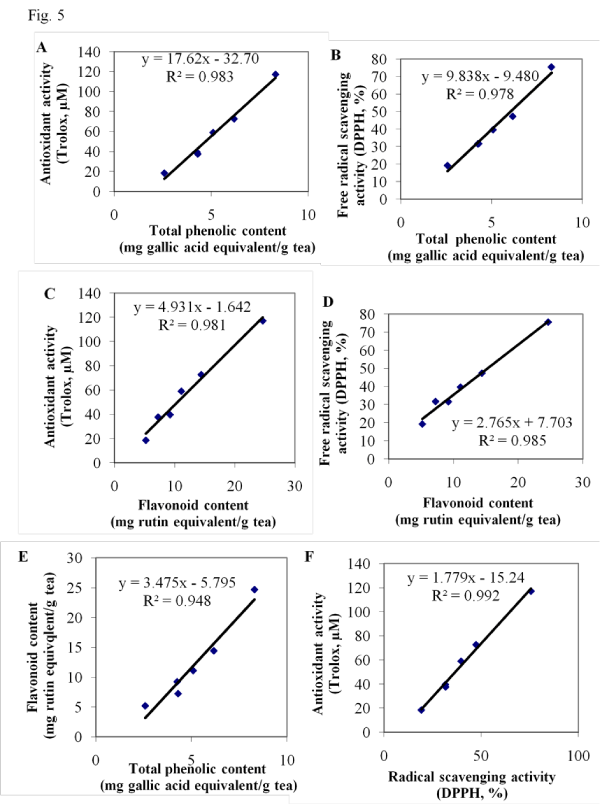
Positive Correlation between the antioxidant activity and contents of flavonoids and⁄or total phenolics in wolfberry tea
Linear regression analysis results revealed that antioxidant activity was positively correlated with contents of both total phenolics and flavonoids in wolfberry bud and leaf tea [Figure 5 A, B, C, D]. The total phenolic content was highly correlated with the flavonoid content [Figure 5 E]. The antioxidant activity results determined by different methods [FRAP and DPPH] were correlated [Figure 5F], which were in good consistent with previous reports [21,26].
Figure 5: Positive correlation between the antioxidant capacity and contents of flavonoids and⁄or total phenolics in the wolfberry tea. Linear regression analysis was conducted in antioxidant activity (FRAP assay) and total phenolics (A), free radical scavenging activity and total phenolics (B), antioxidant activity (FRAP assay) and flavonoids (C), free radical scavenging activity and flavonoids (D), flavonoids and total phenolics (E), and antioxidant activity (FRAP assay) and free radical scavenging activity (F).
Potential bioactive components and the antioxidant activities have been studied in many types of tea products. The benefits of flavonoids and ⁄or phenolics to human health are well understood. In our study, we demonstrated that the antioxidant properties in the wolfberry leaf tea which were comparable to other tea product. However, the wolfberry bud tea had a significantly higher antioxidant activity than that of the wolfberry leaf tea. The contents of total phenolics and flavonoids were directly correlated with the antioxidant capacity in wolfberry bud and⁄or leaf tea. The difference in phenolics, flavonoids, and⁄or antioxidant activity among samples may reflect various wolfberry plant growth conditions, for example, fertilizer, soil, and the weather conditions (e.g. light, water, temperature). Thus, the establishment of sound culture standards for the effective quality control of wolfberry tea is critical to the tea industry.
Conclusion
flavonoids and ⁄or phenolics to human health are well understood. In our study, we demonstrated that the antioxidant properties in the wolfberry leaf tea which were comparable to other tea product. However, the wolfberry bud tea had a significantly higher antioxidant activity than that of the wolfberry leaf tea. The contents of total phenolics and flavonoids were directly correlated with the antioxidant capacity in wolfberry bud and⁄or leaf tea. The difference in phenolics, flavonoids, and⁄or antioxidant activity among samples may reflect various wolfberry plant growth conditions, for example, fertilizer, soil, and the weather conditions (e.g. light, water, temperature). Thus, the establishment of sound culture standards for the effective quality control of wolfberry tea is critical to the tea industry.
References
- Flint HL. Lycium barbarum. Landscape plants for eastern North America: exclusive of Florida and the immediate Gulf Coast. Chilchester: John Wiley & Sons.;1997.
- Carpenter TM, Steggerda M. The food of the present-day Navajo Indians of New Mexico and Arizona. J Nutr. 1939; 18: 297-305.
- Tang L, Zhang Y, Jiang Y, Willard L, Ortiz E. Dietary wolfberry ameliorates retinal structure abnormalities in db/db mice at the early stage of diabetes. Exp Biol Med (Maywood). 2011; 236: 1051-1063.
- Fraser PD, Bramley PM. The biosynthesis and nutritional uses of carotenoids. Prog Lipid Res. 2004; 43: 228-265.
- Le Marchand L. Cancer preventive effects of flavonoids--a review. Biomed Pharmacother. 2002; 56: 296-301.
- Sheng JC, Yu F, Xin ZH, Zhao LY, Zhu XJ, et al. Preparation, identification and their antitumor activities in vitro of polysaccharides from Chlorella pyrenoidosa. Food Chem. 2007; 105:533-539.
- Yang X, Zhao Y, Yang Y, Ruan Y. Isolation and characterization of immunostimulatory polysaccharide from an herb tea, Gynostemma pentaphyllum Makino. J Agric Food Chem. 2008; 56: 6905-6909.
- Ho Y-S, So K-F, Chang R C-C. Anti-aging herbal medicine-how and why can they be used in aging-associated neurodegenerative diseases? Ageing Res Rev. 2010; 9:354-362.
- Lin D, He H, Ji H, Willis J, Willard L, et al. Wolfberries potentiate mitophagy and enhance mitochondrial biogenesis leading to prevention of hepatic steatosis in obese mice: the role of AMP-activated protein kinase a2 subunit. Mol Nutr Food Res. 2013.
- Yu H, Wark L, Ji H, Willard L, Jaing Y. Dietary wolfberry upregulates carotenoid metabolic genes and enhances mitochondrial biogenesis in the retina of db/db diabetic mice. Mol Nutr Food Res. 2013; 57: 1158-1169.
- Kim SY, Lee KH, Chang KS, Bock JY, Jung MY. Taste and flavor compounds in box thorn (Lycium chinense Miller). Food Chem. 1997; 58:297-303.
- Yeh YC, Hahm TS, Sabliov CM, Lo YM. Effects of Chinese wolfberry (Lycium chinense P. Mill.) leaf hydrolysates on the growth of Pediococcus acidilactici. Bioresour Technol. 2008; 99: 1383-1393.
- Kang MH, Park WJ, Choi MK. Anti-obesity and hypolipidemic effects of Lycium chinense leaf powder in obese rats. J Med Food. 2010; 13: 801-807.
- Bae HC, Cho IS, Nam MS. Effects of the biological function of yogurt added with Lycium chinense Miller extract. J Korean Soc Anim Sci Technol. 625.
- Singleton VL, Orthofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. In: Methods in Enzymology, Oxidant and Antioxidants (Part A) (Packer L, ed), pp152-178. San Diego: Academic Press.
- Prior RL, Wu X, Schaich K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J Agric Food Chem. 2005; 53: 4290-4302.
- Huang D, Ou B, Prior RL. The chemistry behind antioxidant capacity assays. J Agric Food Chem. 2005; 53: 1841-1856.
- Sahreen S, Khan MR, Khan RA. Evaluation of antioxidant activities of various solvent extracts of Carissa opaca fruits. Food Chem 2010; 122:1205-1211.
- Meghashri S, Vijay Kumar H, Gopal S. Antioxidant properties of a novel flavonoid from leaves of Leucas aspera. Food Chem. 2010; 122: 105-110.
- Benzie IF, Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of "antioxidant power": the FRAP assay. Anal Biochem. 1996; 239: 70-76.
- Seeram NP, Henning SM, Niu Y, Lee R, Scheuller HS. Catechin and caffeine content of green tea dietary supplements and correlation with antioxidant capacity. J Agric Food Chem. 2006; 54: 1599-1603.
- Duh PD, Yen GC, Yen WJ, Wang BS, Chang LW. Effects of pu-erh tea on oxidative damage and nitric oxide scavenging. J Agric Food Chem. 2004; 52: 8169-8176.
- Seeram NP, Aviram M, Zhang Y, Henning SM, Feng L. Comparison of antioxidant potency of commonly consumed polyphenol-rich beverages in the United States. J Agric Food Chem. 2008; 56: 1415-1422.
- Duan H, Chen Y, Chen G. Far infrared-assisted extraction followed by capillary electrophoresis for the determination of bioactive constituents in the leaves of Lycium barbarum Linn. J Chromatogr A. 2010; 1217: 4511-4516.
- Dong JZ, Lu da Y, Wang Y. Analysis of flavonoids from leaves of cultivated Lycium barbarum L. Plant Foods Hum Nutr. 2009; 64: 199-204.
- Anesini C, Ferraro GE, Filip R. Total polyphenol content and antioxidant capacity of commercially available tea (Camellia sinensis) in Argentina. J Agric Food Chem. 2008; 56: 9225-9229.